CLINICAL TRIALS Fra MITO e REALT SILVIO GARATTINI






































- Slides: 61

CLINICAL TRIALS: Fra MITO e REALTÀ SILVIO GARATTINI Palermo 13 Aprile 2012

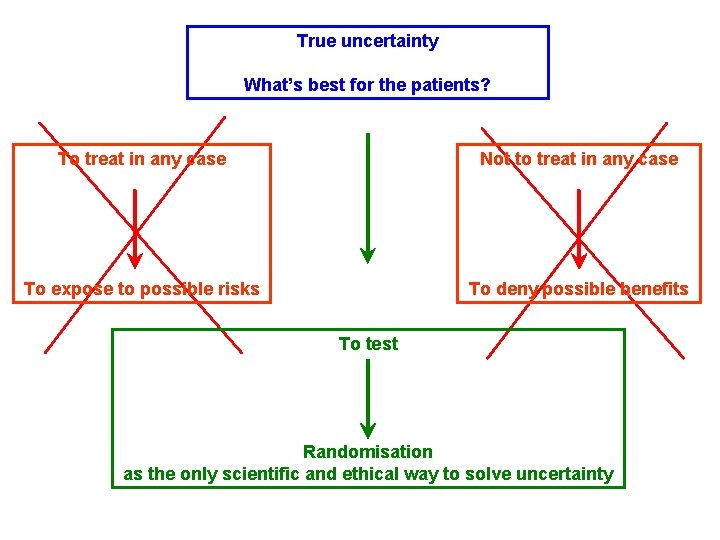
True uncertainty What’s best for the patients? To treat in any case Not to treat in any case To expose to possible risks To deny possible benefits To test Randomisation as the only scientific and ethical way to solve uncertainty

METHODOLOGICAL REQUIREMENTS FOR CLINICAL TRIALS Ask important questions… …answer them reliably The objective is the patient, the goal is his benefit Yusuf S, Collins R, Peto R. Why do we need some large, simple randomized trials? Stat Med 1984; 3: 409 -420

PATIENTS CAN BE INVOLVED IN CLINICAL TRIALS ONLY IF THERE IS A REASONABLE POTENTIAL ADVANTAGE. FOR THEM OR FUTURE PATIENTS THE ADVANTAGE COULD BE INCREASED EFFICACY, DECREASED TOXICITY, DIFFERENT TOXIC PROFILES, BETTER COMPLIANCE. LONGER DURATION OF ACTION, etc.

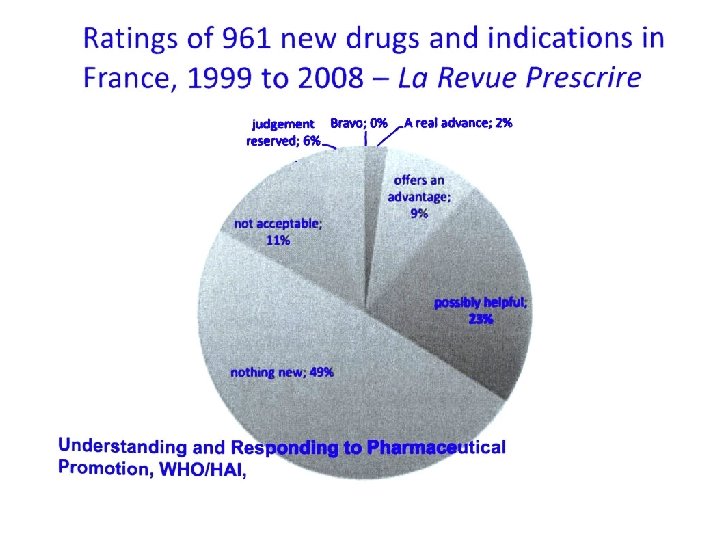
MIGLIORAMENTI INDOTTI DA 281 NUOVI FARMACI MEDIANA 0, 097 QALY 51% < 0, 1 QALY 12% > 1, 0 QALY Basic Clin Pharmacol 2009, 105, Suppl. 1, 032 Walker et al. , 2009


• THE EXCESSIVE USE OF PLACEBO

PLACEBO SHOULD BE USED ONLY WHEN THERE ARE NO EFFECTIVE DRUGS FOR A GIVEN INDICATION INAPPROPIATE USE OF PLACEBO MAY LEAD TO AN OPTIMISTIC EVALUATION OF A NEW DRUG

DENOSUMAB (ANTI-RANK LIGAND MAB) HAS BEEN TESTED AGAINST PLACEBO IN POST-MENOPAUSAL WOMEN WHEN THERE AVAILABLE SEVERAL ANTI-OSTEOPOROTIC AGENTS Scrip 19/9/08


