Clostridial fermentations n n Clostridia physiologically and phylogenically








- Slides: 15

Clostridial fermentations n n Clostridia: physiologically and phylogenically diverse group. 14 related families, at least 7 have organisms traditionally called Clostridium (Collins et al. IJSB 44: 812 -826, 1994) Genus characteristics: n n Gram positive Spore formers Strict anaerobes Butyric acid production.

Clostridia n Saccharolytic: swollen cells with subterminal spores n C. perfringens group n n Intestinal organisms Toxins, proteases Sugars or proteins Butyric acid group n n C. butyricum and C. pasteurianium Solvents by some

Clostridia n Tetani: amino acid users n n n C. tetani Terminal spores Putrefactive group n n n Strictly proteolytic C. sporogenes and C. botulinum Mercaptans, cadaverine, sulfides

Clostridia n Substrate-specific groups n n n Purines: C. acidi-urici, C. cylindrosporum Ethanol/acetate: C. kluyveri Acetogens: C. aceticum n n n Make perdominately acetic acid from sugars Some use hydrogen and carbon dioxide to make acetic acid Eubacterium, Peptostreptococcus are intestinal organisms

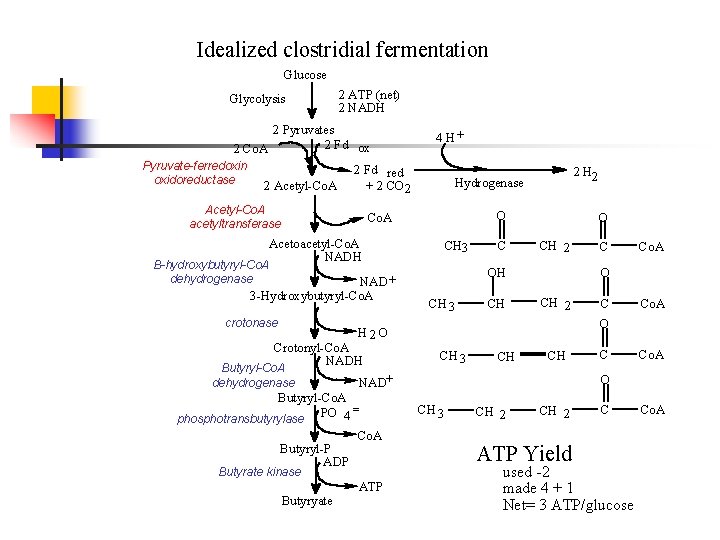
Idealized clostridial fermentation Glucose Glycolysis 2 ATP (net) 2 NADH 2 Pyruvates 2 Fd ox 2 Co. A Pyruvate-ferredoxin oxidoreductase 2 Acetyl-Co. A 4 H+ 2 Fd red + 2 CO 2 Acetyl-Co. A acetyltransferase O Co. A Acetoacetyl-Co. A NADH CH 3 Β-hydroxybutyryl-Co. A dehydrogenase NAD + 3 -Hydroxybutyryl-Co. A crotonase 2 Hydrogenase C O CH 2 OH CH 3 CH Butyryl-Co. A dehydrogenase CH 2 Butyryl-P ADP Co. A Butyrate kinase Butyryate ATP C Co. A O CH 3 CH CH NAD+ Butyryl-Co. A = phosphotransbutyrylase PO 4 Co. A O H 2 O Crotonyl-Co. A NADH C C Co. A O CH 3 CH 2 ATP Yield C used -2 made 4 + 1 Net= 3 ATP/glucose Co. A

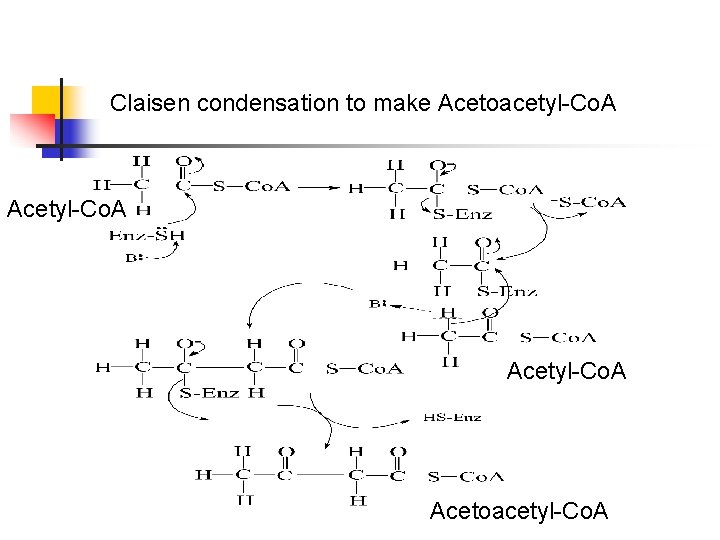
Claisen condensation to make Acetoacetyl-Co. A Acetyl-Co. A Acetoacetyl-Co. A

