Clostridium botulinum Toxin The Neuromuscular Wonder Drug Amy

Clostridium botulinum Toxin: The Neuromuscular Wonder Drug Amy Malhowski Biology 360 March 30, 2005 Figure taken from: http: //www. consultingroom. com/Aesthetics/Product_Display. asp? ID=1

Public Perception of Botulinum Toxin Bioterrorism! Figures taken from: http: //www. safetycentral. com/bottoxfacin. html http: //archives. cnn. com/2002/ALLPOLITICS/06/12/bush. terror/

And of course…Botox® Aka “The Fountain of Youth” Figures taken from:

Outline of Talk • • • Historical background of C. botulinum Transmission of Botulinum toxin Molecular pathogenesis Therapeutic uses of Botulinum toxin Concluding remarks

What is Botulism? • Results in flaccid paralysis of muscles • Caused by toxin produced from C. botulinum • Three types – via route of entry of bacteria – Foodborne, infant, wound • Mainly exists as foodborne outbreaks • Now have bioterrorism threat

The History of Botulinum Toxin • Coined “botulism” in 1700 s from botulus (sausage) after an outbreak from consumption of improperly cooked sausage • Published 1 st case studies on botulinum intoxication • Accurately described neurological symptoms • 1 st to propose therapeutic use of toxin Figure adapted from: Erbguth, 2004.

Symptoms of Botulism Figure taken from Caya, et al. , 2004.

Finding the Culprit • Emile Pierre van Ermengem – – 1 st to connect botulism to bacterium isolated from raw, salted pork & postmortem tissues of botulism victims – Sucessfully isolated bacterium, naming it Bacillus botulinus

Clostridium botulinum • • • Strict anaerobe Gram-positive Bacillus (rod) shape Ubiquitous in terrestrial environment Virulence factor = Botulinum toxin – Released under specific conditions Figure taken from http: //www. jhsph. edu/Publications/Special/cover 2. htm

Botulism and Bioterrorism • Botulinum toxin attempted use as biological weapon during WWII – aborted when toxin did not affect test animals (donkeys) • Great potential in toxicity • Bo. NT no longer considered good biological weapon

Mass Producing Botulinum Toxin • Fort Detrick (1946) – bioweapon research – 1 st time mass produce toxin • Production process- grow, crystallize • 1972 – Nixon terminates all research on biological warfare agents • Research continues – 1979 – Schantz produces batch 79 -11; used until 1997 • 1991 – several batches made – Botox ® by Allergan Inc.

So what? Importance of C. botulinum Research • Bioterrorism/outbreaks • Kerner – brought about idea of therapeutics • Most recent work – harnessing Bo. NT as therapeutic agent for neuromuscular disorders

Outline of Talk • • • Historical background of C. botulinum Transmission of Botulinum toxin Molecular pathogenesis Therapeutic uses of Botulinum toxin Impediments in Treatment Concluding remarks

Transmission of Botulinum Toxin • Most commonly via improperly cooked food • Conditions to produce toxin not completely understood • Complex route of transmission – – Ingestion/injection Neurotoxin produced as progenitor complex Absorbed into tissue circulates blood Docks onto receptors of neuron transcytosis binds up acetylcholine paralysis

Classes of Botulinum Toxin • Seven different subtypes of botulinum toxin – A, B, C 1, D, E, F, and G • Same general mechanism for muscular paralysis • Vary in structure, target site, & toxicity • Only two manufactured for commercial use – A and B

Target Proteins of Botulinum Toxins Serotype Cellular Substrate Target Cleavage Site A SNAP-25 Gln 197 -Arg 198 B VAMP/Synaptobrevin Cellubrevin Gln 76 -Phe 77 Gln 59 -Phe 60? C 1 Syntaxin 1 A, 1 B SNAP-25 Lys 253 -Ala 254 Lys 252 -253 D VAMP/Synaptobrevin (18, 181) Cellubrevin (181) Lys 59 -Leu 60 Ala 67 -Asp 68 Lys 42 -Leu 43? E SNAP-25(46) Arg 180 -Ile 181 F VAMP/Synaptobrevin (181, 182) Cellubrevin Gln 58 -Lys 59 Gln 41 -Lys 42? G VAMP/Synaptobrevin Ala 81 -Ala 82 Figure adapted from: Aoki. 2004. Curr Med Chem. 11: 3085 -3092.

