Colligative Properties Does a solute change the properties





















- Slides: 22

Colligative Properties

Does a solute change the properties of a solvent?

Consider aqueous solutions • Solvent = water How do properties of aqueous solution compare to properties of pure water?

Conductivity • Pure water does not conduct electricity – Some aqueous solutions do • To conduct a solution must contain ions

• Conductivity depends on nature of the particle – molecular (covalent) solutes DO NOT conduct – ionic solutes DO conduct

Colligative Properties • Depend on number particles in solution not type particles

Colligative Properties – doesn’t matter what kind of particles (ions or molecules) – Concentration of particles does matter

What are some colligative properties? • Vapor Pressure – decreases (↓) • Freezing Point – decreases (↓) • Boiling Point – increases (↑) • change in Osmotic Pressure

The higher the concentration of solute particles, the larger the effect on colligative properties

Does it matter if solute is ionic or covalent? YES • number of particles will vary • effected by concentration: • greater concen = greater effect • lower concen = lesser effect

Ionic solutes dissolve & dissociate into (+) & (-) ions more particles per mole of solute

Mg. Cl 2(s) + H 2 O(l) Mg 2+(aq) + 2 Cl(aq) 1 mole salt 3 moles ions Na. Cl(s) + H 2 O(l) Na+1(aq) + Cl-1(aq) 1 mole salt 2 moles ions

Covalent solutes dissolve as molecules 1 -to-1 relationship between moles of solute & moles of dissolved particles

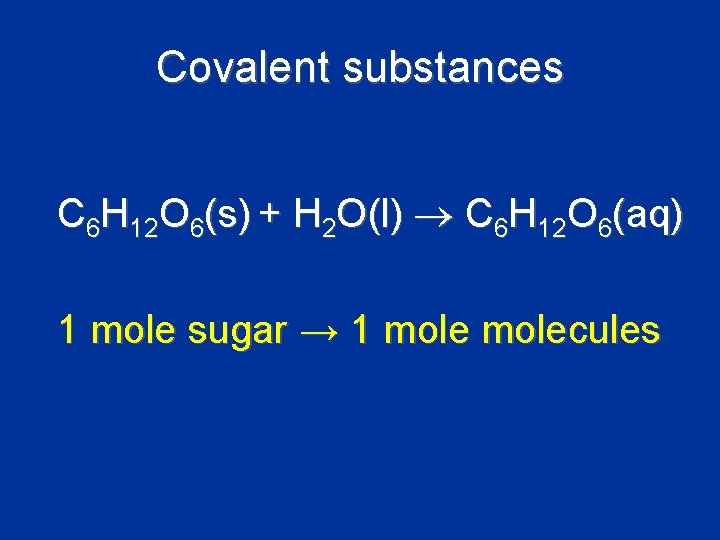
Covalent substances C 6 H 12 O 6(s) + H 2 O(l) C 6 H 12 O 6(aq) 1 mole sugar → 1 molecules

As solute is added to a solvent, what happens to freezing point & boiling point of solution? 1) The freezing point ↓ & the boiling point ↓ 2) The freezing point ↓ & the boiling point ↑ 3) The freezing point ↑ & the boiling point ↓ 4) The freezing point ↑ & the boiling point ↑ Answer: 2

Which solution containing 1 mole of solute dissolved in 1000 g of water has the lowest freezing point? 1) C 2 H 5 OH(aq) 2) Na. Cl(aq) Be careful! - do you want greatest or least change? 3) KOH What if question asked which solution has highest freezing point? 4) Ca. Cl 2 Answer: 4

Of the following solutions, the one that will freeze at the lowest temperature contains 1 mole of nonvolatile solute dissolved in 1) 2) 3) 4) 250 g of solvent 500 g of solvent 750 g of solvent 1000 g of solvent Answer: 1

Which solute, when added to 1000 g of water, will produce a solution with the highest boiling point? 1) 29 g of Na. Cl 2) 58 g of Na. Cl 1 mole of Na. Cl → 2 moles of ions 3) 31 g of C 2 H 6 O 2 4) 62 g of C 2 H 6 O 2 1 mole of C 2 H 6 O 2 → 1 mole of molecules

Which solution will freeze at the lowest temperature? 1) 1 g of Na. Cl dissolved per 100 g of H 2 O 2) 1 g of Na. Cl dissolved per 1000 g of H 2 O 3) 1 g of C 12 H 22 O 11 dissolved per 100 g of H 2 O 4) 1 g of C 12 H 22 O 11 dissolved per 1000 g of H 2 O

Which solution will freeze at the lowest temperature? 1) 1 g of Na. Cl dissolved per 100 g of H 2 O __1 g____ 58. 8 g/mol = 0. 017 mol → 0. 034 mol of ions (Na. Cl) (Na+1 and Cl-1) 2) 1 g of C 12 H 22 O 11 dissolved per 100 g of H 2 O __1 g___ 232 g mol = 0. 0043 mol of molecules

Effect of Nonvolatile Solute • Boiling Point Elevation (↑) • Freezing Point Depression (↓) • The more particles, the bigger the effect!

Applications of colligative properties Salting roads in the winter time Using salted ice to make ice cream