Coagulation Using PAC Michael Beau M Du July

Coagulation Using PAC Michael Beau M. Du July 27, 2015

OBJECTIVES • To separate fish blood from the brine by coagulation using PAC in order to reuse the brine • To use Poly Aluminum Chloride (PAC) as an alternative coagulant in coagulating fish blood.

INTRODUCTION

COAGULATION • To cause transformation of a liquid into or as if into a soft, semisolid, or solid mass • A process using chemical reactions to change a liquid state to a solid state

Poly Aluminum Chloride (PAC) • • Chemical Formula: Al 2(OH)Cl 5 n Clear to yellow powder, not combustible Soluble in water Remains effective at very low temperatures even below 0 deg C • Flocs formed from PAC settle equally well at low and at normal water temperatures

Poly Aluminum Chloride (PAC) Applications • • Coagulation Liquid-solids Separation Wastewater Treatment Water Clarifications

METHODOLOGY

MATERIALS • Poly Aluminum Chloride (30. 03%PAC) (Bought from Alyson’s Chemicals) • Tuna Fish Blood • Dynafine X Salt • Salinometer • Ice • • Weighing Scale Quickchill Cooler Thermometer
Trial 1: Check for Salinity • Prepare 500 ml 24 BE Brine. • Prepare 150 ml Blood. • Mix the blood with the Brine and check for salinity

Trial 2 at -7 deg Celsius • • Prepare 600 ml 24 BE Brine, Prepare 150 ml Blood Dissolve 10 g PAC in 150 ml water Weigh 4. 5 kg of ice and mix it with Quick chill in a separate container • Place the 600 m. L Brine inside the cooler • Pour the ice with Quick chill in the cooler.

Trial 2 at -7 deg Celsius • Wait for the temperature inside the beaker to reach -7 C • Mix the Dynafine X Salt inside the beaker followed by the PAC • Stir and close the cooler • Wait for settling at the bottom, coagulation • Take a photo for the result.

Preparation for Coagulation (July 23)
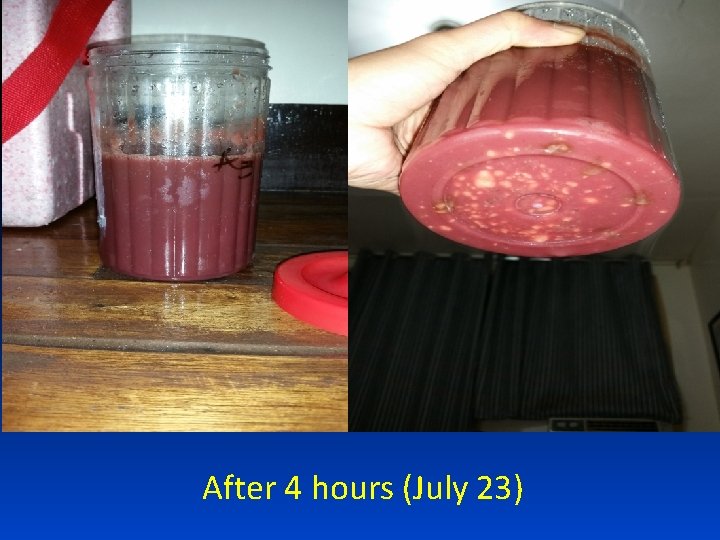
After 4 hours (July 23)

After 1 day (July 24), no coagulation

Trial 3 at -7 deg Celsius • Prepare 600 m. L 24 BE Brine solution • Prepare 50 g PAC to be dissolved in 250 m. L water • Weigh 4. 5 kg of tube ice then mix it with 1. 35 kg of Quickchill • Place the 600 m. L solution inside a cooler then pour the ice to surround the beaker • Wait for temperature inside the beaker to reach -7 C

Trial 3 at -7 deg Celsius • Pour the PAC solution inside the beaker then mix • Wait for coagulation • Take a photo of the result