Outline of Talk • • • Historical background of C. botulinum Transmission of Botulinum toxin Molecular pathogenesis Therapeutic uses of Botulinum toxin Impediments in Treatment Concluding remarks

Molecular Pathogenesis of Bo. NT synthesized as single-chain polypeptide (inactive form) Polypeptide cleaved by protease to create dichain structure (active form) Bo. NT binds to epithelium, transcytosed, reaches general circulation Receptor-mediated endocytosis at peripheral cholinergic nerve endings In cytosol, toxin cleaves target, blocking neurotransmitter release = flaccid paralysis
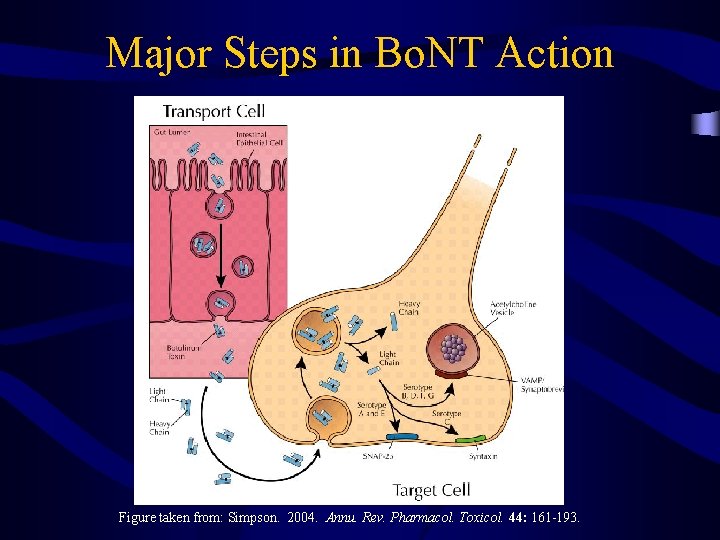
Major Steps in Bo. NT Action Figure taken from: Simpson. 2004. Annu. Rev. Pharmacol. Toxicol. 44: 161 -193.
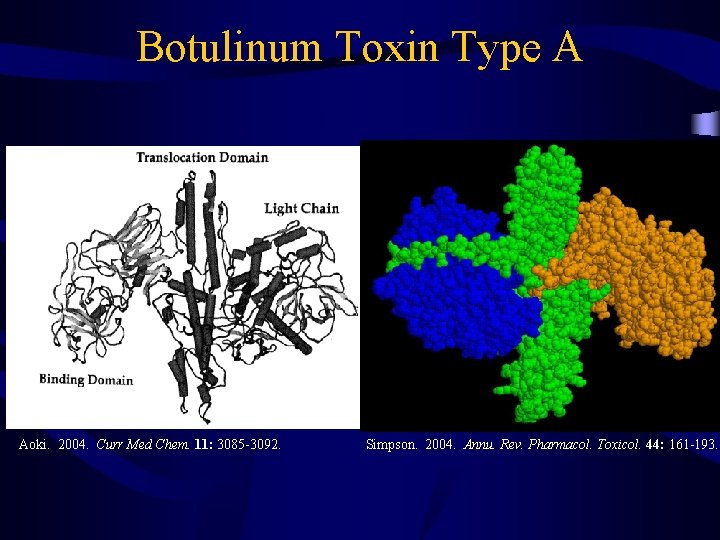
Botulinum Toxin Type A Aoki. 2004. Curr Med Chem. 11: 3085 -3092. Simpson. 2004. Annu. Rev. Pharmacol. Toxicol. 44: 161 -193.
Figure taken from: Arnon, et al. 2001.

Uses of Botulinum Toxin • Bioterrorism agent – Category A • Local paralytic agent – Botox® • Therapeutic agent – Neuromuscular disorders – Pain management

Bo. NT as Local Paralytic Agent • • • Use Botulinum toxin type A (Botox®) Many cosmetic uses Few clinical side effects Fast acting – 6 hrs post injection Effects last 3 -6 months Serial injections required to maintain results

Bo. NT/A Induces Local Paralysis • Local effects = dose dependent • Injection site affects physical outcome Before Botox® Figures adapted from: Mendez-Eastman. 2003. Plast. Surg. Nurs. 23: 64 -70. After Botox®

Outline of Talk • • • Historical background of C. botulinum Transmission of Botulinum toxin Molecular pathogenesis Therapeutic uses of Botulinum toxin Impediments in Treatment Concluding remarks
Bo. NT as a Therapeutic Agent • Botox® used for aesthetics therapeutic use in neuromuscular disorders • Bo. NT/A = Botox ® from Allergan, Inc. • Bo. NT/B = MYOBLOC™ from Elan Pharmaceuticals

