Coagulation disorders of pregnancy By Dr ishraq mohammed

Coagulation disorders of pregnancy By Dr. ishraq mohammed

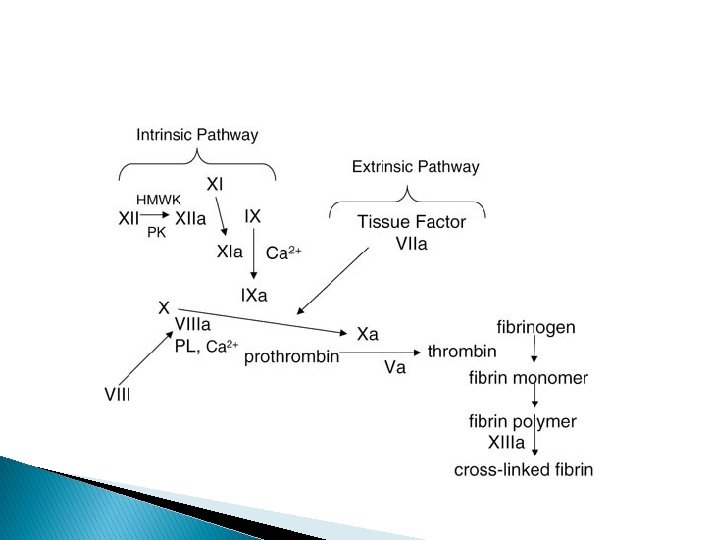
Venous thromboembolism � Venous thromboembolic disease (VTE) is the most common cause of direct maternal death in the UK. Pregnancy is a hypercoagulable state because of an alteration in the thrombotic & fibrinolytic system. There is an increase in clotting factors 8, 9, 10 & fibrinogen level, & a reduction in protein s & anti-thrombin III concentration. The net result of these changes is thought to be an evolutionary response to reduce the likelihood of hemorrhage following delivery.

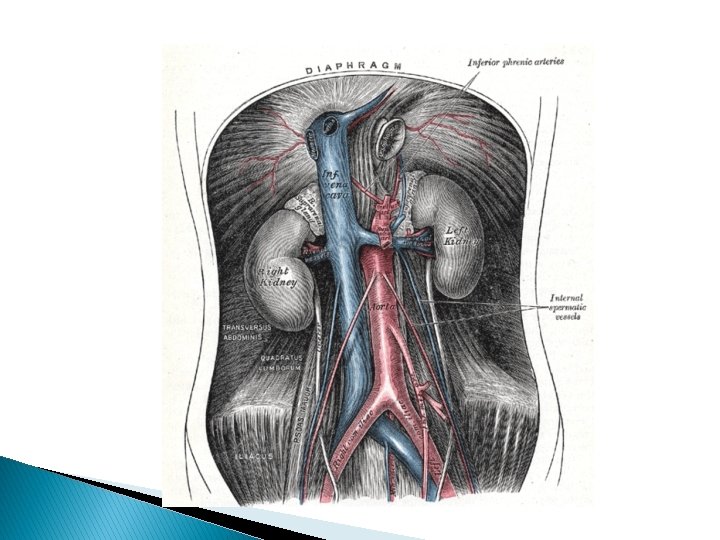
� These physiological changes predispose a woman to thromboembolism & this is further exacerbated by venous stasis in the lower limbs due to the weight of the gravid uterus placing pressure on the inferior vena cava in late pregnancy & immobility, particularly in the puerperium.

� Pregnancy is associated with a 6 -10 fold increase in the risk of VTE compared to nonpregnant situation. Without thromboprophylaxsis, the incidence of nonfatal pulmonary embolism& deep venous thrombosis in pregnancy is about 0, 1 per cent, this increases following delivery to around 1 -2 per cent & is further increased following emergency caesarean section.

