Coal seq 2005 Frank van Bergen Sander Hol
Coal- seq 2005 Frank van Bergen, Sander Hol , Chris Spiers, Colin Peach t 1

Acknowledgements • This project was made possible by funds from the CATO project, Shell International and TNO. • The technical staff members of the Laboratory for High Pressure and Temperature Research of Utrecht University, especially Peter van Krieken and Gert Kastelein, are thanked for their valuable support and suggestions in the experiments and interpretation. • Harry Veld and Kathrin Reimer of TNO are acknowledged for their support and suggestions in the coal characterization and gas analyses. • The Central Mining Institute and the Brzeszcze mine in Poland Delft University of Technology are acknowledged for providing the coal samples t 2

Introduction • CO 2 sequestration in coal while producing (enhanced) coal bed methane (ECBM-CO 2) considered to be a niche option for CO 2 sequestration for those areas with large industrial sources and few sequestration alternatives • Critical factors • Permeability (swelling) • Exchange ratio at reservoir conditions • Field experiment ongoing to test the feasibility • Laboratory experiments required to understand the fundamental processes • This project aims at the integration of field and laboratory results! t 3
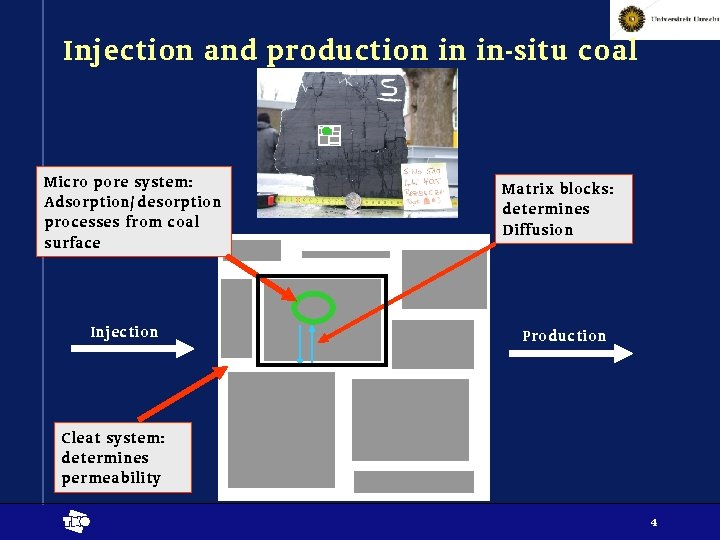
Injection and production in in-situ coal Micro pore system: Adsorption/ desorption processes from coal surface Injection Matrix blocks: determines Diffusion Production Cleat system: determines permeability t 4
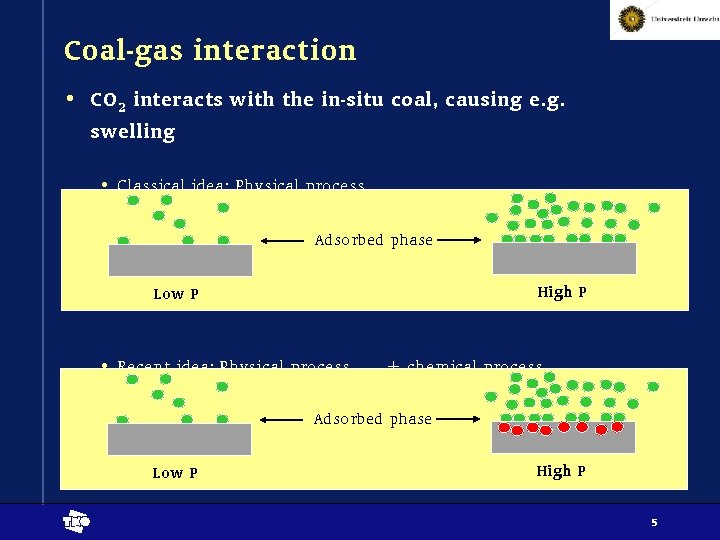
Coal-gas interaction • CO 2 interacts with the in-situ coal, causing e. g. swelling • Classical idea: Physical process Adsorbed phase High P Low P • Recent idea: Physical process + chemical process Adsorbed phase Low P t High P 5

Characteristics pure processes • Physisorption • Effects are largely reversible, partly irreversible • Coal structure can change mechanically • Effects are strongly P dependent • Absolute effects are P dependent (i. e. higher P, more swelling), e. g. by a Langmuir relation • (Negative) relation between P and reaction time • Chemisorption • Effects are largely irreversible, partly reversible • Coal structure changes chemically • Effects are concentration dependent, for a gas thus P dependent • Strong (negative) relation between concentration (P) and reaction time • Absolute effects are P independent • once the sample looses its reactivity, it becomes inert t 6