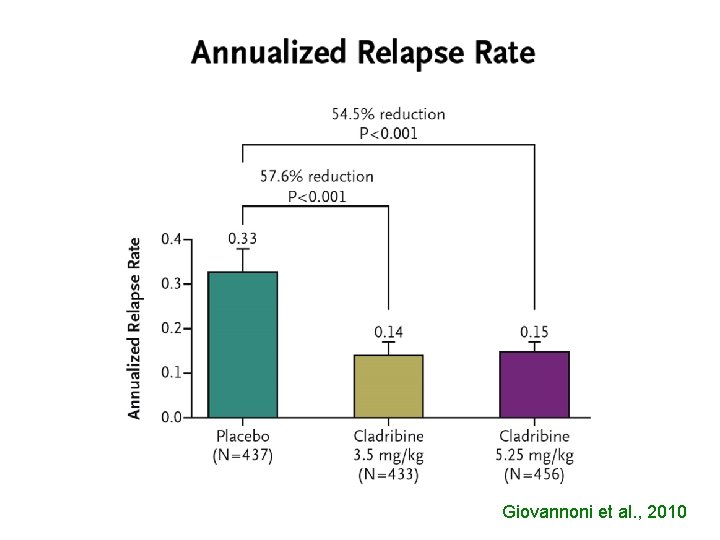
Giovannoni et al. , 2010


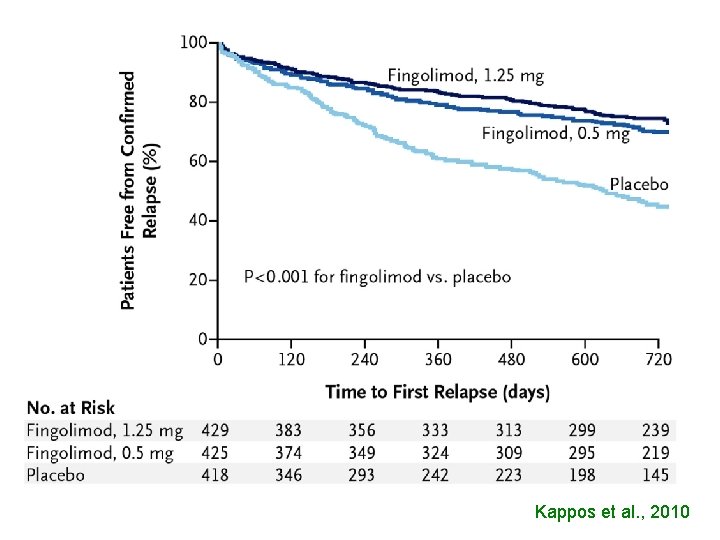
Kappos et al. , 2010

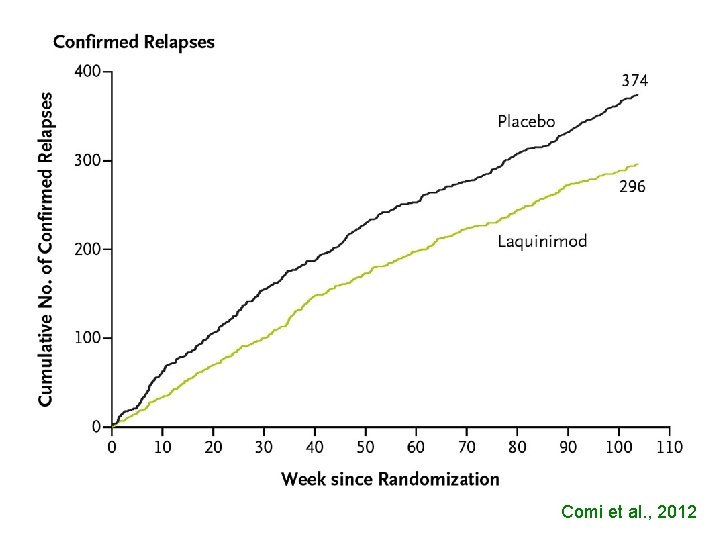
Placebo-Controlled Trial of Oral Laquinimod for Multiple Sclerosis Giancarlo Comi, M. D. , Douglas Jeffery, M. D. , Ludwig Kappos, M. D. , Xavier Montalban, M. D. , Alexey Boyko, M. D. , Maria A. Rocca, M. D. , and Massimo Filippi, M. D. , for the ALLEGRO Study Group* CONCLUSIONS In this phase 3 study, oral laquinimod administered once daily slowed the progression of disability and reduced the rate of relapse in patients with relapsing–remitting multiple sclerosis. (Funded by Teva Pharmaceutical Industries; Clinical. Trials. gov number, NCT 00509145. ) N ENGL J MED 2012. 366; 11 - 1000

Comi et al. , 2012

RELATIVE RISK REDUCTION OF RELAPSES IN RESPECT TO PLACEBO INFb-1 b 28 % (1995) INFb-1 a 32 % (1996) GLATIRAMER 29 % (1995)

EXCESS OF RELAPSES THAT COULD BE AVOIDED IF A COMPARATOR WOULD HAVE BEEN USED INSTEAD OF PLACEBO CLADRIBINE 79 DIRUCOTIDE 21 NATALIZUMAB 138 TERIFLUNOMIDE 123 FINGOLIMOD 100 LAQUIMOD 130 TOTAL RELAPSES 591 Bertelè et al. , 2012

