Clinical Practice Guidelines Liver transplantation About these slides

Clinical Practice Guidelines Liver transplantation

About these slides • These slides give a comprehensive overview of the EASL clinical practice guidelines on liver transplantation • The guidelines were published in full in the February 2016 issue of the Journal of Hepatology – The full publication can be downloaded from the Clinical Practice Guidelines section of the EASL website – Please cite the published article as: European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Liver transplantation. J Hepatol 2016; 64: 433– 85 • Please feel free to use, adapt, and share these slides for your own personal use; however, please acknowledge EASL as the source

About these slides • Definitions of all abbreviations shown in these slides are provided within the slide notes • When you see a home symbol like this one: , you can click on this to return to the outline or topics pages, depending on which section you are in These slides are intended for use as an educational resource and should not be used in isolation to make patient management decisions. All information included should be verified before treating patients or using any therapies described in these materials • Please send any feedback to: slidedeck_feedback@easloffice. eu

Guideline panel • Chair: – Patrizia Burra • Panel members: – Andrew Burroughs, * Ivo Graziadei, Jacques Pirenne, Juan Carlos Valdecasas, Paolo Muiesan, Didier Samuel, Xavier Forns *Andrew Burroughs passed away during the preparation of this guideline, Giacomo Germani and Emmanuel Tsochatzis contributed to its completion EASL CPG LT. J Hepatol 2016; 64: 433– 85

Outline Methods Background Guidelines Appendix EASL CPG LT. J Hepatol 2016; 64: 433– 85 • Grading evidence • Introduction to liver transplantation • Key recommendations • Additional recommendations
Methods Grading evidence

Grading evidence • Grading is adapted from the GRADE system 1 Grade of recommendation I Randomized controlled trials II-1 Controlled trials without randomization II-2 Cohort or case-control analytic studies II-3 Multiple time series, dramatic uncontrolled experiments III Opinions of respected authorities, descriptive epidemiology 1. Guyatt GH, et al. BMJ 2008: 336: 924– 6; EASL CPG LT. J Hepatol 2016; 64: 433– 85

Background Introduction to liver transplantation
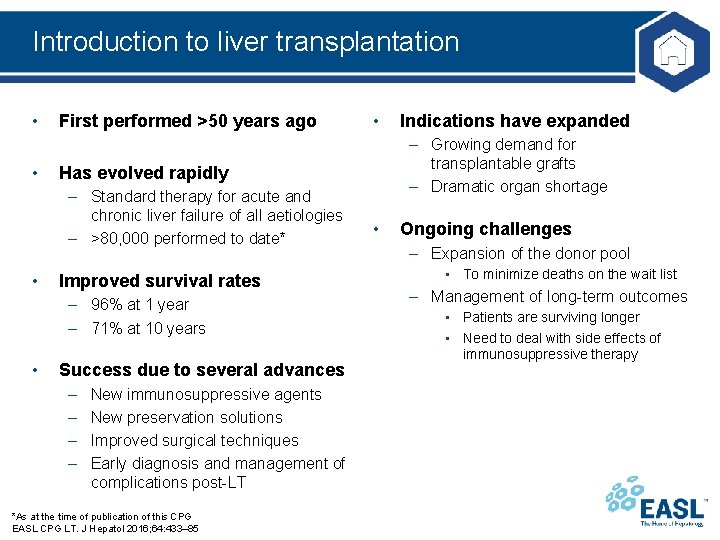
Introduction to liver transplantation • • First performed >50 years ago Improved survival rates – 96% at 1 year – 71% at 10 years • Success due to several advances – – New immunosuppressive agents New preservation solutions Improved surgical techniques Early diagnosis and management of complications post-LT *As at the time of publication of this CPG EASL CPG LT. J Hepatol 2016; 64: 433– 85 Indications have expanded – Growing demand for transplantable grafts – Dramatic organ shortage Has evolved rapidly – Standard therapy for acute and chronic liver failure of all aetiologies – >80, 000 performed to date* • • • Ongoing challenges – Expansion of the donor pool • To minimize deaths on the wait list – Management of long-term outcomes • Patients are surviving longer • Need to deal with side effects of immunosuppressive therapy