Bo. NT as Therapeutic Agent in Neuromuscular Disorders • Purified Bo. NT/A = Botox ® • Treat medical conditions characterized by muscle hyperactivity/spasm – blepharospasm, strabismus, cervical dystonia, glabellar lines, spastic dystonia, limb spasticity, tremors, chronic anal fissure, hyperhidrosis, etc. • Currently only FDA approved for 4 disorders – – Blepharospasm (aka focal dystonia) Strabismus Cervical dystonia Hyperhidrosis

Bo. NT/A & Muscle Hyperactivity Cervical Dystonia (CD) • CD – involuntary contractions of neck and shoulder muscles • FDA approved injections with Bo. NT/A (2000) • Bo. NT/A is injected into affected muscles to reduce muscle contraction • Bo. NT/A effectively reduces muscle spasticity and pain associated with CD

Cervical Dystonia Study with Botox® by Allergan, Inc. • Phase 3 randomized, multi-center, double blind, placebo-controlled study on treatment of CD with Botox ® (1998) • 170 subjects (88 in Botox® group, 82 in placebo group), analyzed until 10 wks postinjection • Study suggested that majority of patients had beneficial response by 6 th week

Cervical Dystonia Study with Bo. NT/A as Dysport® • A multicenter, double-blind, randomized, controlled trial with Dysport® to treat CD in the USA (2005) • Patients (80) randomly assigned to receive Dysport® (500 U) or placebo • Dysport was significantly more effective than placebo at weeks 4, 8, and 12 • Dysport group had 38% with positive treatment response, with median duration of response of 18. 5 weeks