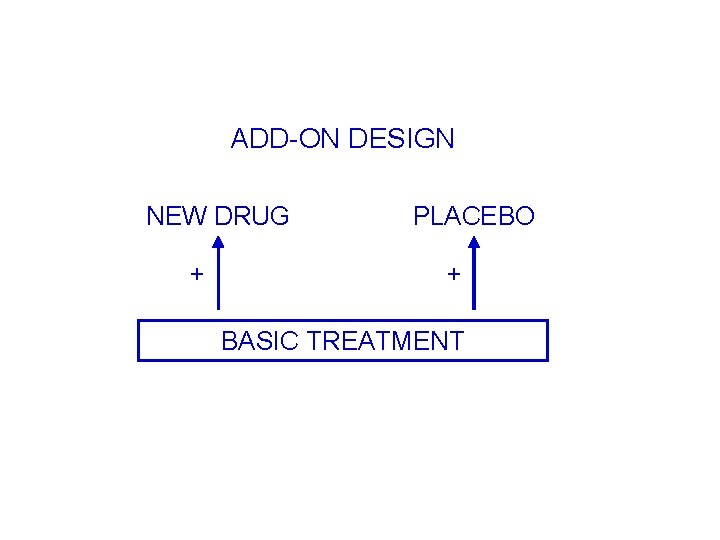
ADD-ON DESIGN NEW DRUG + PLACEBO + BASIC TREATMENT

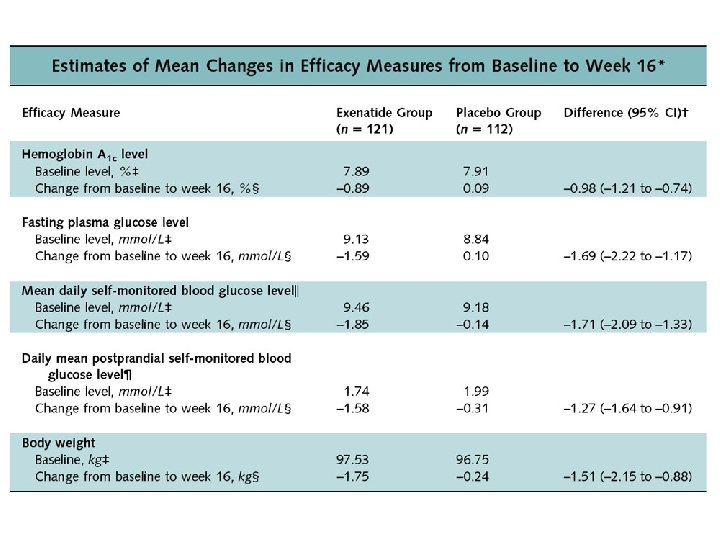
Zinman et al. , 2007


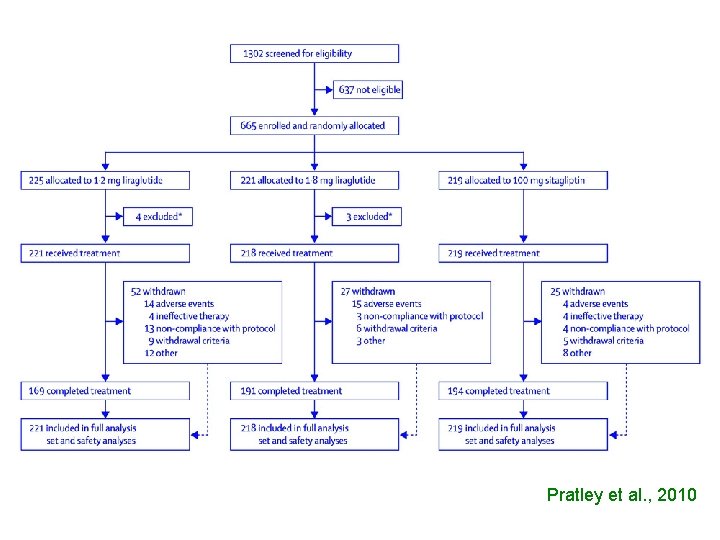
A FAIR COMPARISON WOULD HAVE BEEN TO USE INSTEAD OF PLACEBO ONE OF THE MANY ANTIDIABETIC AGENTS AVAILABLE ON THE MARKET