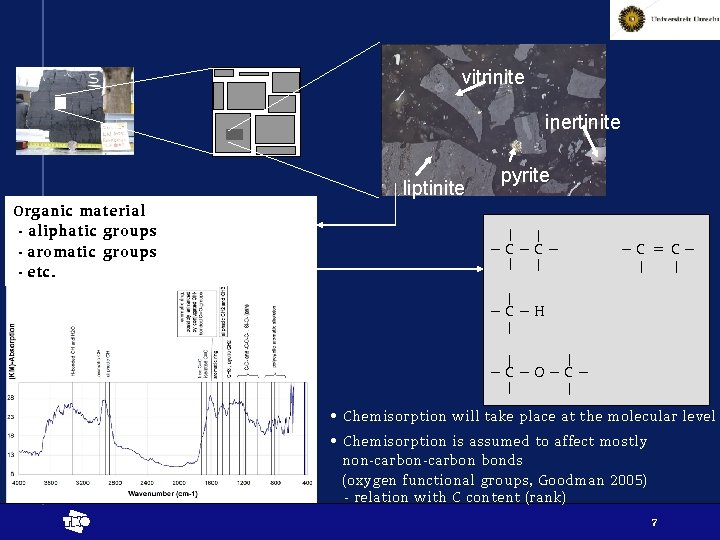
vitrinite inertinite liptinite Organic material - aliphatic groups - aromatic groups - etc. pyrite –C–C– –C=C– –C–H –C–O–C– • Chemisorption will take place at the molecular level • Chemisorption is assumed to affect mostly non-carbon bonds (oxygen functional groups, Goodman 2005) - relation with C content (rank) t 7

Laboratory Experiments Approach • Focused on • Registration of exerted stress after introduction of CO 2 • Determination of volumetric expansion of coal under the influence of CO 2 • Reversibility / irreversibility of expansion, possible indication of chemical processes t 8
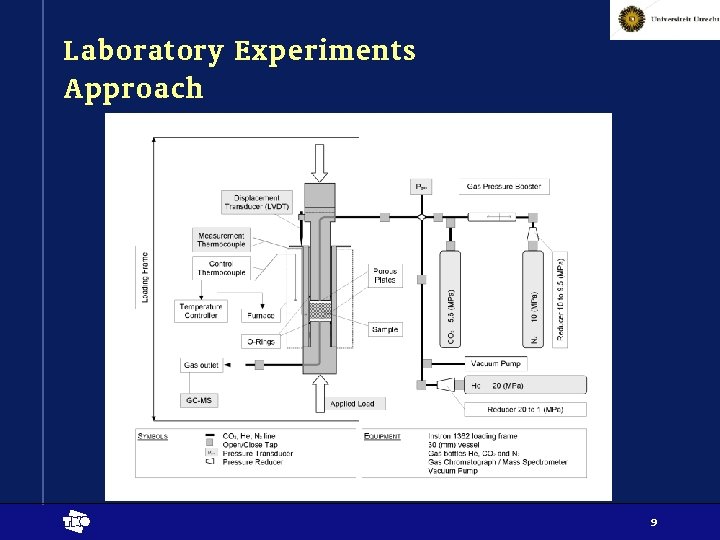
Laboratory Experiments Approach t 9

Laboratory Experiments Approach • Sample • Activated Carbon (reference material) • Low volatile bituminous coal (Germany) • High volatile bituminous coal (Poland) • Sample treatment • Dried and physically ‘homogenised’ • 63 -212 (µm), approx. 10 (gram) • Pre-compaction under vacuum at 65 (MPa) • He porosity circa 15 -20% • Gas • CO 2 • Helium (Reference gas) • Nitrogen (data still under evaluation) t 10
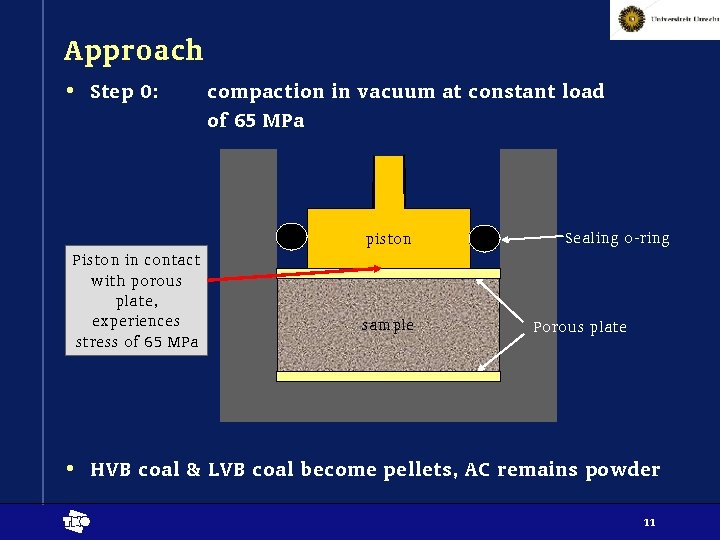