Some Risk Factors Associated with an Increased Risk for Thromboembolism Risk Factor Obstetrical 1. Cesarean delivery 2. Diabetes 3. Hemorrhage and anemia 4. Hyperemesis 5. Immobility—prolonged bed rest 6. Multifetal gestation 7. Multiparity 8. Preeclampsia 9. Puerperal infection

General 1. 2. 3. 4. 5. 6. 7. 8. 9. Age 35 years or older Cancer Connective-tissue disease Dehydration Immobility—long-distance travel Infection and inflammatory disease Myeloproliferative disease Nephrotic syndrome Obesity

Oral contraceptive use 11. Orthopedic surgery 12. Paraplegia 13. Prior thromboembolism 14. Sickle cell disease 15. Smoking 16. Thrombophilia 10.

Thrombophilias � Several important regulatory proteins act as inhibitors in the coagulation cascade. Inherited or acquired deficiencies of these inhibitory proteins are collectively referred to as thrombophilias. These can lead to hypercoagulability and recurrent venous thromboembolism. Although these disorders are collectively present in about 15 percent of white European populations, they are responsible for more than 50 percent of all thromboembolic events during pregnancy. Recommendations for antepartum and postpartum thromboprophylaxis for women diagnosed with a thrombophilia are discussed later in this chapter (Thrombophilia Prophylaxis to Prevent Adverse Pregnancy Outcomes).

� Protein C Deficiency � Antithrombin Deficiency � Protein S Deficiency � Activated Protein C Resistance (Factor V Leiden Mutation) � Prothrombin G 20210 a Mutation � Hyperhomocysteinemia

Antiphospholipid Antibodies These autoantibodies are detected in about 2 percent of patients who have nontraumatic venous thrombosis. The antibodies are directed against cardiolipin(s) or against phospholipid-binding proteins such as 2 -glycoprotein I. They are commonly found in patients with systemic lupus erythematosus, (SLE). Women with moderate-to-high levels of these antibodies may have antiphospholipid syndrome, which is defined by a number of clinical features such as thromboembolism or certain obstetrical complications that include. � at least one otherwise unexplained fetal death at or beyond 10 weeks; � at least one preterm birth before 34 weeks; or � at least three consecutive spontaneous abortions before 10 weeks. �

� In these patients, thromboembolism—either venous or arterial—most commonly involves the lower extremities. Importantly, the syndrome also should be considered in women with thromboses in unusual sites, such as the portal, mesenteric, splenic, subclavian, and cerebral veins. Antiphospholipid antibodies are also a predisposing factor for arterial thromboses. In fact, they account for up to 5 percent of arterial strokes in otherwise healthy young women. Thromboses may occur in relatively unusual locations, such as the retinal, subclavian, brachial, or digital arteries.

Deep-Venous Thrombosis Clinical Presentation � Most cases of venous thrombosis during pregnancy are probably confined to the deep veins of the lower extremity. The frequency and extent to which they involve the pelvic veins is not known precisely, but preliminary observations indicate that iliac vein thrombosis may be frequent in those after cesarean delivery (The signs and symptoms vary greatly and depend in large measure on the degree of occlusion and the intensity of the inflammatory response. Interestingly, most cases during pregnancy occur in the left leg. 90 percent of lower extremity thromboses involved the left leg. Greer (2003) hypothesizes that this may result from compression of the left iliac vein by the right iliac artery and ovarian artery, both of which cross the vein only on the left side.

� Classical thrombosis involving the lower extremity is abrupt in onset, and there is pain and edema of the leg and thigh. The lower-extremity thrombus typically involves much of the deep venous system to the iliofemoral region. Occasionally, reflex arterial spasm causes a pale, cool extremity with diminished pulsations. Conversely, there may be appreciable clot, yet little pain, heat, or swelling. Importantly, calf pain, either spontaneous or in response to squeezing or to stretching the Achilles tendon—Homans sign—may be caused by a strained muscle or a contusion. � A fourth of untreated cases have an associated pulmonary embolism. Anticoagulation reduces this risk to less than 5 percent as discussed subsequently.