Bo. NT/A & Pain Management • Testing Bo. NT use in controlling painassociated disorders • Data suggests Bo. NT acts in complex manner – not just controlling overactive muscle • Appears that Bo. NT inhibits the release of neurotransmitters (glutamate and substance P) involved in pain transmission
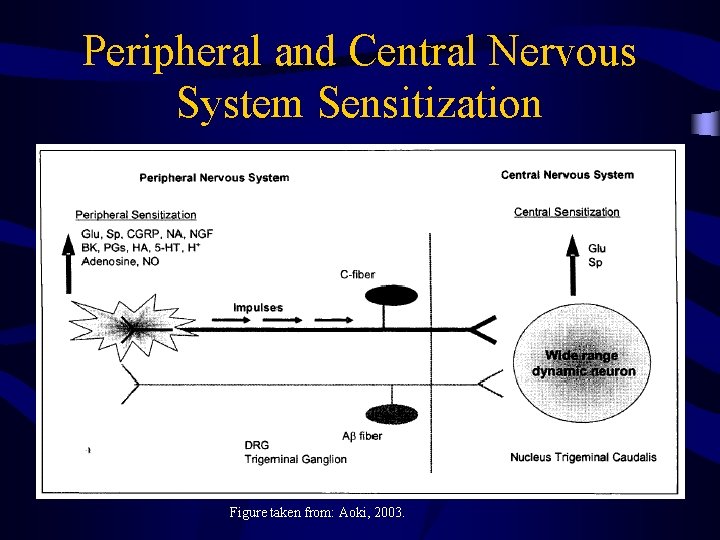
Peripheral and Central Nervous System Sensitization Figure taken from: Aoki, 2003.
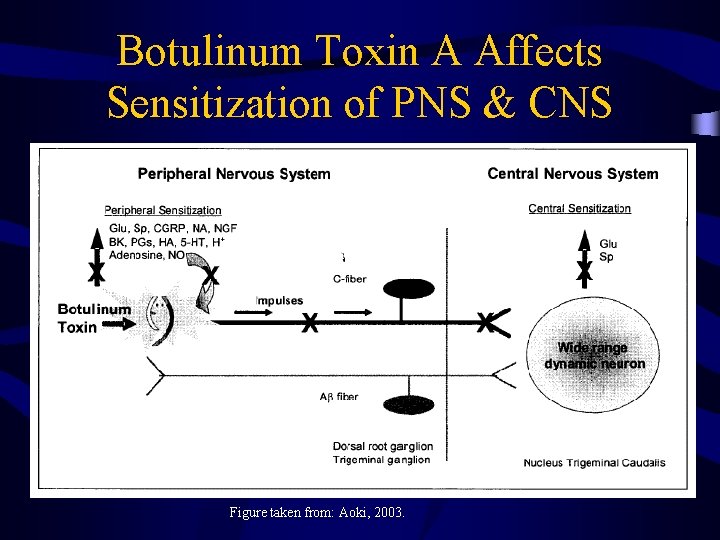
Botulinum Toxin A Affects Sensitization of PNS & CNS Figure taken from: Aoki, 2003.
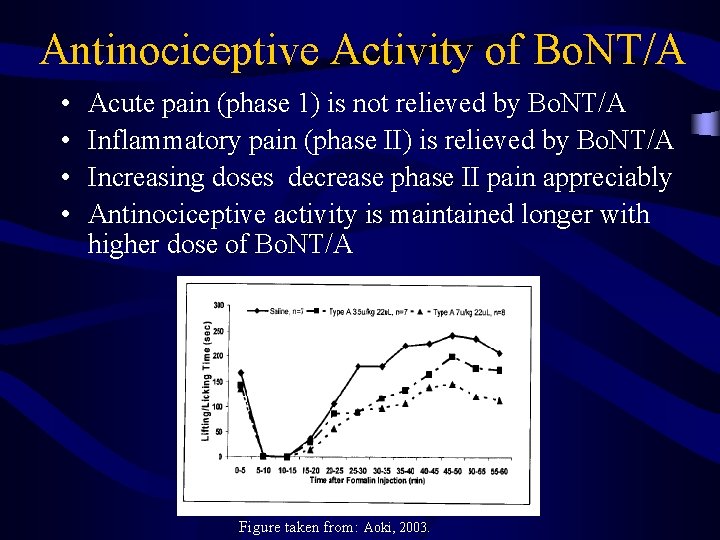
Antinociceptive Activity of Bo. NT/A • • Acute pain (phase 1) is not relieved by Bo. NT/A Inflammatory pain (phase II) is relieved by Bo. NT/A Increasing doses decrease phase II pain appreciably Antinociceptive activity is maintained longer with higher dose of Bo. NT/A Figure taken from: Aoki, 2003.
Bo. NT/A Injection Reduces Formalin -induced Pain • Upon formalin challenge 5 days post-injection, dosedependent decrease in Glut release is observed • Injection of Bo. NT/A prevents increase of formalininduced Glut release Figure taken from: Aoki, 2003.
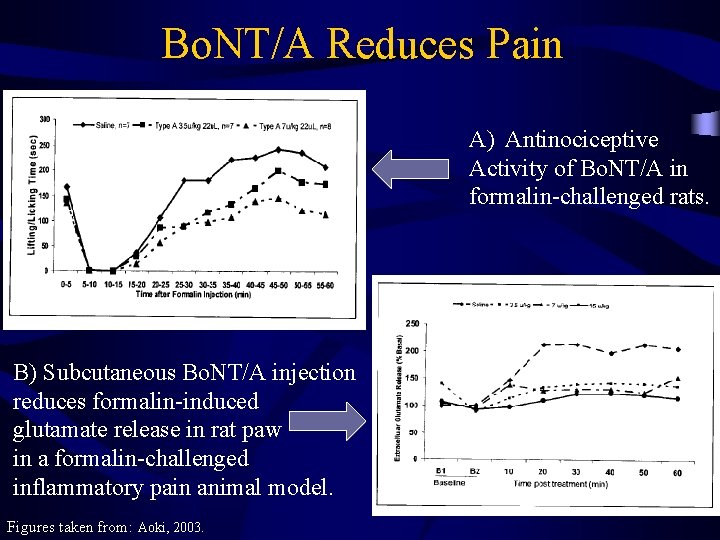
Bo. NT/A Reduces Pain A) Antinociceptive Activity of Bo. NT/A in formalin-challenged rats. B) Subcutaneous Bo. NT/A injection reduces formalin-induced glutamate release in rat paw in a formalin-challenged inflammatory pain animal model. Figures taken from: Aoki, 2003.

Conclusions on Therapeutics • Bo. NT mechanism = specific • Uses are diverse – Local flaccid paralysis – Reducing muscle spasticity – Reducing pain • Currently, Bo. NT therapy on muscle disorders and associated pain

Outline of Talk • • • Historical background of C. botulinum Transmission of Botulinum toxin Molecular pathogenesis Therapeutic uses of Botulinum toxin Impediments in Treatment Concluding remarks

Impediments in Treating with Bo. NT • FDA approval pending for many disorders • Fleeting effects – need repeated injections • Socioeconomics – less expensive than surgery but not permanent • Social constraints – – not up to snuff on research – stigma in using deadly toxin for good use

Concluding Remarks • Toxin = great therapeutic agent! • Research to understand mechanism of release of Bo. NT from C. botulinum • Impediments in therapeutics • Future with Botox® is bright!

