Clinical trials in Europe Professor Bruce Morland Birmingham

Clinical trials in Europe Professor Bruce Morland Birmingham, UK




Change of Paradigm in Oncology Drug Development & Era of High-throughput Technologies - When Biology Meets Clinics …. . ~ 1000 Anticancer compounds yearly under development Targeted anticancer compounds • • New mechanisms of action New profile of activity Distinct profile of toxicity Oral and prolonged administration
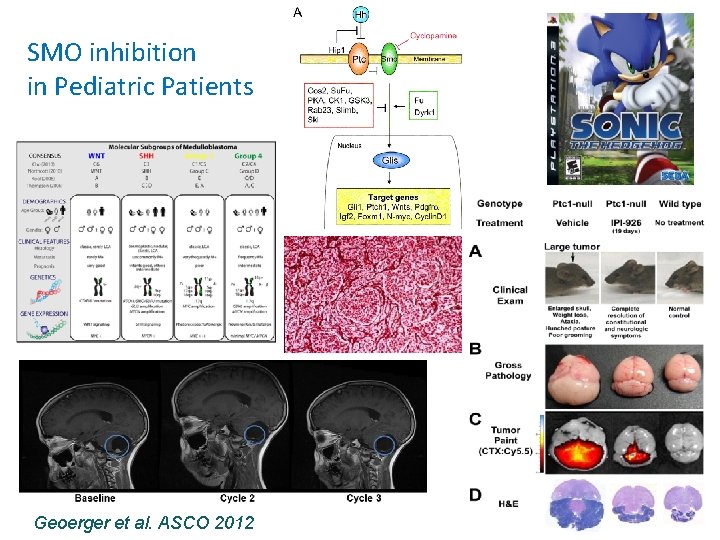
SMO inhibition in Pediatric Patients Geoerger et al. ASCO 2012
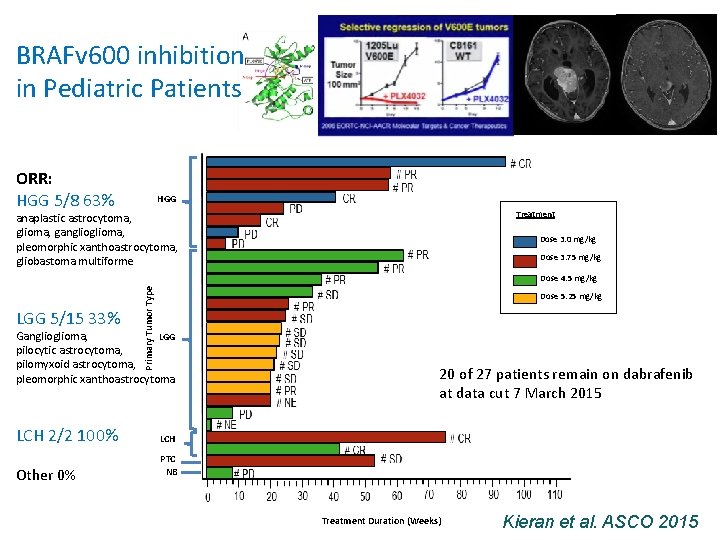
BRAFv 600 inhibition in Pediatric Patients ORR: HGG 5/8 63% HGG Treatment anaplastic astrocytoma, glioma, ganglioma, pleomorphic xanthoastrocytoma, gliobastoma multiforme Dose 3. 0 mg/kg Dose 3. 75 mg/kg LGG 5/15 33% Primary Tumor Type Dose 4. 5 mg/kg Dose 5. 25 mg/kg Ganglioma, LGG pilocytic astrocytoma, pilomyxoid astrocytoma, pleomorphic xanthoastrocytoma LCH 2/2 100% Other 0% 20 of 27 patients remain on dabrafenib at data cut 7 March 2015 LCH PTC NB Treatment Duration (Weeks) Kieran et al. ASCO 2015
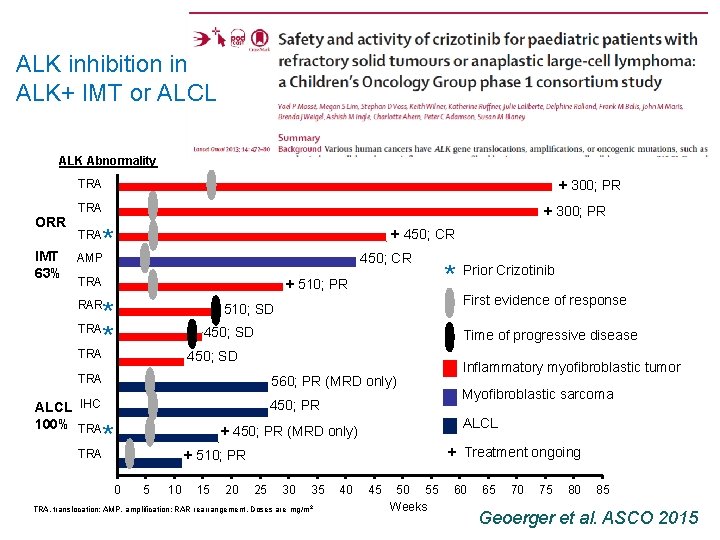
ALK inhibition in ALK+ IMT or ALCL ALK Abnormality + 300; PR TRA ORR IMT 63% TRA + 300; PR * AMP + 450; CR TRA + 510; PR * * TRA RAR 450; SD 560; PR (MRD only) ALCL IHC TRA Time of progressive disease 450; SD TRA 100% TRA First evidence of response 510; SD TRA 450; PR * 10 15 20 Myofibroblastic sarcoma + Treatment ongoing + 510; PR 5 Inflammatory myofibroblastic tumor ALCL + 450; PR (MRD only) 0 * Prior Crizotinib 25 30 35 TRA, translocation; AMP, amplification; RAR rearrangement, Doses are mg/m 2 40 45 50 55 Weeks 60 65 70 75 80 85 Geoerger et al. ASCO 2015
Hallmarks of Cancer
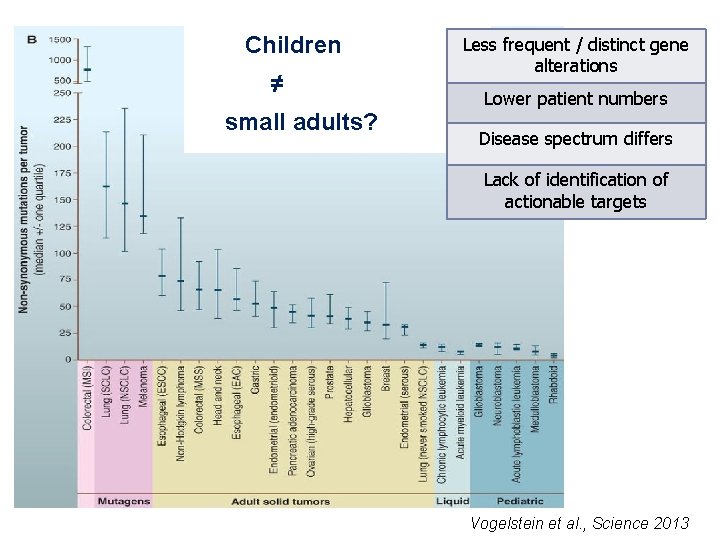
Children ≠ small adults? Less frequent / distinct gene alterations Lower patient numbers Disease spectrum differs Lack of identification of actionable targets Vogelstein et al. , Science 2013

