Collaboration with FDA and NIH December 2017 NIH

Collaboration with FDA and NIH December 2017

NIH Enhancing Stewardship of Clinical Trials Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION *

NIH-FDA Protocol Template Initiative March 2015: NIH–FDA Joint Leadership Council agreed to jointly develop a clinical trial protocol template to improve quality of protocols received for review Goal of the template Provide a standard protocol format Provide instructions for writing a complete protocol Working group consisted of NIH and FDA staff NIH Program staff, FDA reviewers, IRB members, Statisticians, clinical operations, etc. Reviewed over 13 NIH protocol templates Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION * 3
Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION *

NIH-FDA Protocol Template Development February 2016: Draft phase 2 and 3 clinical trial protocol template posted for public comment. Numerous comments received from over 60 respondents and incorporated into revised template NIH-FDA Working Group met regularly with Trans. Celerate to harmonize format of templates Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION * 5
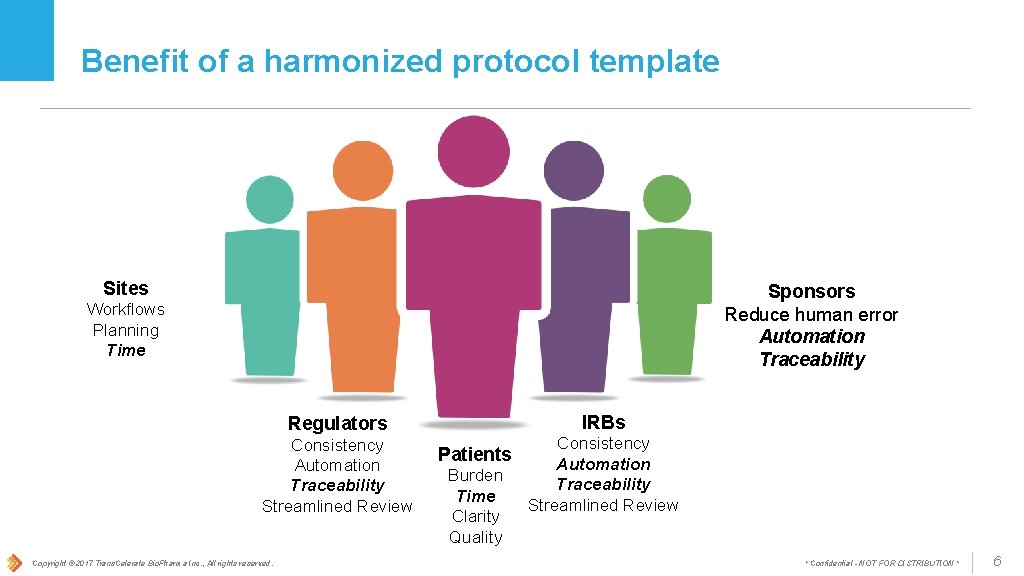
Benefit of a harmonized protocol template Sites Sponsors Reduce human error Automation Traceability Workflows Planning Time IRBs Regulators Consistency Automation Traceability Streamlined Review Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. Patients Burden Time Clarity Quality Consistency Automation Traceability Streamlined Review * Confidential - NOT FOR DISTRIBUTION * 6

CPT and NIH-FDA Templates: Where we started CPT Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. NIH-FDA * Confidential - NOT FOR DISTRIBUTION *

Review of Trans. Celerate CPT and NIH-FDA templates • Reviewed TOC and instructional content • Found many similarities • Used “best of” from each template • Learned from comments from public, IRBs and FDA • Agreed on flexibility within the template (2 nd & 3 rd level headings) • Resulted in significant updates from previous versions How did we do this and what did we find? Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION * 8