And remember… Sometimes wrinkles aren’t all that bad!

Thank you! • Chris White-Ziegler • My readers: Caitlin Reed & Natalia Grob • Bio 360 students Figure taken from: http: //www. jwolfe. clara. net/Humour/Med. Miscel. htm

References • • • Aoki, K. R. 2001. A Comparison of the Safety Margins of Botulinum Neurotoxin Serotypes A, B, and F in Mice. Toxicon. 39: 1815 -1820. Aoki, K. R. 2003. Evidence for Antinociceptive Activity of Botulinum Toxin Type A in Pain Management. Headache. 43: S 9 -S 15. Aoki, K. R. 2004. Botulinum Toxin: A Successful Therapeutic Protein. Curr. Med. Chem. 11: 3085 -3092. Bossi, P. , A. Tegnell, A. Baka, F. Van Loock, J. Hendriks, A. Werner, H. Maidhof, and G. Gouvras. 2004. Bichat Guidelines for the Clinical Management of Botulism and Bioterrorism-related Botulism. Eurosurveillance. 9: 1 -4. Breldenbach, M. A. and A. T. Brunger. 2004. Substrate Recognition Strategy for Botulinum Neurotoxin Serotype A. Nature. 432: 925 -929. Casadevall, A. , E. Dadachova, and L. Pirofski. 2004. Passive Antibody Therapy for Infectious Diseases. Nat Rev Microbiol. 2: 695 -703. Casadevall, A. and L. Pirofski. 2004. The Weapon Potential of a Microbe. Trends Microbiol. 12: 259 -263. Caya, J. G. , R. Agoi, and J. E. Miller. 2004. Clostridium botulinum and the Clinical Laboratorian: A Detailed Review of Botulism, Including Biological Warfare Ramifications of Botulinum Toxin. Arch Pathol Lab Med. 128: 653 -662. CDC Botulism Emergency Preparedness & Response. http: //www. bt. cdc. gov/agent/botulism/

References • • • Chaddock, J. A. , J. R. Purkiss, L. M. Friis, J. D. Broadbridge, M. J. Duggan, S. J. Fooks, C. C. Shone, C. P. Quinn, and K. A. Foster. Inhibition of Vesicular Secretion In Both Neuronal and Nonneuronal Cells by a Retargeted Endopeptidase Derivative of Clostridium botulinum Type A. Infect. Immun. 68: 2587 -2593. Coffield, J. A. , N. M. Bakry, A. B. Maksymowych, and L. L. Simpson. 1999. Characterization of a Vertebrate Neuromuscular Junction That Demonstrates Selective Resistance to Botulinum Toxin. J. Pharmacol. Exp. Ther. 289: 1509 -1516. Coffield, J. A. , N. M. Bakry, R. -d. Zhang, J. Carlson, L. G. Gomella, and L. L. Simpson. 1997. In Vitro Characterization of Botulinum Toxin Types A, C and D Action on Human Tissues: Combined Electrophysiologic, Pharmacologic and Molecular Biologic Approaches. J. Pharmacol. Exp. Ther. 280: 1489 -1498. Cui, M. , S. Khanijou, J. Rubino, and K. R. Aoki. 2004. Subcutaneous Administration of Botulinum Toxin A Reduces Formalin-Induced Pain. 107: 125 -133. De Paiva, A. , F. A. Meunier, J. Molgo, K. R. Aoki, and J. O. Dolly. 1999. Functional Repair of Motor Endplates After Botulinum Neurotoxin Type A Poisoning: Biphasic Switch of Synaptic Activity Between Nerve Sprouts and their Parent Terminals. Proc. Natl. Acad. Sci. USA. 96: 3200 -3205.