Pratley et al. , 2010

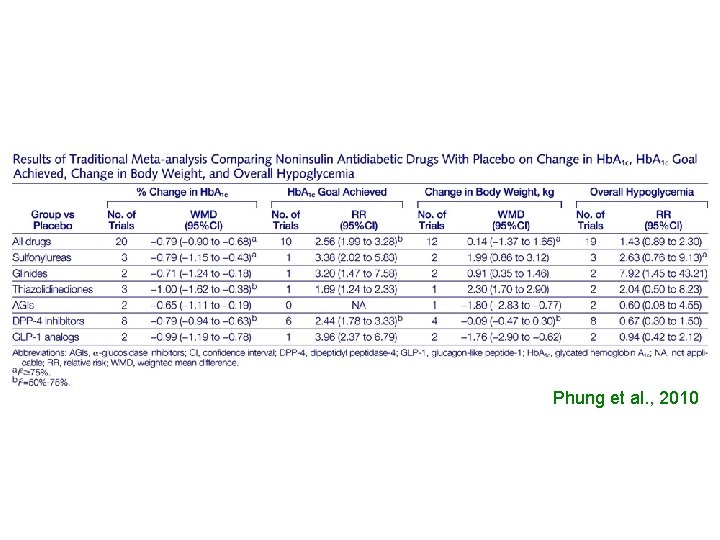
Effect of Noninsulin Antidiabetic Drugs Added to Metformin Therapy on Glycemic Control, Weight Gain, and Hypoglycemia in Type 2 Diabetes Olivia J. Phung, Pharm. D Jennifer M. Scholle, Pharm. D Mehak Talwar, BS Craig I. Coleman, Pharm. D Conclusion When added to maximal metformin therapy, all noninsulin antidiabetic drugs were associated with similar Hb. A 1 c reductions but differed in their associations with weight gain and risk of hypoglycemia. JAMA. 2010; 303(14): 1410 -1418

Phung et al. , 2010

• DESIGN OF CLINICAL TRIALS

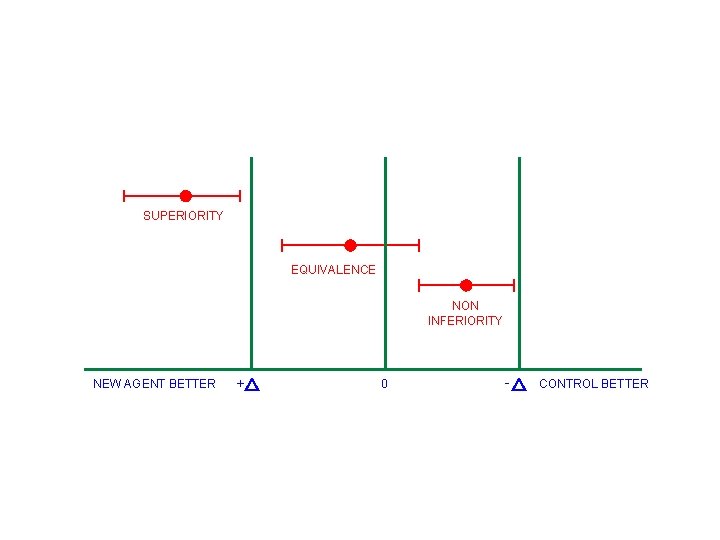
CLINICAL TRIALS MAY BE DESIGNED TO DEMONSTRATE • SUPERIORITY • EQUIVALENCE • NON INFERIORITY

IN THE NON-INFERIOR TYPE OF TRIAL WITH AN ACTIVE CONTROL, INVESTIGATORS ARE TESTING THE NULL HYPOTHESIS THAT A NEW DRUG IS WORSE THAN THE ACTIVE CONTROL (STANDARD) AND WHEN THEY CAN REJECT THE NULL HYPOTHESIS THEY ACCEPT THE ALTERNATIVE, THAT THE NEW DRUG IS NOT WORSE THAN THE ACTIVE CONTROL. ARAS, 2001 DRUG INFORMATION J. 35, 1157

SUPERIORITY EQUIVALENCE NON INFERIORITY NEW AGENT BETTER + 0 - CONTROL BETTER

Are there specific reasons for allowing a non-inferiority approach? • There may be non-responders to current treatments and products with comparable activity may offer a useful alternative. If the target is non-responders to current treatments, why not test their superiority over drugs with little effect in this subset of patients?

18 ANTICANCER AGENTS 21 INDICATIONS 12 9 ONLY PHASE III 6 EQUIVALENCE OR NON INFERIORITY 3 SUPERIORITY

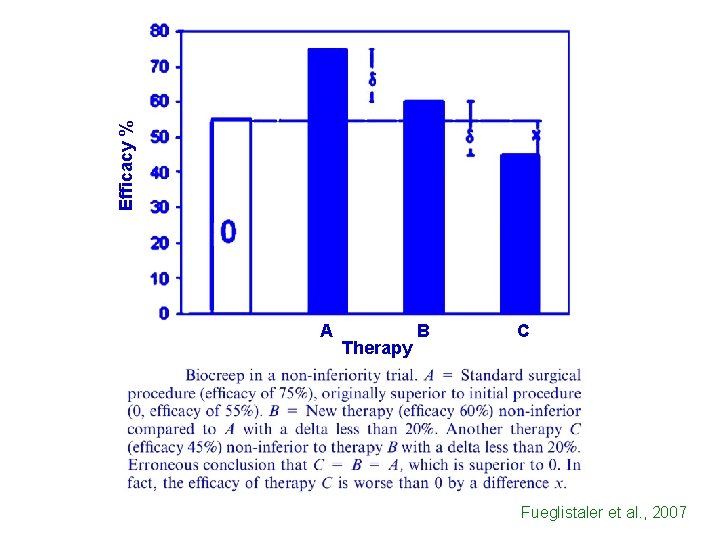
Efficacy % A Therapy B C Fueglistaler et al. , 2007

OUT OF 383 CLINICAL TRIALS 64 % COULD DETECT A DIFFERENCE > 50 % 84 % COULD DETECT A DIFFERENCE > 25 % MOHER et al. , 1994

Between 2002 and 2009, of 43 NDAs containing data from at least one noninferiority trial, the FDA approved 18 on the basis of pivotal evidence from noninferiority trials but found nine NDAs had poorly designed noninferiority trials that could not be used to establish the efficacy of the new drug. Nature Medicine 2010, 16, 1049

THE DESIGN OF N. I. TRAILS REQUIRES ALWAYS THE USE OF STANDARD AND PLACEBO WILE A SUPERIORITY DESIGN REQUIRES ONLY THE USE OF STANDARD.

