Collisions between electrons and atoms Your Specification Says

Collisions between electrons and atoms Your Specification Says: Collisions of electrons with atoms The electron volt. Ionisation and excitation; understanding of ionization and excitation in the fluorescent tube.
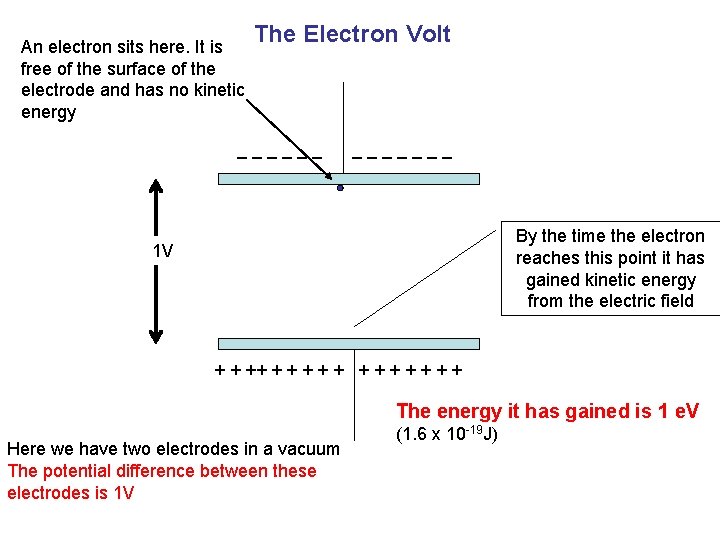
An electron sits here. It is free of the surface of the electrode and has no kinetic energy The Electron Volt _______ By the time the electron reaches this point it has gained kinetic energy from the electric field 1 V + + + + The energy it has gained is 1 e. V Here we have two electrodes in a vacuum The potential difference between these electrodes is 1 V (1. 6 x 10 -19 J)

THE ELECTRON VOLT The electron volt is defined as “the energy acquired by an electron when it is accelerated through an electrical potential difference of 1 V” Learn this definition

The Gas Discharge Tube A “gas-discharge tube” is just a tube containing a gas at low pressure. There are two electrodes in the tube an anode (+) and a cathode(-) A high potential difference is set up between the electrodes to give the electrons enough energy to emerge from the cathode and accelerate towards the anode cathode + Collisions between these accelerating electrons and the gaseous atoms in the tube give rise to the emission of light

Collisions With Atoms The accelerating electrons collide with atoms in the gas causing ionizations. That is electrons are ejected from the gas. Other electrons fall into the vacated levels to take their place. It is this process that creates an emission spectrum.

Collisions With Atoms The accelerating electrons collide with atoms in the gas causing ionizations. That is electrons are ejected from the gas. Other electrons fall into the vacated levels to take their place. It is this process that creates an emission spectrum. Many transitions occur producing a range of lines at different frequencies within the emitted spectrum

The Fluorescent Tube A fluorescent tube used for lighting has the air removed and a noble gas at very low pressure (0. 3%) of atmospheric pressure. Noble gases are unreactive so unwanted chemical reactions like oxidation of the electrodes is prevented anode . Mercury vapour is also introduced into the tube cathode + The mercury vapour undergoes excitation in the manner described emitting electromagnetic radiation much of which is in the ultraviolet region of the spectrum. _
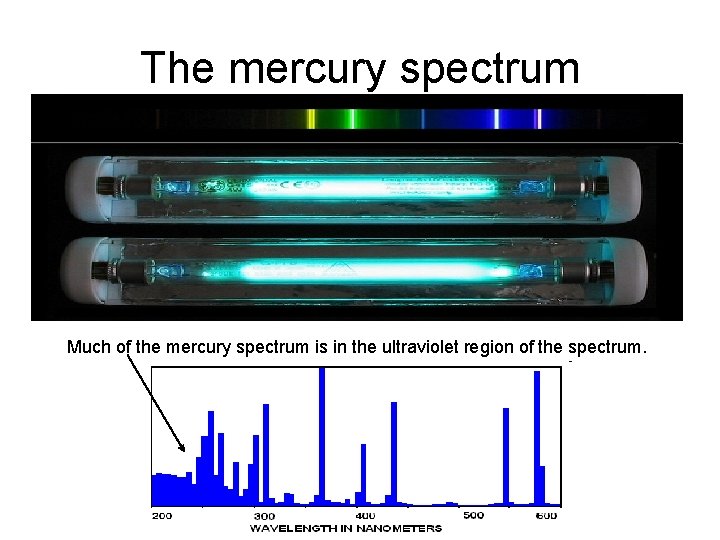
The mercury spectrum Much of the mercury spectrum is in the ultraviolet region of the spectrum.

Producing Visible Light • Most of the energy of a fluorescent lighting tube is emitted as ultraviolet light which is useless for lighting purposes. • For this reason the inside of a fluorescent tube is coated in a material called a “phosphor”. • Phosphors absorb ultra violet energy and reemit the energy at visible frequencies

Fluorescent tubes coated in phosphors
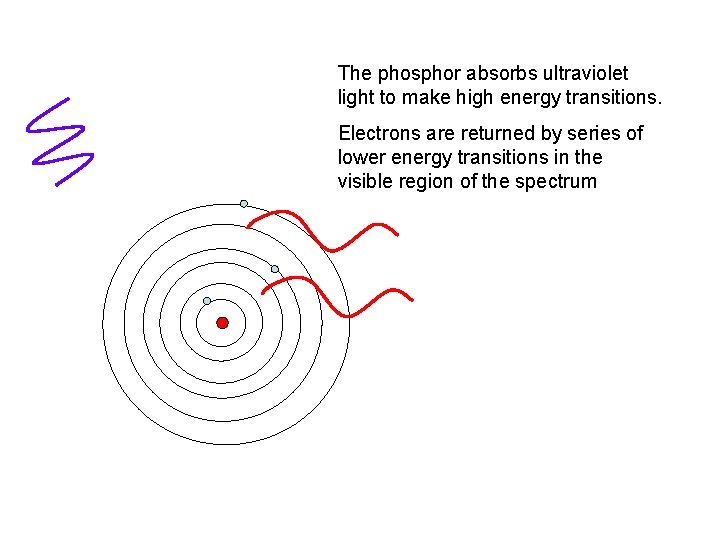
The phosphor absorbs ultraviolet light to make high energy transitions. Electrons are returned by series of lower energy transitions in the visible region of the spectrum
- Slides: 11