References • • • Erbguth, F. J. 2004. Historical Notes on Botulism, Clostridium botulinum, Botulinum Toxin, and the Idea of the Therapeutic Use of the Toxin. Mov Dis. 19: S 2 -S 6. Fernandez-Salas, E. , H. Ho, P. Garay, L. E. Steward, and K. R. Aoki. 2004. Is the Light Chain Subcellular Localization an Important Factor in Botulinum Toxin Duration of Action? Mov. Dis. 19: S 23 -S 34. Fernandez-Salas, E. , L. E. Steward, H. Ho, P. E. Garay, S. W. Sun, M. A. Gilmore, J. V. Ordas, J. Wang, J. Francis, and K. R. Aoki. 2004. Plasma Membrane Localization Signals in the Light Chain of Botulinum Neurotoxin. Proc. Natl. Acad. Sci. USA. 101: 3208 -3213. Foran, P. G. , N. Mohammed, G. O. Lisk, S. Nagwaney, G. W. Lawrence, E. Johnson, L. Smith, K. R. Aoki, and J. O. Dolly. 2003. Evaluation of the Therapeutic Usefulness of Botulinum Neurotoxin B, C 1, E, and F Compared with the Long Lasting Type A: Basis for Distinct Durations of Inhibition of Exocytosis in Central Neurons. J. Biol. Chem. 278: 1363 -1371. Franciosa, G. , M. Pourshaban, A. De Luca, A. Buccino, B. Dallapiccola, and P. Aureli. 2004. Identification of Type A, B, E, and F Botulinum Neurotoxin Genes and of Botulinum Neurotoxigenic Clostridia by Denaturing High-Performance Liquid Chromatography. App Env Microbiol. 70: 4170 -4176.

References • • Hall, Y. H. J. , J. A. Chaddock, H. J. Moulsdale, E. R. Kirby, F. C. G. Alexander, J. D. Marks, and K. A. Foster. 2004. Novel Application of an in vitro Technique to the Detection and Quantification of Botulinum Neurotoxin Antibodies. J. Immun. Met. 288: 55 -60. Hong, B. , L. Jiang, Y. Hu, D. Fang, and H. Guo. 2004. J. Microbiol. Met. 58: 403 -411. Jathoul, A. P. , J. L. Holley, and H. S. Garmory. 2004. Efficacy of DNA Vaccines Expressing the Type F Botulinum Toxin Hc Fragment Using Different Promoters. Vaccine. 22: 3942 -3946. Johnston, M. D. , S. Lawson, and J. A. Otter. 2005. Evaluation of Hydrogen Peroxide Vapour as a Method for the Decontamination of Surfaces Contaminated with Clostridium botulinum Spores. J. Microbiol. Met. 60: 403 -411. Lacy, D. B. , W. Tepp, A. C. Cohen, B. R. Das. Gupta, and R. C. Stevens. 1998. Crystal Structure of Botulinum Neurotoxin Type A and Implications for Toxicity. Nat. Struct. Biol. 5: 898 -902. Lovenklev, J. E. Holst, E. Borch, and P. Radstrom. 2004. Relative Neurotoxin Gene Expression in Clostridium botulinum Type B, Determined Using Quantitative Reverse Transcription-PCR. App Env Microbiol. 70: 2919 -2927. Maksymowych, A. B. and L. L. Simpson. 1998. Binding and Transcytosis of Botulinum Neurotoxin by Polarized Human Colon Carcinoma Cells. J. Biol. Chem. 273: 21950 -21957.

References • • Maksymowych, A. B. , M. Reinhard, C. J. Malizio, M. C. Goodnough, E. A. Johnson, and L. L. Simpson. 1999. Pure Botulinum Neurotoxin Is Absorbed from the Stomach and Small Intestine and Produces Peripheral Neuromuscular Blockade. Infect. Immun. 67: 4708 -4712. Marvaud, J. C. , S. Raffestin, M. Gibert, and M. R. Popoff. 2000. Regulation of the Toxigenesis in Clostridium botulinum and Clostridium tetani. Biol. Cell. 92: 455 -457. Mc. Laughlin, J. B. , J. Sobel, T. Lynn, E. Funk, and J. P. Middaugh. 2004. Botulism Type E Outbreak Associated with Eating a Beached Whale, Alaska. Emerg. Infect. Dis. 10: 1685 -1687. Mendez-Eastman, S. K. 2003. BOTOX: A Review. Plast Surg Nurs. 23: 64 -70. Merrison, A. F. A. , K. E. Chidley, J. Dunnett, and K. A. Sieradzan. 2005. Lesson of the Week: Wound Botulism Associated with Subcutaneous Drug Use. BJM. 325: 1020 -1021. Park, J. and L. L. Simpson. 2003. Inhalational Poisoning by Botulinum Toxin and Inhalation Vaccination with Its Heavy-Chain Component. Infect & Immun. 71: 1147 -1154. Park, J. and L. L. Simpson. 2004. Progress Toward Development of an Inhalation Vaccine Against Botulinum Toxin. Expert Rev. Vacc. 3: 477487.