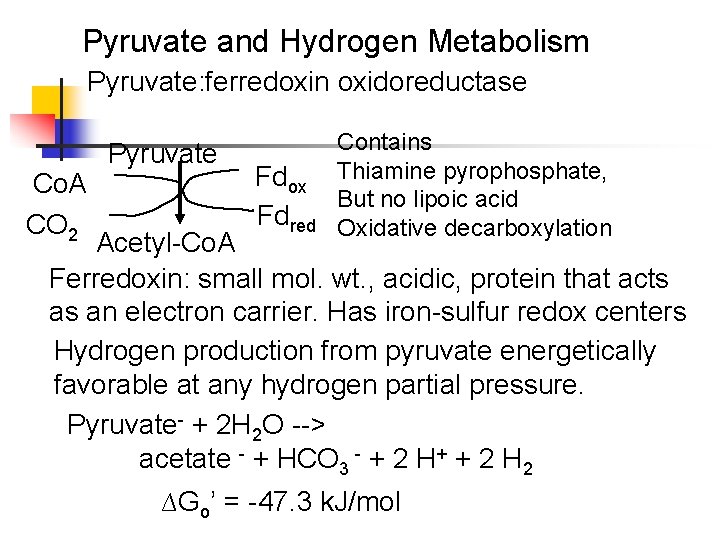
Pyruvate and Hydrogen Metabolism Pyruvate: ferredoxin oxidoreductase Co. A CO 2 Pyruvate Fdox Fdred Contains Thiamine pyrophosphate, But no lipoic acid Oxidative decarboxylation Acetyl-Co. A Ferredoxin: small mol. wt. , acidic, protein that acts as an electron carrier. Has iron-sulfur redox centers Hydrogen production from pyruvate energetically favorable at any hydrogen partial pressure. Pyruvate- + 2 H 2 O --> acetate - + HCO 3 - + 2 H+ + 2 H 2 ∆Go’ = -47. 3 k. J/mol

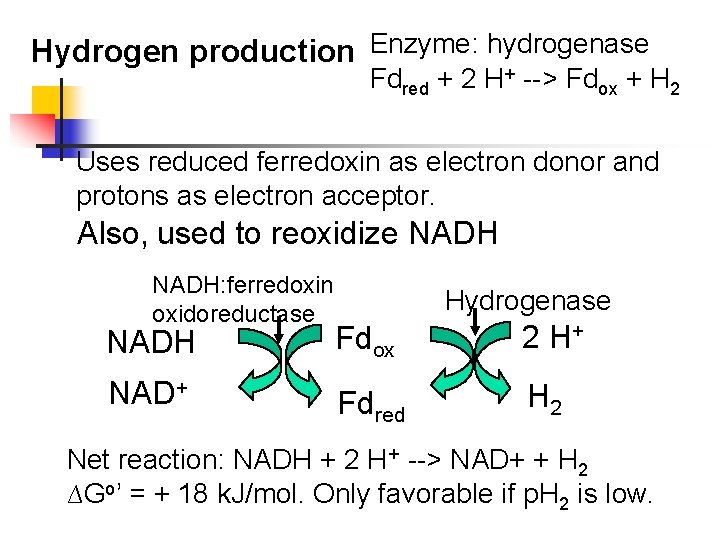
Hydrogen production Enzyme: hydrogenase Fdred + 2 H+ --> Fdox + H 2 Uses reduced ferredoxin as electron donor and protons as electron acceptor. Also, used to reoxidize NADH: ferredoxin oxidoreductase Hydrogenase NADH Fdox 2 H+ NAD+ Fdred H 2 Net reaction: NADH + 2 H+ --> NAD+ + H 2 ∆Go’ = + 18 k. J/mol. Only favorable if p. H 2 is low.

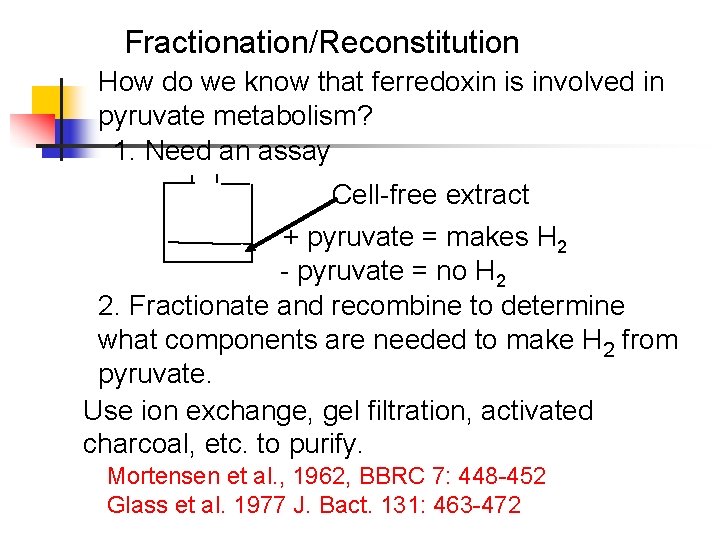
Fractionation/Reconstitution How do we know that ferredoxin is involved in pyruvate metabolism? 1. Need an assay Cell-free extract + pyruvate = makes H 2 - pyruvate = no H 2 2. Fractionate and recombine to determine what components are needed to make H 2 from pyruvate. Use ion exchange, gel filtration, activated charcoal, etc. to purify. Mortensen et al. , 1962, BBRC 7: 448 -452 Glass et al. 1977 J. Bact. 131: 463 -472