Off to a good start? CPT NIH-FDA 12 Section Headers, including Appendices Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. 17 Section Headers, plus Appendices * Confidential - NOT FOR DISTRIBUTION *
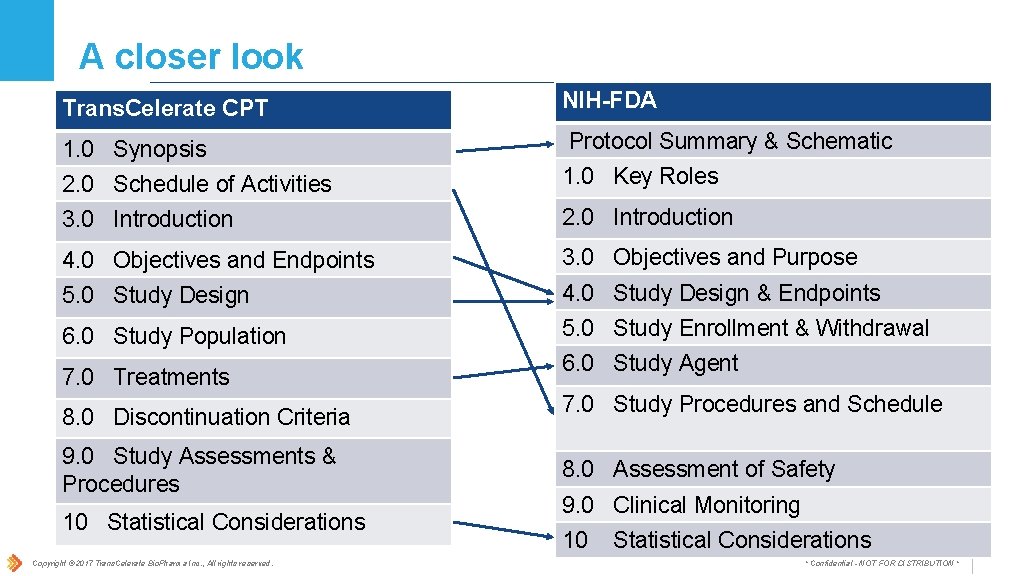
A closer look Trans. Celerate CPT NIH-FDA 1. 0 Synopsis Protocol Summary & Schematic 2. 0 Schedule of Activities 1. 0 Key Roles 3. 0 Introduction 2. 0 Introduction 4. 0 Objectives and Endpoints 3. 0 Objectives and Purpose 5. 0 Study Design 4. 0 Study Design & Endpoints 6. 0 Study Population 5. 0 Study Enrollment & Withdrawal 7. 0 Treatments 8. 0 Discontinuation Criteria 9. 0 Study Assessments & Procedures 10 Statistical Considerations Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. 6. 0 Study Agent 7. 0 Study Procedures and Schedule 8. 0 Assessment of Safety 9. 0 Clinical Monitoring 10 Statistical Considerations * Confidential - NOT FOR DISTRIBUTION *

Alignment = Organization + Prioritization + Semantics Harmonized Templates • Simplified TOC 1. 0 Protocol Summary • Moved sections and sub-sections: 2. 0 Introduction 3. 0 Objectives and Endpoints 4. 0 Study Design 5. 0 Study Population 6. 0 Study Intervention 7. 0 Discontinuation of SI & Ppt DC/Wd 8. 0 Study Assessments & Procedures 9. 0 Statistical Considerations 10 Supporting Doc & Oper. Consid. 11 References Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. – Prioritized sections for the user: investigator & reviewers • Moved Schedule of Activities to Protocol Summary • Harmonized Heading Titles – Treatment v. Study Agent = Study Intervention • Harmonized subsections – Introduction: Study Rationale, Background, Benefit/Risk Assessment * Confidential - NOT FOR DISTRIBUTION *