Draft informed consent Garattini S, Bertele’ V. How can research ethics committees protect patients better? BMJ 2003; 326: 1199 -201 “Let us treat you with something that at best is the same as what you would have had before, but might also reduce - though this is unlikely - most of the advantages previously attained in your condition. It might even benefit you more than any current therapy but, should that actually happen, we shall not be able to prove it. Nor have we enough chance to let you know whether the new treatment may somehow bother or even harm you more than the standard one”.

NON-INFERIORITY TRIALS FEW PATIENTS WOULD AGREE TO PARTICIPATE IF THIS MESSAGE IS CLEAR IN THE INFORMED CONSENT FORM.

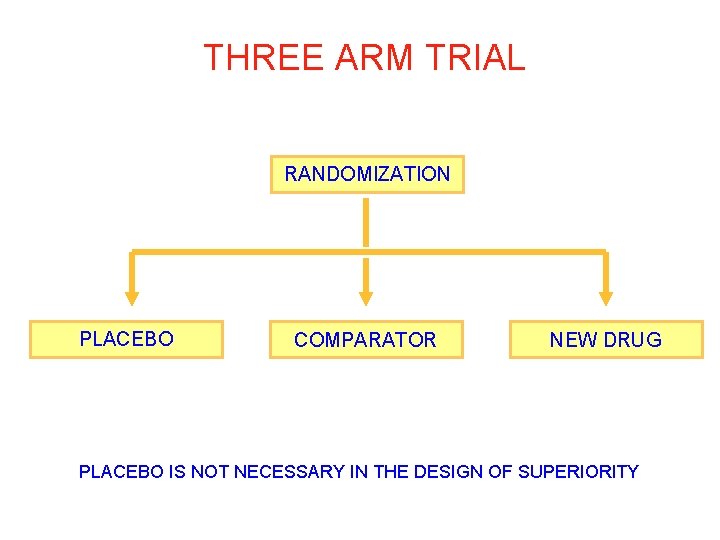
THREE ARM TRIAL RANDOMIZATION PLACEBO COMPARATOR NEW DRUG PLACEBO IS NOT NECESSARY IN THE DESIGN OF SUPERIORITY

• INADEQUATE COMPARATOR

TACROLIMUS VS CYCLOSPORINE 32. 5 % 51. 3 % ACUTE REJECTIONS ACUTE RENAL REJECTIONS ARE MINIMIZED WHEN TROUGH LEVELS ARE KEPT BETWEEN 330 - 430 ng/ml MARGREITER et al. , 2002 TRIAL CYCLOSPORINE < 300 ng/ml

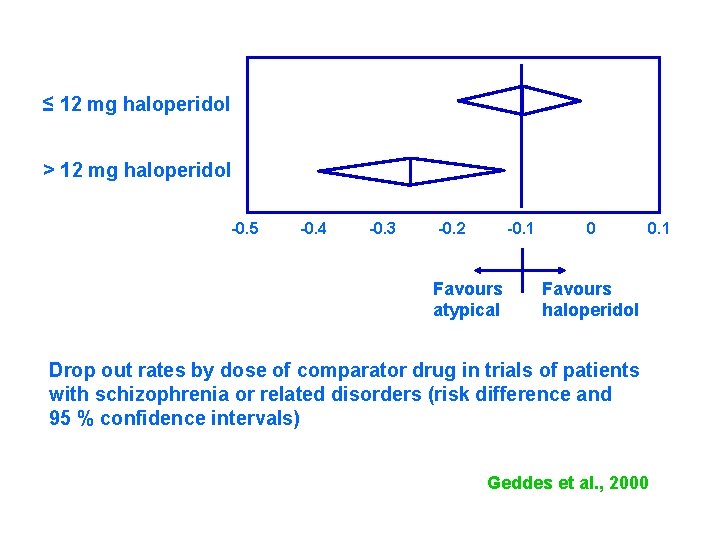
≤ 12 mg haloperidol > 12 mg haloperidol -0. 5 -0. 4 -0. 3 -0. 2 -0. 1 Favours atypical 0 0. 1 Favours haloperidol Drop out rates by dose of comparator drug in trials of patients with schizophrenia or related disorders (risk difference and 95 % confidence intervals) Geddes et al. , 2000