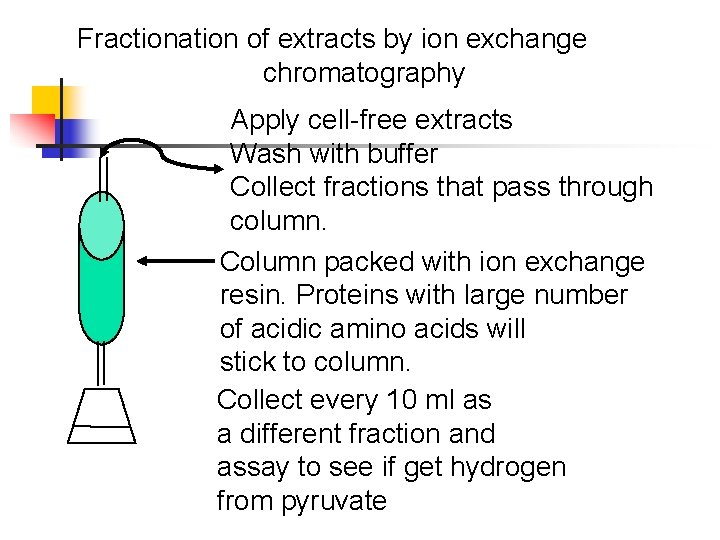
Fractionation of extracts by ion exchange chromatography Apply cell-free extracts Wash with buffer Collect fractions that pass through column. Column packed with ion exchange resin. Proteins with large number of acidic amino acids will stick to column. Collect every 10 ml as a different fraction and assay to see if get hydrogen from pyruvate

Assay results After ion exchange chromatography Hydrogen from pyruvate? DEAE-treated + pyr no H 2 - pyr no H 2 DEAE-treated with methyl viologen + pyr 6. 4 µmol H 2 - pyr no H 2 Methyl viologen: dye that serves as low potential electron carrier. Conclude: something was removed by chromatography that helps to make hydrogen.

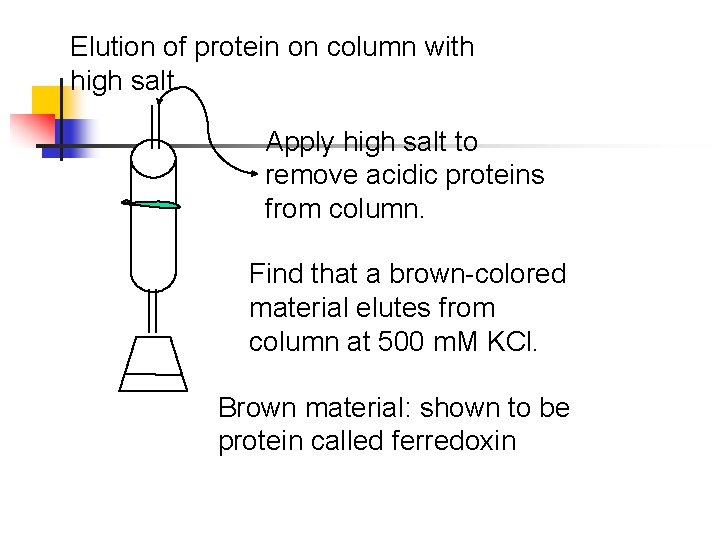
Elution of protein on column with high salt. Apply high salt to remove acidic proteins from column. Find that a brown-colored material elutes from column at 500 m. M KCl. Brown material: shown to be protein called ferredoxin

Assay results: After ion exchange chromatography and 500 m. M KCl treatment. Hydrogen from pyruvate? DEAE-treated + pyr no H 2 - pyr no H 2 DEAE-treated with brown material + pyr 6. 3 µmol H 2 - pyr no H 2 Addition of ferredoxin restored activity. Conclude: need ferredoxin to make hydrogen from pyruvate.

Clostridial fermentations n n In the idealized case, no acetate was formed since all acetyl-Co. A was used to make acetoacetyl-Co. A to reoxidize NADH In real fermentations, clostridia make a mixture of acetate and butyrate. How can this occur? By having NADH reoxidized by hydrogen production. Only favorable if hydrogen partial pressure is low

Summary n n n Butryic acid formation allows high ATP yield Claisen condensation: common mechanism in biosynthesis using acyl-Co. A substrates. Pyruvate: Fd oxidoreductase important enzyme in anaerobic metabolism Hydrogen production from NADH allow fermentative bacteria to maximize ATP gain. Fractionation/reconstitution is how most pathways were elucidated.