Approach • Step 0: compaction in vacuum at constant load of 65 MPa piston Piston in contact with porous plate, experiences stress of 65 MPa sample Sealing o-ring Porous plate • HVB coal & LVB coal become pellets, AC remains powder t 11
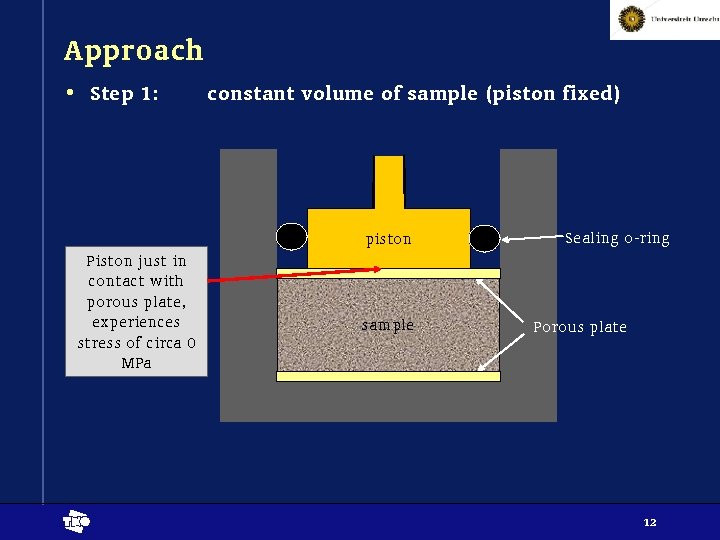
Approach • Step 1: constant volume of sample (piston fixed) piston Piston just in contact with porous plate, experiences stress of circa 0 MPa t sample Sealing o-ring Porous plate 12
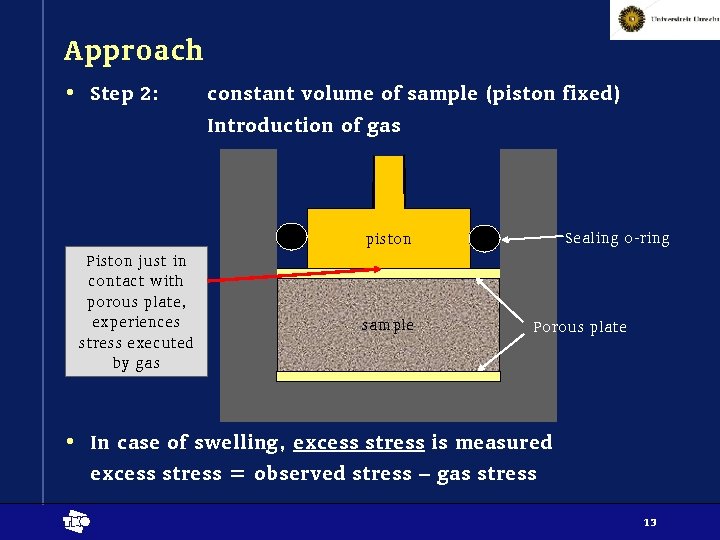
Approach • Step 2: constant volume of sample (piston fixed) Introduction of gas Sealing o-ring piston Piston just in contact with porous plate, experiences stress executed by gas sample Porous plate • In case of swelling, excess stress is measured excess stress = observed stress – gas stress t 13
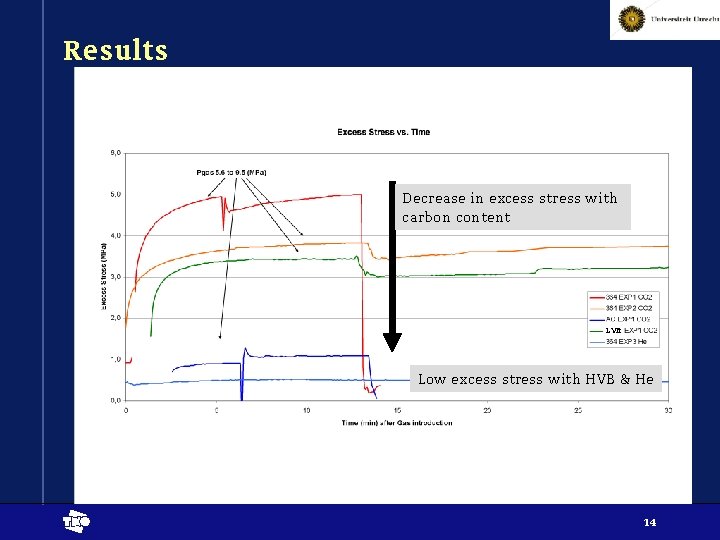
Results Decrease in excess stress with carbon content LVB Low excess stress with HVB & He t 14
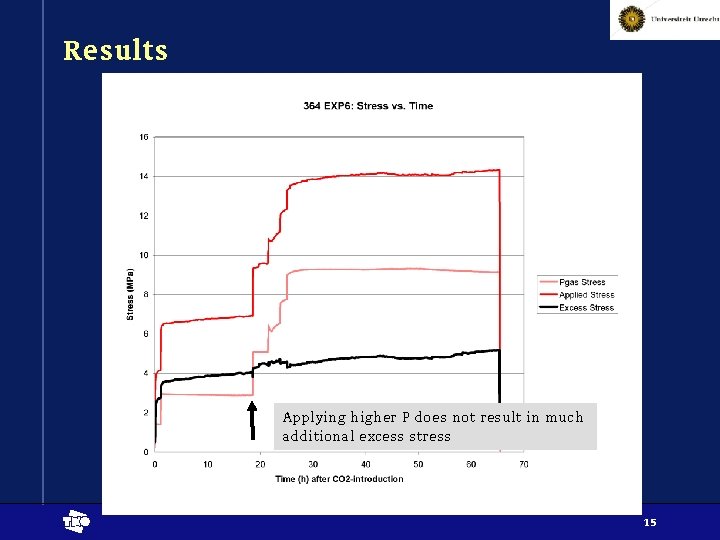
Results Applying higher P does not result in much additional excess stress t 15

Results - interpretation • Exerted force by the sample after introduction of CO 2 is significant • P relationship indicates possibility of chemical reactions • However, similar behavior could be expected from physisorption t 16