� Pretest risk score calculated by assigning 1 point for each of the nine following characteristics: active cancer, immobilization, bed rest more than 3 days or surgery within 12 weeks, local tenderness, entire leg swollen, asymmetric calf swelling greater than 3 cm when measured 10 cm below tibial tuberosity, pitting edema only in symptomatic leg, collateral nonvaricose superficial veins, or prior deep-venous thrombosis. Two points are subtracted if an alternative diagnosis is at least as likely as deep-venous thrombosis. a. High risk = 3 or more points, moderate risk = 1 or 2 points, low risk = 0 or less points. (From Lockwood, 2007, with permission. )

Diagnosis: � Venography � Invasive contrast venography remains the standard to exclude lower extremity deepvenous thrombosis But venography is associated with significant complications, including thrombosis, and it is time consuming and cumbersome. Thus, noninvasive methods are usually used to confirm the clinical diagnosis.

Compression Ultrasonography � This noninvasive technique is currently the most-used first -line test to detect deep-venous thrombosis (Greer, 2003). The diagnosis is based on the noncompressibility and typical echoarchitecture of a thrombosed vein (Davis, 2001). In symptomatic nonpregnant patients, examination of the femoral, popliteal, and calf trifurcation veins is more than 90 -percent sensitive and more than 99 -percent specific for proximal thrombosis. In pregnant women, the important caveat is that normal findings with venous ultrasonography results do not always exclude a pulmonary embolism. This is because thrombosis may have already embolized or because it arose from deep pelvic veins inaccessible to ultrasound evaluation �

� Computed Tomography � Spiral computed tomography (CT) scanning is widely available and very useful for detecting lower extremity deep-venous thrombosis as well as those within the vena cava and iliac and pelvic venous systems. Although radiation and contrast agents are required, the benefits of CT outweigh any theoretical risks if lead shielding is used. Fetal radiation exposure is negligible unless the pelvic veins are imaged

� Magnetic Resonance (MR) Imaging � D-Dimer Screening Tests � These specific fibrin degradation products are generated when fibrinolysin degrades fibrin, as occurs in thromboembolism. Their measurement is frequently incorporated into diagnostic algorithms for venous thromboembolism in nonpregnant patients. Screening with the Ddimer test in pregnancy, however, is problematic for a number of reasons. Depending on assay sensitivity, D-dimer serum levels increase with gestational age along with substantively elevated plasma fibrinogen concentrations

Management � Anticoagulation is initiated with either unfractionated or low-molecular-weight heparin. During pregnancy, heparin therapy is continued, and for postpartum women, anticoagulation is begun simultaneously with warfarin. Recall that pulmonary embolism develops in about 25 percent of patients with untreated venous thrombosis, and anticoagulation decreases this risk to less than 5 percent. In nonpregnant patients, the mortality rate is less than 1 percent). � Over several days, leg pain dissipates. After symptoms have abated, graded ambulation should be started. Elastic stockings are fitted anticoagulation is continued. Recovery to this stage usually takes 7 to 10 days.
Heparinization � Treatment of thromboembolism during pregnancy is with either unfractionated or low -molecular-weight heparin. Although either type is acceptable, most recommend one of the low-molecular-weight Unfractionated Heparin (UFH) � Treatment is with an intravenous heparin bolus followed by continuous infusion titrated to achieve full anticoagulation. � heparins.

� an initial bolus of intravenous unfractionated heparin at a dose of 80 units/kg. This is followed by continuous infusion of at least 30, 000 IU for 24 hours, titrated to achieve an activated partial thromboplastin time (a. PTT) of 1. 5 to 2. 5 times control values. . Intravenous anticoagulation should be maintained for at least 5 to 7 days, after which, treatment is converted to subcutaneous heparin. Injections are then given every 8 hours to maintain the a. PTT to at least 1. 5 to 2. 5 times control throughout the dosing interval. For women with antiphospholipid syndrome, a. PTT does not accurately assess heparin anticoagulation, and thus anti-factor Xa levels are preferred.