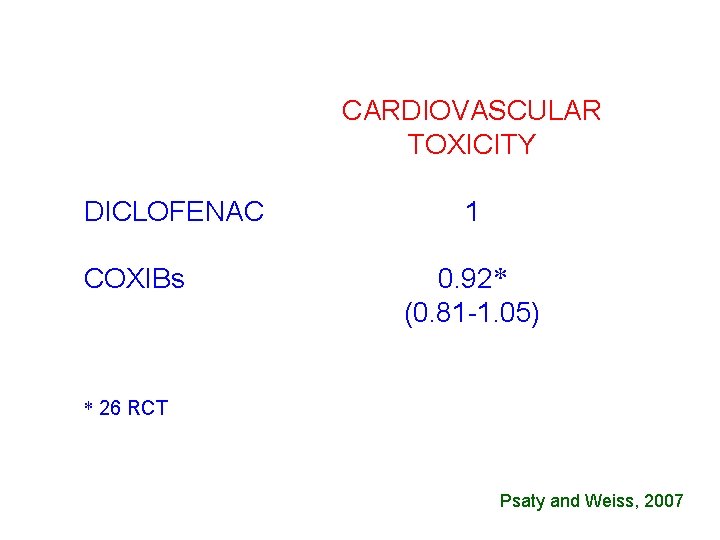
CARDIOVASCULAR TOXICITY DICLOFENAC COXIBs 1 0. 92* (0. 81 -1. 05) * 26 RCT Psaty and Weiss, 2007

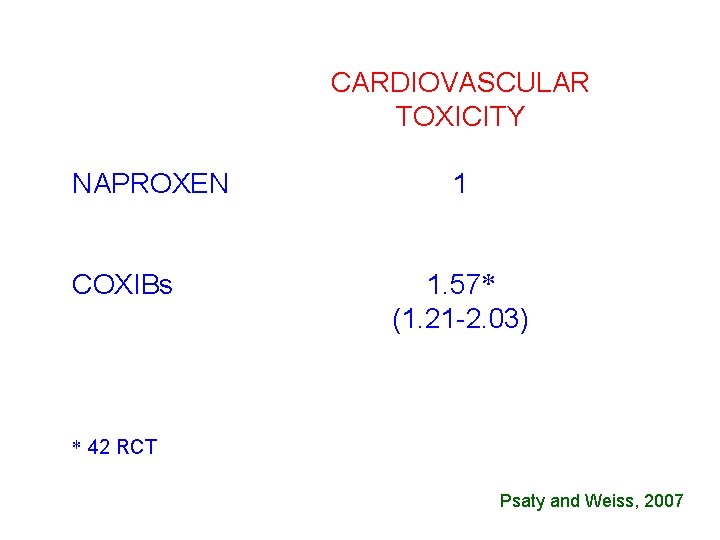
CARDIOVASCULAR TOXICITY NAPROXEN COXIBs 1 1. 57* (1. 21 -2. 03) * 42 RCT Psaty and Weiss, 2007

• SURROGATE END POINTS

QUALITY OF LIFE, MORBILITY, MORTALITY SHOULD BE THE FINAL OBJECTIVES OF CLINICAL TRIALS

• ENCAINIDE AND FLECAINIDE DECREASE ARRHYTHMIAS BUT INCREASE MORTALITY • OESTROGENS INCREASE HDL-CHOLESTEROL BUT DO NOT PREVENT CARDIOVASCULAR EVENTS • TORCETRAPIB INCREASES HDL-CHOLESTEROL BUT INCREASES MORTALITY

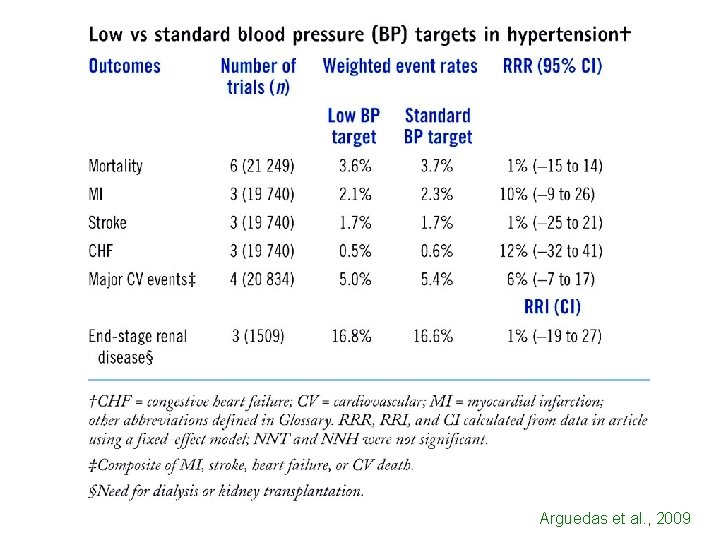
Hb. A 1 c IS NOT A SURROGATE END-POINT FOR CARDIOVASCULAR DISEASES IN TYPE 2 DIABETES SULFONYLUREAS Hb. A 1 C MYOCARDIAL INFARCTION ROSIGLITAZONE Hb. A 1 C HEART FAILURE NEED TO EVALUATE PARAMETERS IMPORTANT FOR PATIENTS