References • • Purkiss, J. , M. Welch, S. Doward, and K. Foster. 2000. Capsaicin-Stimulated Release of Substance P from Cultured Dorsal Root Ganglion Neurons: Involvement of Two Distinct Mechanisms. Biochem. Pharm. 59: 14031406. Raffestin, S. B. Dupuy, J. C. Marvaud, and M. R. Popoff. 2005. Bot. R/A and Tet. R are Alternative RNA Polymerase Sigma Factors Controlling the Expression of the Neurotoxin and Associated Protein Genes in Clostridium botulinum Type A and Clostridium tetani. Molec. Microbiol. 55: 235 -249. Rohrich, R. J. , J. E. Janis. , S. Fagien, and J. M. Stuzin. 2003. The Cosmetic Use of Botulinum Toxin. CME Plast. Reconstr. Surg. 112: S 177 -S 187. Sahai, A, M. Khan, C. J. Fowler, and P. Dasgupta. 2005. Botulinum Toxin for the Treatment of Lower Urinary Tract Symptoms: A Review. Neurourology. 24: 2 -12. Scott, A. B. 2004. Development of Botulinum Toxin Therapy. Dermatol. Clin. 22: 131 -133. Simpson, L. L. 1986. A Preclinical Evaluation of Aminopyridines as Putative Therapeutic Agents in the Treatment of Botulism. Infect. Immun. 52: 858862. Simpson, L. L. , A. B. Maksymowych, and S. Hao. 2001. The Role of Zinc Binding in the Biological Activity of Botulinum Toxin. J. Biol. Chem. 276: 2703427041.

References • • • Simpson, L. L. , H. Zepeda, and I. Ohishi. 1988. Partial Characterization of the Enzymatic Activity Associated with the Binary Toxin (Type C 2) Produced by Clostridium botulinum. Infect. Immun. 56: 24 -27. Simpson, L. L. , J. A. Coffield, and N. Bakry. 1993. Chelation of Zinc Antagonizes the Neuromuscular Blocking Properties of the Seven Serotypes of Botulinum Neurotoxin as well as Tetanus Toxin. J. Pharmacol. Exp. Ther. 267: 720 -727. Simpson, L. L. , Y. Kamata, and S. Kozaki. 1990. Use of Monoclonal Antibodies as Probes for the Structure and Biological Activity of Botulinum Neurotoxin. J. Pharmacol Exp Ther. 255: 227 -232. Sobel, J. , N. Tucker, A. Sulka, J. Mc. Laughlin, and S. Maslanka. 2004. Foodborne Botulism in the United States, 1990 -2000. Emerg. Infect. Dis. 10: 1606 -1611. Ting, P. T. and A. Freiman. 2004. The Story of Clostridium botulinum: from Food Poisoning to Botox. Clin. Med. 4: 258261. Truong, D. D. Duane, J. Jankovic, C. Singer, L. C. Seeberger, C. L. Comella, M. F. Lew, R. L. Rodnitzky, F. O. Danisi, J. P. Sutton, P. D. Charles, R. A. Hauser, G. L. Sheean. Efficacy and Safety of Botulinum Type A Toxin (Dysport) in Cervical Dystonia: Results of the First US Randomized, Double-blind, Placebo-controlled Study. 2005. Mov. Dis.

References • Yoneda, S. , M. Shimazawa, M. Kato, A. Nonoyama, Y. Torii, H. Nishino, N. Sugimoto, S. Kozaki, and H. Hara. 2004. Comparison of the Therapeutic Indexes of Different Molecular Forms of Botulinum Toxin Type A. Eur. J Pharm. 508: 223229. • Yowler, B. C. and C. Schengrund. 2004. Glycosphingolipids—Sweets for Botulinum Neurotoxin. Glycoconjugate. 21: 287 -293. • Zhang, L. , W. Lin, S. Li, and K. R. Aoki. 2003. Complete DNA Sequences of the Botulinum Neurotoxin Complex of Clostridium botulinum Type A-Hall (Allergan) Strain. Gene. 315: 21 -32.

Miscellaneous Info.