� The duration of full anticoagulation varies, and there are several acceptable schemes. recommends anticoagulation throughout pregnancy and for 6 weeks postpartum but for a minimum total duration of 6 months. � Prophylactic doses of subcutaneous unfractionated heparin can range from 5000 to 10, 000 units every 12 hours titrated to maintain an antifactor Xa level of 0. 1 to 0. 2 units, measured 6 hours after the last injection. If the venous thromboembolism occurs during the postpartum period, Lockwood (2007) recommends a minimum of 6 months of anticoagulation treatment

Low-Molecular-Weight Heparin � This is a family of derivatives of unfractionated heparin, and their molecular weights average 4000 to 5000 daltons compared with 12, 000 to 16, 000 daltons for conventional heparin. None of these heparins cross the placenta, and all exert their anticoagulant activity by activating antithrombin. The primary difference is in their relative inhibitory activity against factor Xa and thrombin (Specifically, unfractionated heparin has equivalent activity against factor Xa and thrombin, but low-molecular-weight heparins have greater activity against factor Xa than thrombin. They also have a more predictable anticoagulant response and fewer bleeding complications than unfractionated heparin because of their better bioavailability, longer half-life, dose-independent clearance, and decreased interference with platelets. �

Complications of Anticoagulation � Three significant complications associated with anticoagulation are hemorrhage, thrombocytopenia, and osteoporosis. The latter two are unique to heparin, and their risk may be reduced with low-molecular-weight heparins � Heparin-Induced Thrombocytopenia. There are two types of heparin-induced thrombocytopenia —commonly referred to as HIT. The most common is a nonimmune, benign, reversible form that occurs within the first few days of therapy and resolves in about 5 days without cessation of therapy

� The incidence of HIT is approximately 3 to 5 percent in nonpregnant individuals � If thrombocytopenia is severe, heparin therapy must be stopped and alternative anticoagulation initiated. Low-molecularweight heparin may not be an entirely safe alternative because it has some cross reactivity with unfractionated heparin. In these cases, many recommend danaparoid—a sulfated glycosaminoglycan heparinoid.

� Heparin-Induced Osteoporosis � Bone loss may develop with long-term heparin administration—usually 6 months or longer—and is more prevalent in cigarette smokers although such a result is less likely than with unfractionated heparin. Women treated with any heparin should be encouraged to take a daily supplement of 1500 mg of calcium (Low-molecular-weight heparins may cause less adverse effects.

Pulmonary Embolism � Although it causes about 10 percent of maternal deaths, pulmonary embolism is relatively uncommon during pregnancy and the puerperium. The incidence averages about 1 in 7000 pregnancies. There is an almost equal prevalence for antepartum and postpartum embolism, but those developing postpartum have a higher mortality rate. According to Marik and Plante (2008), 70 percent of women presenting with a pulmonary embolism have associated clinical evidence of deep-venous thrombosis. In women presenting with a deep-venous thrombosis, almost half will have a silent pulmonary embolism.

Clinical Presentation � Common symptoms included dyspnea in 82 percent, chest pain in 49 percent, cough in 20 percent, syncope in 14 percent, and hemoptysis in 7 percent. Other predominant clinical findings typically include tachypnea, apprehension, and tachycardia

� Right axis deviation and T-wave inversion in the anterior chest leads may be evident on the electrocardiogram. On chest radiography, there may be loss of vascular markings in the region of the lungs supplied by the obstructed artery. Although most women are hypoxemic, it is emphasized that a normal arterial blood gas analysis does not exclude pulmonary embolism. Approximately a third of patients younger than 40 years will have p. O 2 values >80 mm Hg. In contrast, the alveolar-arterial oxygen tension difference is a more useful indicator of disease, as more than 86 percent of patients with acute pulmonary embolism will have an alveolar-arterial difference of more than 20 mm Hg ). Even with massive pulmonary embolism, signs, symptoms, and laboratory data to support the diagnosis may be deceptively nonspecific.