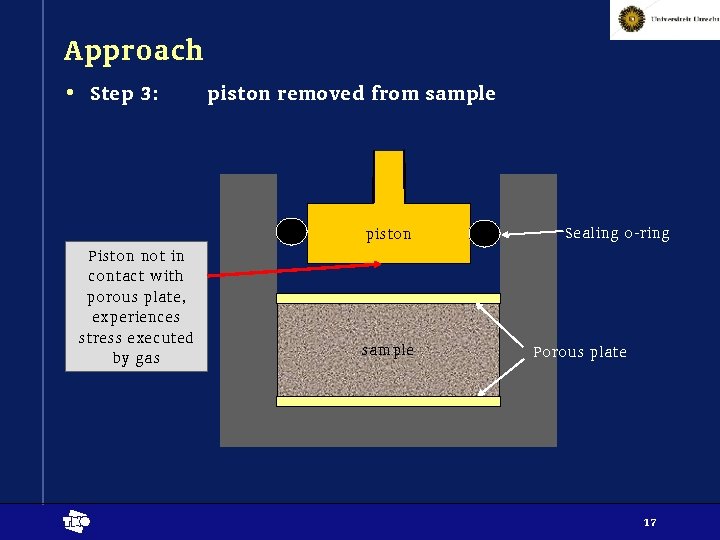
Approach • Step 3: piston removed from sample piston Piston not in contact with porous plate, experiences stress executed by gas t sample Sealing o-ring Porous plate 17
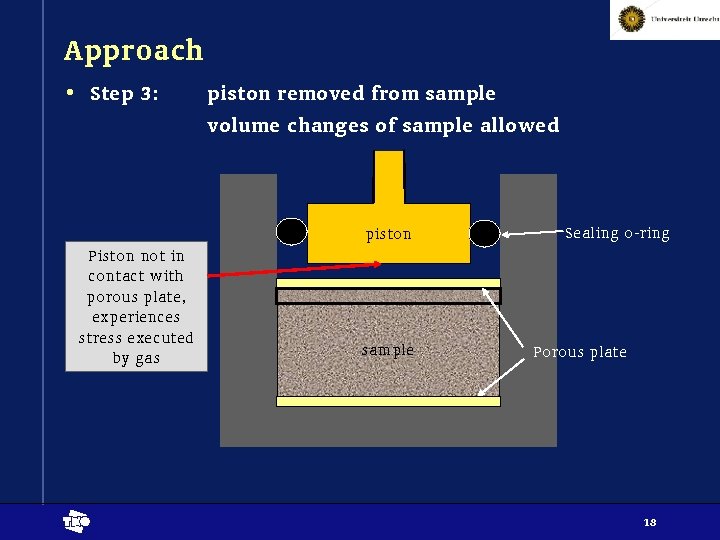
Approach • Step 3: piston removed from sample volume changes of sample allowed piston Piston not in contact with porous plate, experiences stress executed by gas t sample Sealing o-ring Porous plate 18
Approach • Step 3: piston put back on sample, determination of new sample volume piston Piston just in contact with porous plate, experiences stress executed by gas sample Sealing o-ring Porous plate (measured sample volume – initial sample volume) • Strain = initial sample volume t 19
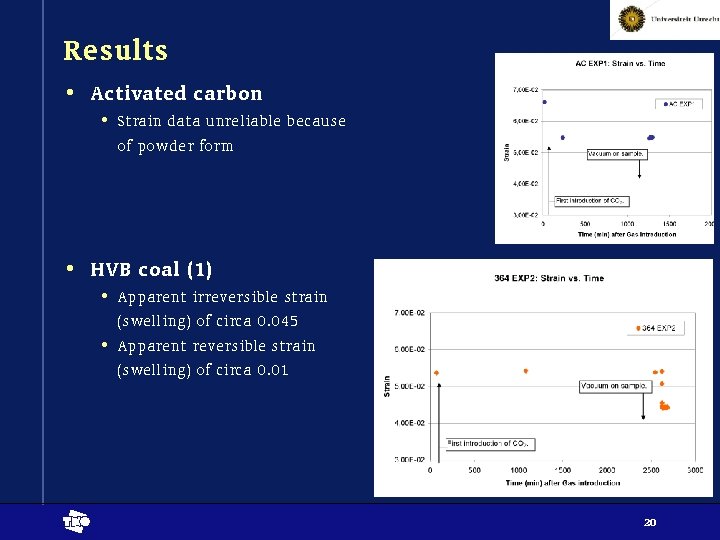
Results • Activated carbon • Strain data unreliable because of powder form • HVB coal (1) • Apparent irreversible strain (swelling) of circa 0. 045 • Apparent reversible strain (swelling) of circa 0. 01 t 20
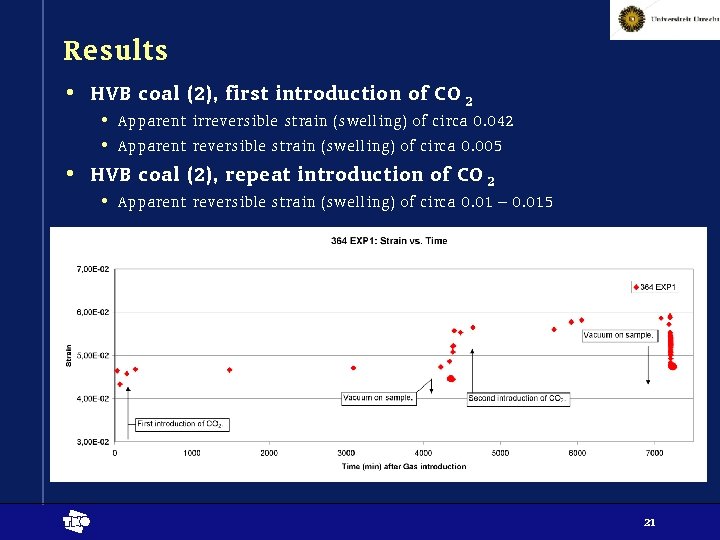
Results • HVB coal (2), first introduction of CO 2 • Apparent irreversible strain (swelling) of circa 0. 042 • Apparent reversible strain (swelling) of circa 0. 005 • HVB coal (2), repeat introduction of CO 2 • Apparent reversible strain (swelling) of circa 0. 01 – 0. 015 t 21