Bo. NT in Treating Lower UTIs • Describe disease • What toxin does as treatment • After effects of toxin – how change disorder • Show mechanism and data to support effects
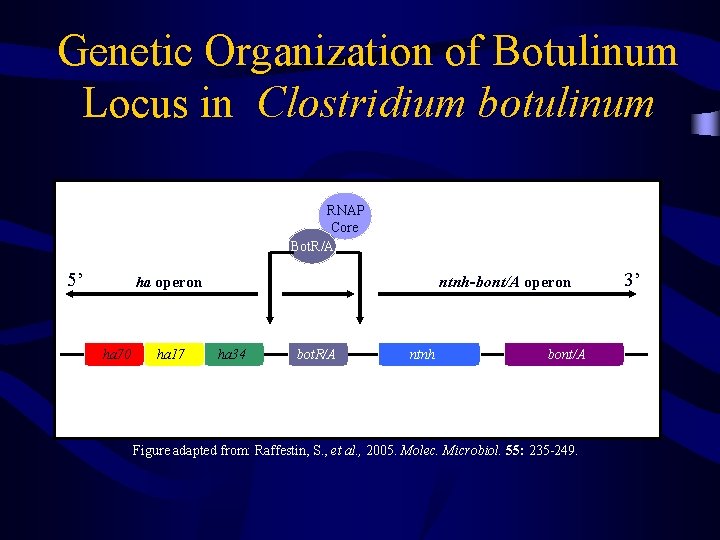
Genetic Organization of Botulinum Locus in Clostridium botulinum RNAP Core Bot. R/A 5’ ha operon ntnh-bont/A operon v ha 70 ha 17 ha 34 bot. R/A ntnh bont/A Figure adapted from: Raffestin, S. , et al. , 2005. Molec. Microbiol. 55: 235 -249. 3’
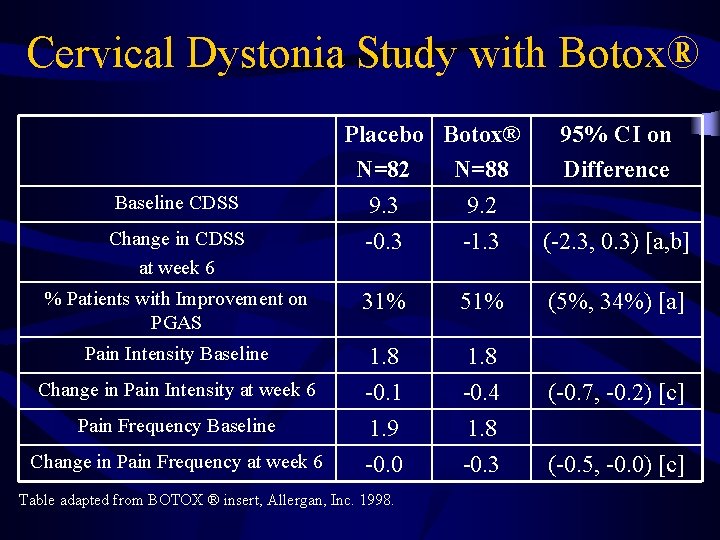
Cervical Dystonia Study with Botox® Baseline CDSS Placebo Botox® N=82 N=88 9. 3 9. 2 95% CI on Difference Change in CDSS at week 6 -0. 3 -1. 3 (-2. 3, 0. 3) [a, b] % Patients with Improvement on PGAS 31% 51% (5%, 34%) [a] Pain Intensity Baseline 1. 8 Change in Pain Intensity at week 6 -0. 1 1. 9 -0. 0 -0. 4 1. 8 -0. 3 Pain Frequency Baseline Change in Pain Frequency at week 6 Table adapted from BOTOX ® insert, Allergan, Inc. 1998. (-0. 7, -0. 2) [c] (-0. 5, -0. 0) [c]
Transcytosis of Bo. NT • Bo. NT targets gut epithelial – Absorptive enterocytes – M cells of Peyer’s Patches Figures taken from: Simpson. 2004. Annu. Rev. Pharmacol. Toxicol. 44: 161 -193.
Major Steps in Bo. NT Action Productive Internalization Binding Low/High Affinity Site Expose Occult Domains Endocytosis Insert Into Membrane Dissociation of Zinc Transcytosis Intracellular Poisoning (Cleavage Site) Cross Membrane Reduce Disulfide Bond Enzymatic Activity Binding Chain Separation (SNAP-25, VAMP Syntaxin) Renature Light Chain Reassociation of Zinc Figure adapted from: Simpson. 2004. Annu. Rev. Pharmacol. Toxicol. 44: 161 -193.
- Slides: 56