Pediatric cancers are (relatively) simple … Courtesy of Stefan Pfister
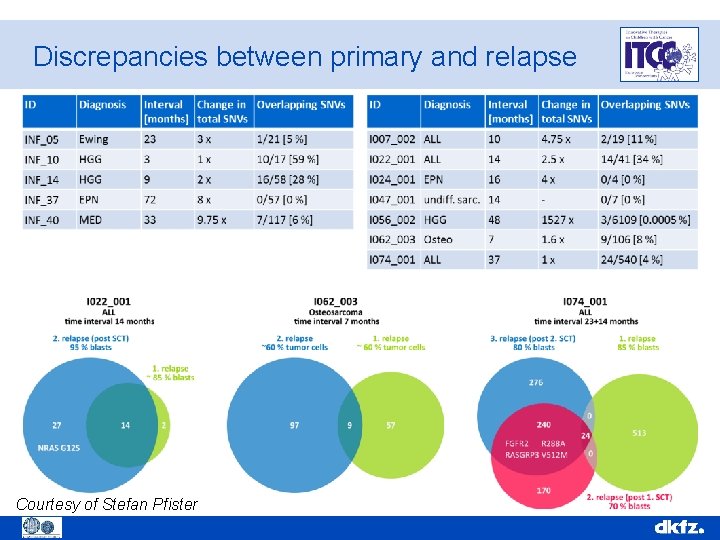
Discrepancies between primary and relapse Courtesy of Stefan Pfister
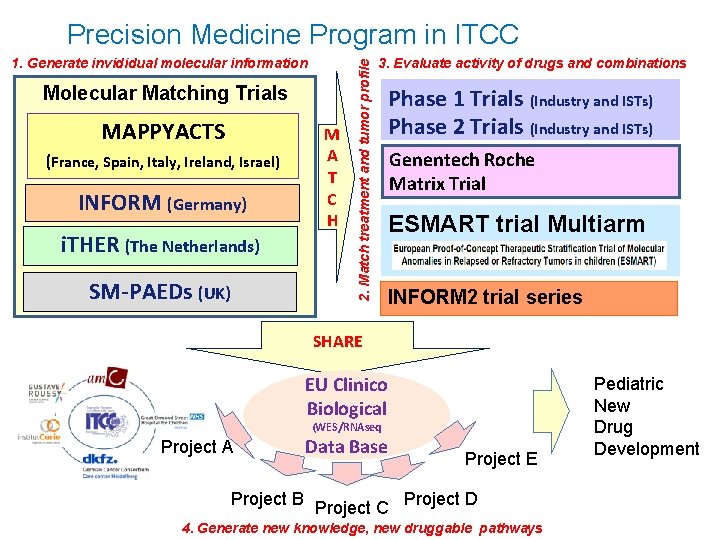
1. Generate invididual molecular information Molecular Matching Trials MAPPYACTS (France, Spain, Italy, Ireland, Israel) INFORM (Germany) i. THER (The Netherlands) SM-PAEDs (UK) M A T C H 2. Match treatment and tumor profile Precision Medicine Program in ITCC 3. Evaluate activity of drugs and combinations Phase 1 Trials (Industry and ISTs) Phase 2 Trials (Industry and ISTs) Genentech Roche Matrix Trial ESMART trial Multiarm INFORM 2 trial series SHARE EU Clinico Biological (WES/RNAseq Project A Project B Data Base Project C Project E Project D 4. Generate new knowledge, new druggable pathways Pediatric New Drug Development