Diagnosis: � As with deep-venous thrombosis, the diagnosis of pulmonary embolism requires an initial high index of suspicion followed by objective testing. the initial imaging evaluation for suspected pulmonary embolism during pregnancy generally includes ventilation-perfusion scintigraphy, computed tomography, or bilateral compression ultrasonography. Of note, a chest radiograph should be performed if there is underlying suspicion for other diagnoses. � Spiral CT. � Ventilation–Perfusion Scintigraphy—Lung Scan � Magnetic Resonance Angiography (MRA) � Pulmonary Angiography

Abnormalities on Chest Radiography: 14% Normal 68% Atelectasis or parenchymal density 48% Pleural Effusion 35% Pleural based opacity 24% Elevated diaphragm 15% Prominent central pulmonary artery 7% Cardiomegally 5% Pulmonary edema.

ECG of Acute pulmonary embolus: -An S 1 Q 3 T 3 pattern: prominent S wave in lead I, Q wave and inverted T wave in lead III -Sinus tachycardia -T wave inversion in leads V 1 - V 3 -Right Bundle Branch Block -low amplitude deflections.

Management � Immediate treatment for pulmonary embolism is full anticoagulation similar to that for deep -venous thrombosis as discussed in Pharmacokinetics in Pregnancy. A number of complementary procedures may be indicated � Vena Caval Filters � Thrombolysis � Embolectomy

Antenatal thromboprophylaxis risk assessment & management: A-High risk: Requires antenatal prophylaxis with LMWH. Including single previous VTE + 1 -thrombophilia or family history. 2 -unprovoked/oestrogen-related previous recurrent VTE(>1).

B-Intermediate risk: Consider antenatal prophylaxis with LMWH. Which include: 1 -single previous VTE with no family history or thrombophilia. 2 -thrombophilia+no VTE. 3 -medical cormorbidities, eg: heart or lung disease, SLE, cancer, inflammatory conditions, nephrotic syndrome , sickle cell disease, intravenous drug user. 4 -surgical procedure, eg. appendicectomy.

C-Lower risk: Mobilization and avoidance of dehydration. Which include: 1 -Age >35 years. 2 -obesity(BMI>30 Kg/m 2). 3 -parity >=3. 4 -smoker. 5 -Gross varicose veins. 6 -current systemic infection. 7 -Immobility, eg. paraplegia, SPD, long-distance travel. 8 -pre-eclampsia. 9 -Dehydration/hyperemesis/OHSS. 10 -Multiple pregnancy or ART. Note: if 3 or more risk factors and 2 or more if admitted considered as high+intermediate risk, but less than 3 risk factors considered as low risk.

Postnatal thromboprophylaxis risk assessment & management: A-High risk: At least 6 weeks postnatal prophylactic LMWH. Any previous VTE + anyone requiring antenatal LMWH. B-Intermediate risk: At least 7 days postnatal prophylactic LMWH. NOTE: if persisting or >3 risk factors, consider extending thrombophylaxis with LMWH. Which includes: 1 -caesarean section in labour.

2 -Asymptomatic thrombophilia (inherited or acquired). 3 -BMI>40 kg/m 2. 4 -prolonged hospital admission. 5 -medical cormorbidities, eg. heart or lung disease, SLE, cancer, inflammatory conditions, nephrotic syndrome, sickle cell disease, intravenous drug user.

C-Lower risk: Mobilization and avoidance of dehydration. Which includes: 1 -Age>35 years. 2 -obesity (BMI>30 kg/m 2). 3 -parity >=3. 4 -smoker. 5 -Elective caesarean section. 6 -Any surgical elective in puerperium. 7 -Gross varicose vein.

8 -Current systemic infection. 9 -Immobility, eg. paraplegia, SPD, long-distance travel. 10 -pre-eclampsia. 11 -mid-cavity rotational operative delivery. 12 -prolonged labour >24 hours. 13 -PPH>1 litre or blood transfusion. NOTE: if 2 or more risk factors is considered as intermediate or high risk but if less than 2 risk factors is considered as lower risk.

Thanks
- Slides: 43