Arguedas et al. , 2009

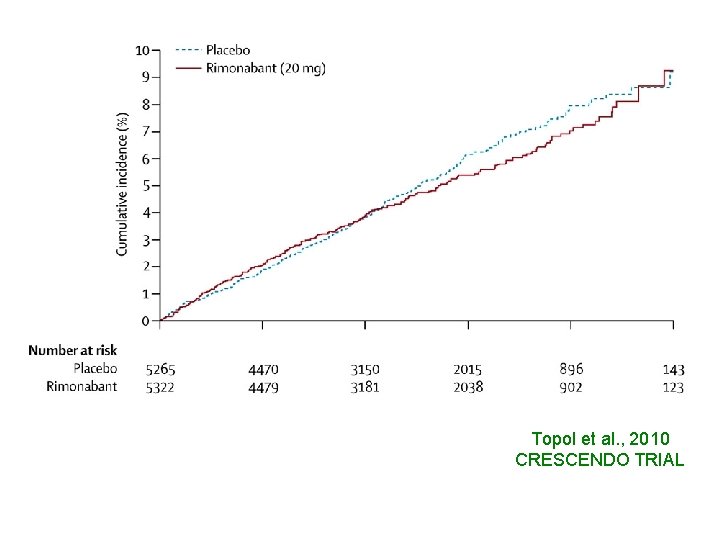
RIMONABANT antagonist of cannabinoid-1 receptor • DECREASE OF BODY WEIGHT • INCREASE OF HDL, ADIPONECTIN • DECREASE OF TRIGLYCERIDES, BLOOD GLUCOSE, FASTING INSULIN, LEPTIN • DECREASE OF TOTAL VOLUME ATHEROMA

Topol et al. , 2010 CRESCENDO TRIAL

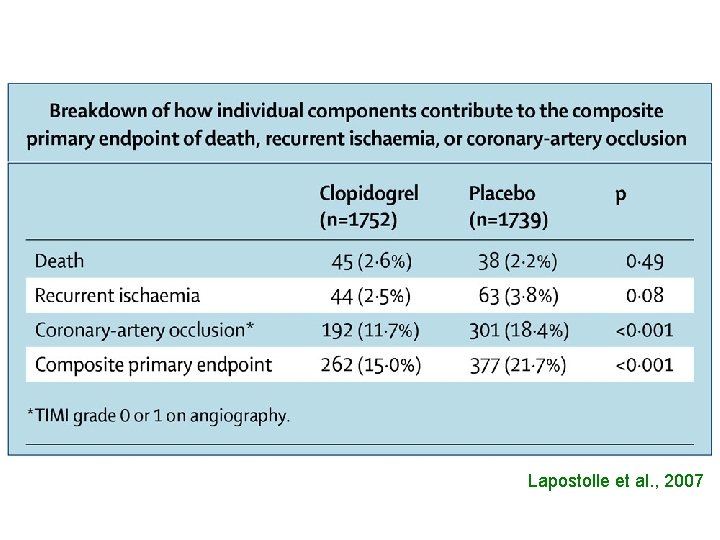
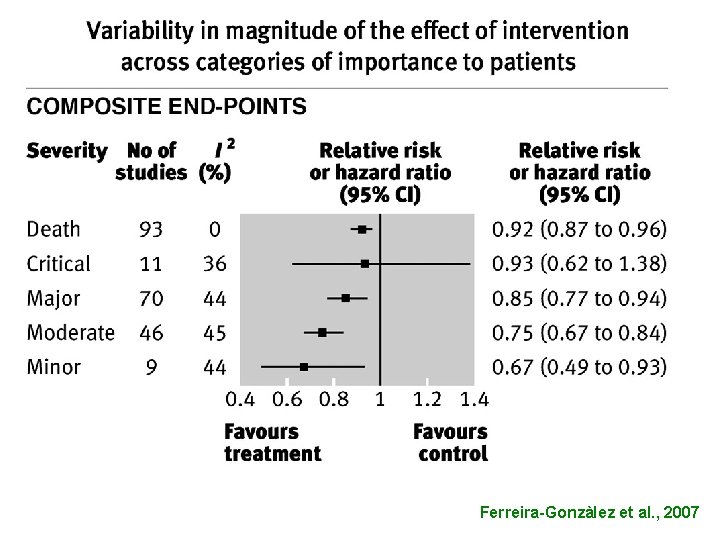
• UTILIZATION OF COMPOSITE END-POINTS

Lapostolle et al. , 2007

Ferreira-Gonzàlez et al. , 2007

• SELECTIVE PUBLICATIONS

SELECTIVE PUBLICATIONS OF 5 SSRI IN TOTAL 42 STUDIES • 21 STUDIES SHOW DRUG BETTER THAN PLACEBO • 19 PRIMARY PUBLICATIONS Sweedish Drug Regulatory Authority Melander et al. , BMJ 2003

SELECTIVE PUBLICATIONS OF 5 SSRI IN TOTAL 42 STUDIES • 21 STUDIES SHOW DRUG BETTER THAN PLACEBO • 19 PRIMARY PUBLICATIONS • 21 STUDIES SHOW NEGATIVE RESULTS • 6 PRIMARY PUBLICATIONS Sweedish Drug Regulatory Authority Melander et al. , BMJ 2003