Best practices for intended users • Quality in protocol development • Site staff have one format • FDA & IRB reviewers have one format • Standards reduce error • Facilitates automation and re-use • Standard template supports a better INDs application • FDA modernizes workflow, extract data elements Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION * 12
NIH-FDA Phase 2 and 3 IND/IDE Clinical Trial Protocol Template Released May 2017 Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION *
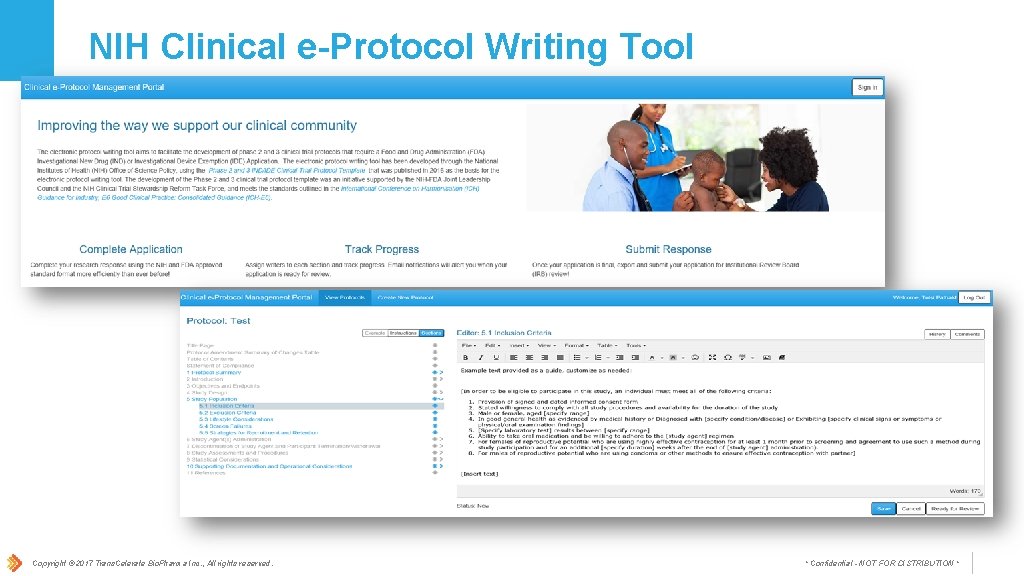
NIH Clinical e-Protocol Writing Tool Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION *

Future Plans for NIH-FDA Template • Develop behavioral intervention and phase 1 protocol templates using NIHFDA protocol format • Add behavioral and phase 1 protocol templates and instructions to electronic writing tool • Consider developing ability to integrate with Clinical. Trials. gov registration, IRB submissions, etc. Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION * 15

Work Group Members From Several Offices • Office of New Drugs (OND) • Office of Strategic Programs (OSP) • Office of Translational Sciences (OTS) • Office of Biostatistics (OB) • Office of Clinical Pharmacology (OCP) • Office of Computational Sciences (OCS) • Office of the Director (OD)/ Center for Biological Evaluation and Research (CBER) • Office of Cellular, Tissue and Gene Therapies (OCTGT)/ CBER Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION * 16
NIH-FDA Joint Protocol NIH FDA Michelle Culp Melissa Robb Cynthia Boucher David Cho Christine Cutillo Owen Faris Peter Kim Daniel Falk Shanda Finnigan Peter Marks Eileen Navarro. Adam Haim Almario Barbara Karp Petra Kaufmann Vaishali Popat Lynette Nieman Rachel Sherman Sarah Read Megan Ryan Galia Siegel Steven Sparenborg Betty Tai Aynur Unalp-Arida James Witter Anne Zajicek FDA CPT Working Group Vaishali Popat, OND IO Peter Kim, OND/OAP/DAIP Lisa Soule, OND/ODEIII/ DBRUP Martin Rose, OND/ODEI/DCRP Bob Temple, CDER/OCD Milena Lolic, CDER/OCD/PASE Ray Chiang, CDER/OMP Stephanie Shapley, CDER/OMP Melissa Robb, CDER/OMP Steve Wilson, OTS/OB Robert ONeill, CDER/OTS Sirisha Mushti, OTS/OB Che Smith, OTS/OB David Petullo, OTS/OB Lili Garrard, OTS/OB Ron Fitzmartin, CDER/OSP Ta-Jen Chen, CDER/OSP Jeff Florian, OTS/OCP Eileen Navarro Almario, OTS/OCS Daniela Vanco, CBER/OD Robert Sokolic, CBER/ OCTGT Trans. Celerate CPT Mitzi Allred, Merck Ralf Bilke, BI TK Booker, Amgen Elizabeth Bygate, GSK Susan Colby, BMS Vivian Combs, Lilly Rob Di. Cicco, GSK Robert Ferendo Nareen Katta, Abb. Vie Diane Klatzman, J&J Bill Lander, GSK Amy Leishman, Lilly Mark Makurath, Merck Frederik Malfait Virginia Nido, Roche Nils Schlote, Roche Cathy Stein-Izsak, Allergan Stacy Tegan, Accenture Provided comment separately on CPT: OSI, CDRH offices, OND/DPP (Mitchell Mathis) Copyright © 2017 Trans. Celerate Bio. Pharma Inc. , All rights reserved. * Confidential - NOT FOR DISTRIBUTION *

Thank you
- Slides: 18