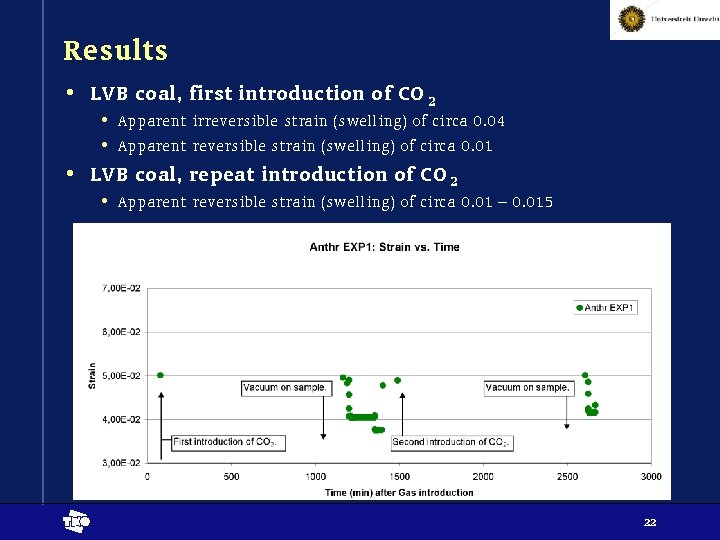
Results • LVB coal, first introduction of CO 2 • Apparent irreversible strain (swelling) of circa 0. 04 • Apparent reversible strain (swelling) of circa 0. 01 • LVB coal, repeat introduction of CO 2 • Apparent reversible strain (swelling) of circa 0. 01 – 0. 015 t 22

Results - interpretation • Volumetric expansion is significant • Strong indications for irreversible chemical reactions, in addition to expansion due to physical sorption t 23
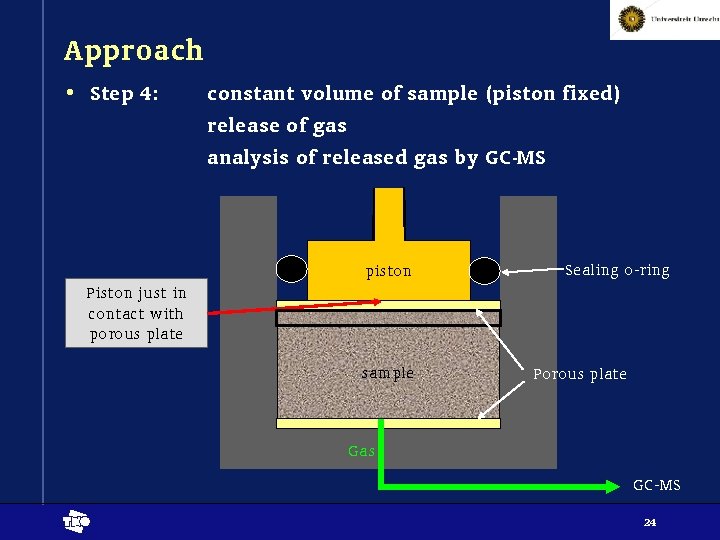
Approach • Step 4: constant volume of sample (piston fixed) release of gas analysis of released gas by GC-MS piston Sealing o-ring Piston just in contact with porous plate sample Porous plate Gas GC-MS t 24
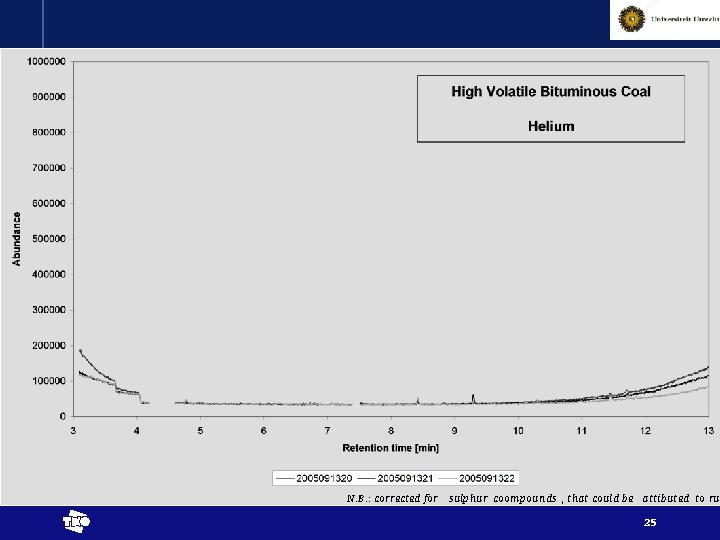
N. B. : corrected for sulphur coompounds , that could be attibuted to rub t 25
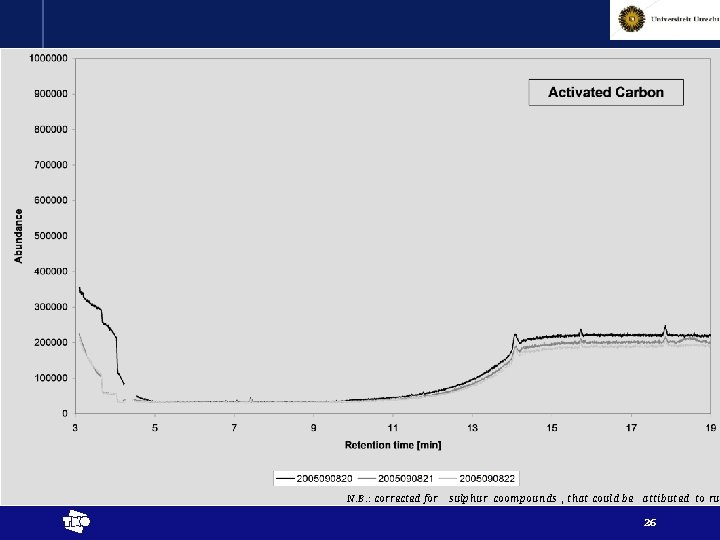
N. B. : corrected for sulphur coompounds , that could be attibuted to rub t 26
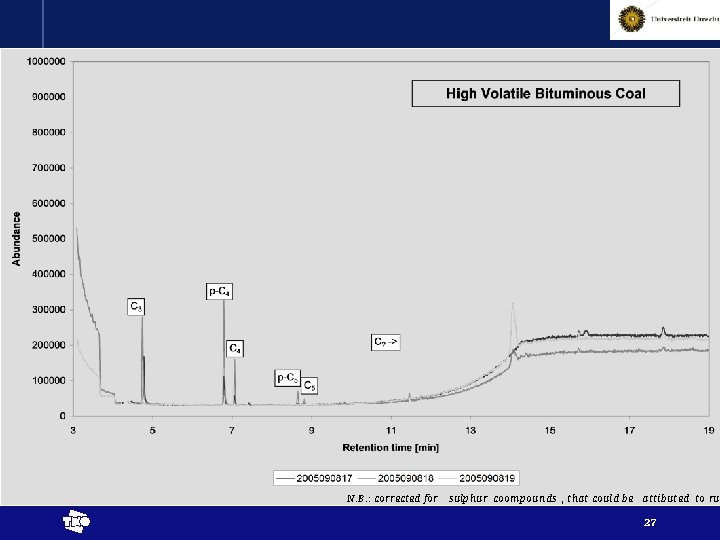
N. B. : corrected for sulphur coompounds , that could be attibuted to rub t 27
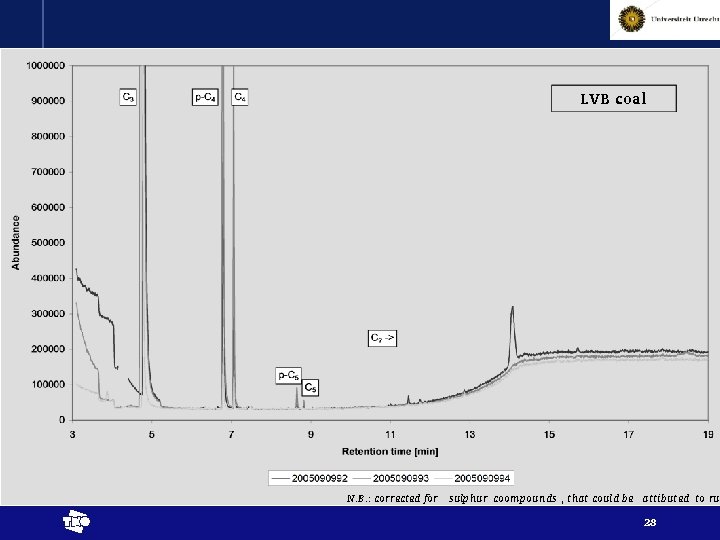