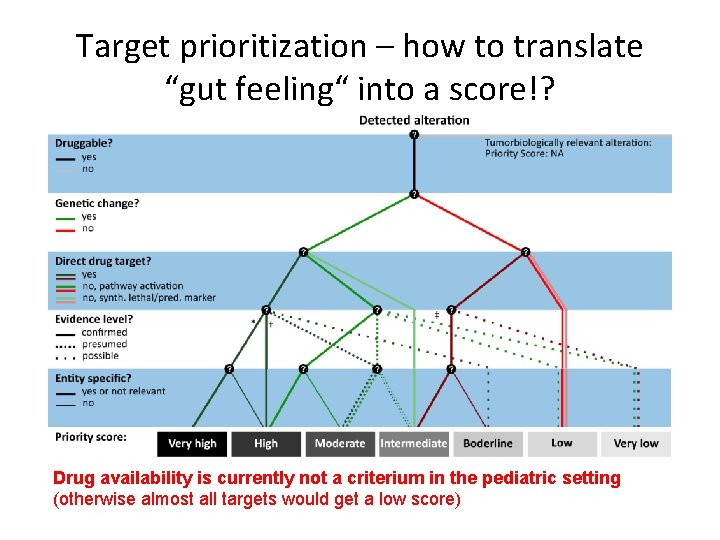
Target prioritization – how to translate “gut feeling“ into a score!? Drug availability is currently not a criterium in the pediatric setting (otherwise almost all targets would get a low score)