Coagulated Part

Trial 4 at -7 deg Celsius • Prepare 600 m. L 24 BE Brine solution • Prepare 60 g PAC and 80 g Dynafine to be dissolved in 250 m. L water • Weigh 4. 5 kg of tube ice then mix it with 1. 35 kg of Quickchill • Place the 600 m. L solution inside a cooler then pour the ice to surround the beaker • Wait for temperature inside the beaker to reach -7 C

Trial 4 at -7 deg Celsius • Pour the PAC and Dynafine solution inside the beaker then mix • Wait for coagulation • Take a photo of the result

Coagulated Part

Trial 5 at -7 deg Celsius • Prepare 600 m. L 24 BE Brine solution • Prepare 60 g PAC to be dissolved in 250 m. L water • Weigh 4. 5 kg of tube ice then mix it with 1. 35 kg of Quickchill • Place the 600 m. L solution inside a cooler then pour the ice to surround the beaker • Wait for temperature inside the beaker to reach -7 C

Trial 5 at -7 deg Celsius • Pour the PAC solution inside the beaker then mix • Wait for coagulation • Take a photo of the result

Coagulated Part

Trial 6 at -7 deg Celsius • Prepare 600 m. L 24 BE Brine solution • Prepare 50 g PAC and 80 g Dynafine to be dissolved in 250 m. L water • Weigh 4. 5 kg of tube ice then mix it with 1. 35 kg of Quickchill • Place the 600 m. L solution inside a cooler then pour the ice to surround the beaker • Wait for temperature inside the beaker to reach -7 C

Trial 6 at -7 deg Celsius • Pour the PAC and Dynafine solution inside the beaker then mix • Wait for coagulation • Take a photo of the result

Coagulated Part

CONCLUSION

CONCLUSION The result did not give a clear solution when the blood was coagulated from the brine. The mixture using only PAC coagulated the blood more even at the bottom of the flask compared to the mixture with Dynafine X Salt. The PAC with Dynafine X Salt created a gap in the middle of the brine wherein blood was coagulated at the top and at the bottom of the flask.

CONCLUSION Trial 6, having made two layers of blood, gave a more solid form of blood due to it containing a higher amount of PAC compared to that of

Recommendation • Try using PAC from a different supplier • Make sure the blood used in the experiment does not contain any fish remains

What’s Next • Test what grain size of the salt will be compatible with the PAC powder. • Prepare a costing of the product that will depend on the formulation. (weight of PAC per weight of salt, Size of the package according to the size of the vessel). • Observe the behavior of the PAC when mixed with salt. • Test different quantities of PAC powder to determine the minimum amount PAC to effective and economical.

What’s Next • Chemical analysis for the clarified bloody brine in order to test for traces of residual PAC, in order to test for food grade quality. • Ask the purse seiner operation proceeds, the brine in the fish tank dilutes. Study the remaining salinity after harvesting of the fish in order to compute the amount of salt with PAC per volume of the brine in the fish tank.

What’s Next • PAC powder (35 pesos/kg) retail. • Ask how many minutes/hours between cycles in the fish tank before the next brining in order to determine the optimal/minimum concentration of PAC and still give enough time fore the reaction to occur. • After optimization, try a field trial in Gen. San or in Navotas. • Do a computation of savings for a fishing customer base on actual consumption of salt during one fishing trip.

PMI • Plus • Given enough inputs/instructions • Minus Ø Limited fish blood Ø Limited PAC powder – Interesting – There are other coagulant such as : Similar to PAC such as ( Aluminum Chlorohydrate, see the article “Polyaluminum Coagulant pdf. )

REFERENCES: • http: //neosolutionsinc. com/blog/2013/04/aluminumsulfate-vs-pac-in-potable-water-treatment/ • • http: //wedc. lboro. ac. uk/resources/conference/20/Mal hotra. pdf • • http: //www 4. ncsu. edu/~hubbe/PAC. htm • • http: //www. niir. org/profiles/profile/2040/polyaluminum-chloride. html

Your Success is Our Success!

(http: //www. chemicalland 21. com/industrialc hem/inorganic/POLYALUMINIUM%20 CHL ORIDE. htm)

Poly Aluminum Chloride (PAC) • Lower doses are required for equivalent results in comparison with aluminium sulphate. • remains efficient over a larger p. H range
- Slides: 38