SELECTIVE PUBLICATIONS OF 5 SSRI EVIDENCE B(I)ASED MEDICINE IN TOTAL 42 STUDIES • 21 STUDIES SHOW DRUG BETTER THAN PLACEBO • 19 PRIMARY PUBLICATIONS • 21 STUDIES SHOW NEGATIVE RESULTS • 6 PRIMARY PUBLICATIONS Sweedish Drug Regulatory Authority Melander et al. , BMJ 2003

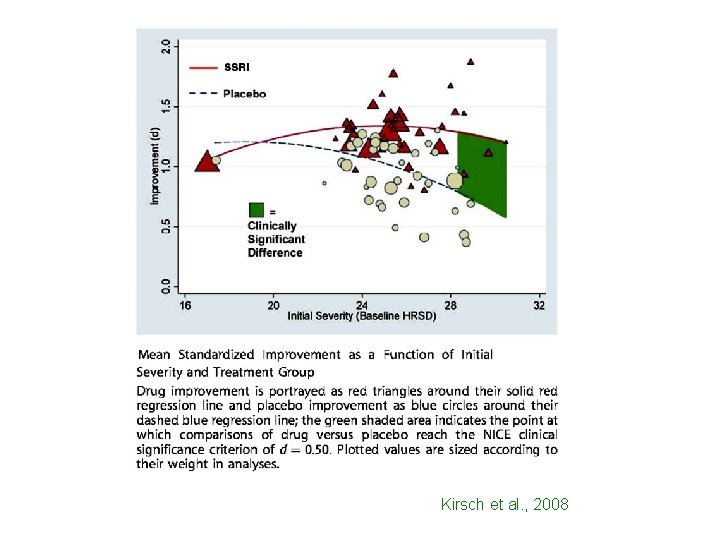
Kirsch et al. , 2008


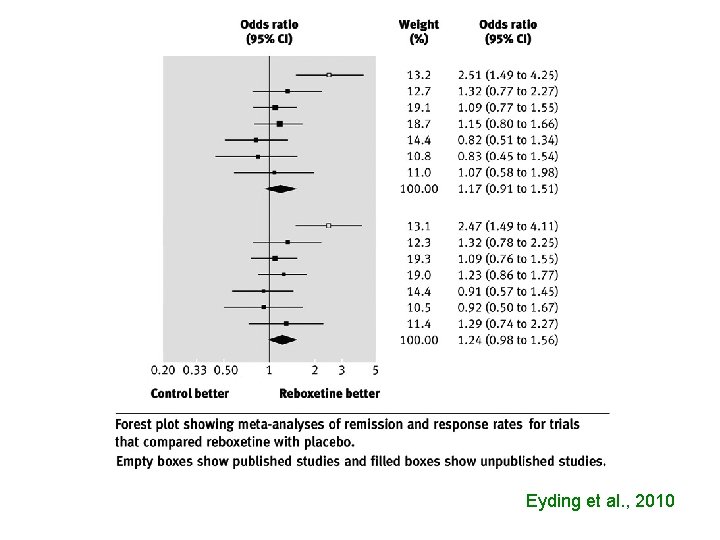
Eyding et al. , 2010

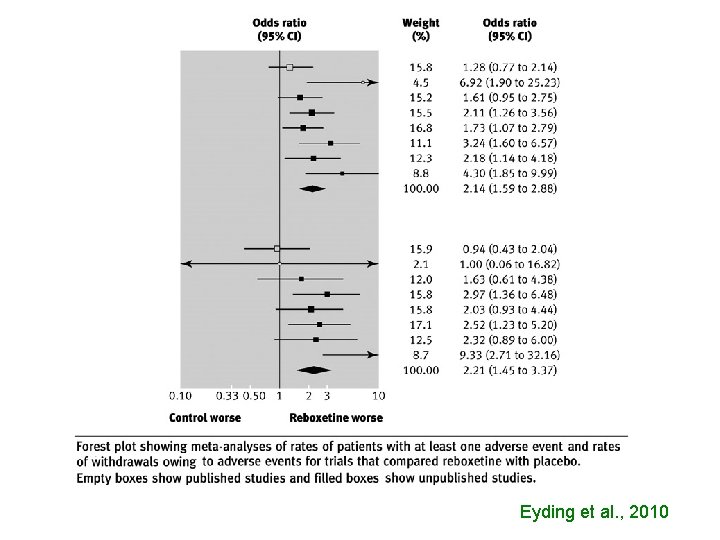
Eyding et al. , 2010

SELECTIVE REPORTING OF CLINICAL TRIALS MAY BIAS META-ANALYSES AND MISDIRECT GUIDE-LINE. IN A RETROSPECTIVE REVIEW OF 130 TRIALS, THOSE WITH POSITIVE RESULTS WERE THREE TIMES MORE LIKELY TO BE PUBLISHED WITH A SIGNIFICANT SHORTER TIME TO PUBLICATION THAN THOSE WITH NEGATIVE RESULTS. STERN, SIMES, 1997