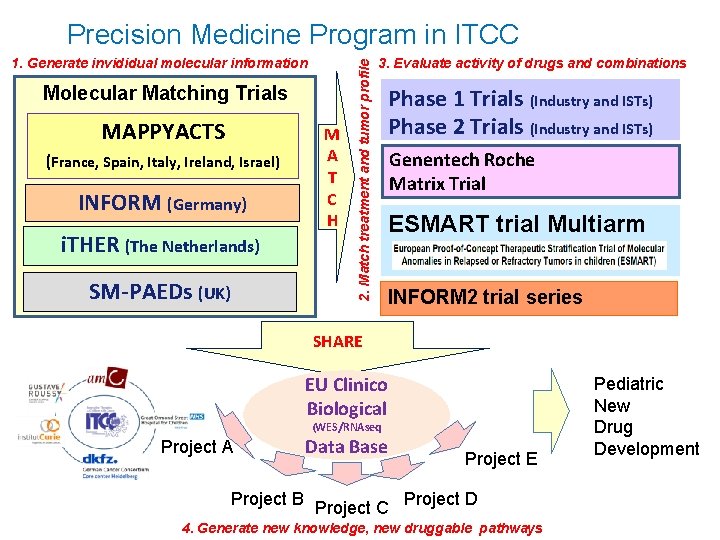
1. Generate invididual molecular information Molecular Matching Trials MAPPYACTS (France, Spain, Italy, Ireland, Israel) INFORM (Germany) i. THER (The Netherlands) SM-PAEDs (UK) M A T C H 2. Match treatment and tumor profile Precision Medicine Program in ITCC 3. Evaluate activity of drugs and combinations Phase 1 Trials (Industry and ISTs) Phase 2 Trials (Industry and ISTs) Genentech Roche Matrix Trial ESMART trial Multiarm INFORM 2 trial series SHARE EU Clinico Biological (WES/RNAseq Project A Project B Data Base Project C Project E Project D 4. Generate new knowledge, new druggable pathways Pediatric New Drug Development







Ewing, rhabdo, neuroblastoma, Wilms, hepatoblastoma on backbone of temozolomide /irinotecan


A phase I/II study of atezolizumab in pediatric and young adult patients with refractory/relapsed solid tumors (i. MATRIX-Atezolizumab). Geoerger et al. J Clin Oncol 35, 2017 (suppl; abstr 10524) • 74 patients (median age 14 years; range 2– 29) were enrolled: osteosarcoma, n = 12; Ewing sarcoma, n = 11; neuroblastoma, n = 11; rhabdomyosarcoma (RMS), n = 10; non-RMS soft tissue sarcoma, n = 10; Wilms tumor, n = 6; Hodgkin lymphoma (HL), n = 5; non-HL, n = 1; other tumor types, n = 8 • 17 patients (24%) had treatment-related grade 3– 4 AEs. One AE (grade 3 transaminase increase) led to study drug discontinuation. Common AEs were pyrexia (37%), fatigue (34%) and constipation (32%) • 2/5 patients with HL had a partial response (PR); the only patient with atypical rhabdoid tumor (RT) had an unconfirmed PR • Ongoing with rhabdoid/ATRT

Conclusions • ITCC is a successful academic partnership • Biology-led/precision medicine programme is deliverable • Partnership/collaboration with Pharma is key • Joint discussion with clinicians, regulators, pharma and parent groups, Accelerate has proven very successful (www. accelerateplatform. eu) • Portfolio expanding



- Slides: 43