LVB coal N. B. : corrected for sulphur coompounds , that could be attibuted to rub t 28

Results - interpretation • Chemical reactions proven by release of higher alkanes (at least up to pentane) t 29
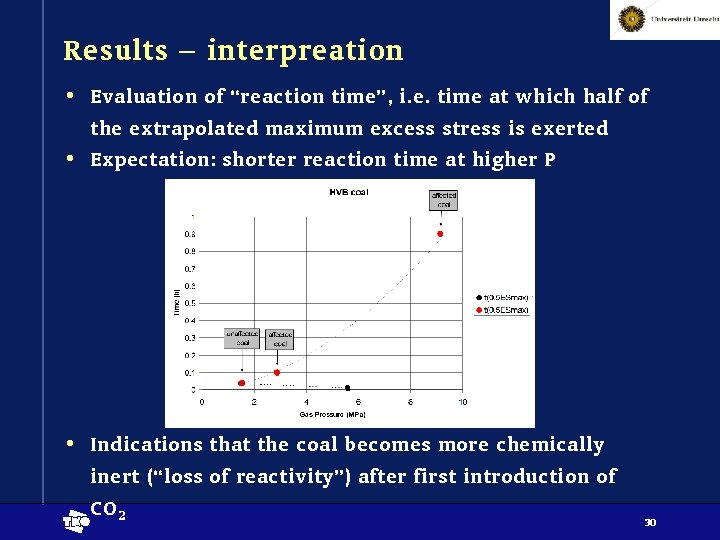
Results – interpreation • Evaluation of “reaction time”, i. e. time at which half of the extrapolated maximum excess stress is exerted • Expectation: shorter reaction time at higher P • Indications that the coal becomes more chemically inert (“loss of reactivity”) after first introduction of CO 2 t 30

Conclusion and implications • After CO 2 -introduction in coal, chemical reactions are likely to occur next to physisorption • Results in permanent coal expansion • Force executed by coal expansion is significant • Observed effects probably dependent on coal characteristics • Rank, composition, etc. • Numerical models usually relate adsorption and swelling to P alone, while coal characteristics seem to play an important role • Coal becomes chemically “inert” after being in contact with CO 2 • 2 -step modelling? First stage “chemical” modelling and second stage “physical modelling? • Observed (chemical) expansion highly relevant to field applications • RECOPOL results showed a decrease in injectivity, attributed to swelling, which was irreversible • Returning to a similar injection P after build-up and fall-off did not result in similar injectivity t 31
Swelling or shrinkage ? • Preliminary experiments under constant load show shrinkage and swelling, depending on stress CO 2 t 32

Swelling or shrinkage ? • Preliminary experiments under constant load show shrinkage and swelling, depending on stress CO 2 t 33

Workshop observations (Frank & Saikat) • Other possibilities besides bi-modal pore structure to explain two different diffusion times (Andreas & Nikolai). • Dirk was able to explain the sorption CH 4 by the a bimodal pore distribution but had difficulties with CO 2. • Several groups did observe differences in response to multiple cycles of CO 2 exposure • More effort should go behind looking into the right way of doing diffusion experiments ( e. g polycyclic stress conditions) t 34

Workshop observations (Frank & Saikat) • More dynamic void volume corrections to sorption data (using swelling coeff. ) • Nikolai: First pressure steps fast (order of hrs. ) and subsequent steps slow (order of >3 days) • Swelling cannot be explained simply by the volume of the adsorbed phase (Nikolai) t 35
- Slides: 35