Guidelines Key recommendations

Topics 1. 2. 3. 4. 5. 6. The candidate for liver transplantation Organ donation Liver transplantation Immunosuppression Medical complications Lifestyle in long-term follow-up Click on a topic to skip to that section
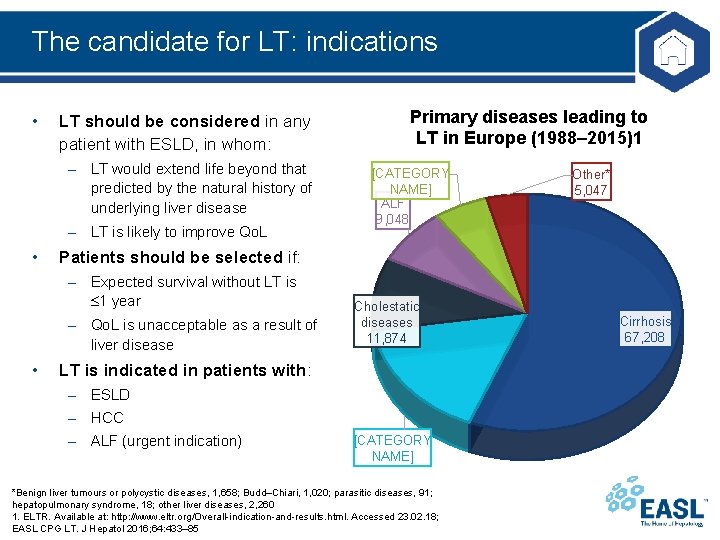
The candidate for LT: indications • LT should be considered in any patient with ESLD, in whom: – LT would extend life beyond that predicted by the natural history of underlying liver disease – LT is likely to improve Qo. L • [CATEGORY NAME] ALF 9, 048 Other* 5, 047 Patients should be selected if: – Expected survival without LT is 1 year – Qo. L is unacceptable as a result of liver disease • Primary diseases leading to LT in Europe (1988– 2015)1 Cholestatic diseases 11, 874 LT is indicated in patients with: – ESLD – HCC – ALF (urgent indication) [CATEGORY NAME] *Benign liver tumours or polycystic diseases, 1, 658; Budd–Chiari, 1, 020; parasitic diseases, 91; hepatopulmonary syndrome, 18; other liver diseases, 2, 260 1. ELTR. Available at: http: //www. eltr. org/Overall-indication-and-results. html. Accessed 23. 02. 18; EASL CPG LT. J Hepatol 2016; 64: 433– 85 Cirrhosis 67, 208

The candidate for LT: Score and prognostic factors for ESLD • Timing of LT is crucial – Before life-threatening complications occur – Not so early that benefits are outweighed by the risk of surgery and immunosuppression for life • Priority for LT is based on Child–Pugh classification and MELD – MELD score ≥ 15 is recommended to list patients with ESLD – Only MELD >35 predicts post-LT mortality • MELD does not reflect the impact of all complications – MELD exceptions – In some cases, extra points should be given to prioritize for LT EASL CPG LT. J Hepatol 2016; 64: 433– 85 Exceptions to MELD score Manifestations of cirrhosis • Refractory ascites • Recurrent gastrointestinal bleeding • Recurrent encephalopathy or chronic encephalopathy • Hepatopulmonary syndrome • Portopulmonary hypertension • Intractable pruritus Miscellaneous liver diseases • Budd–Chiari syndrome • Familial amyloidotic polyneuropathy • Cystic fibrosis • Hereditary haemorrhagic telangiectasia • Polycystic liver disease • Primary oxaluria • Recurrent cholangitis • Uncommon metabolic disease Malignancy • CCA, HCC • Uncommon liver tumours

The candidate for LT: patient evaluation • MELD score drives the allocation of grafts in many European countries – Final decision based on multiple factors • Suitable donors and local/regional priorities – Responsibility of multidisciplinary group within each expert centre after considering the risks and benefits for individual patients Recommendations Grade of recommendation Consider evaluation for LT when a major complication of cirrhosis occurs II-2 MELD score is good to predict short-term pre-transplant mortality risk II-1 MELD score can be used in organ allocation II-1 Experts should recognize and assign a different priority to patients requiring LT whose liver disease severity is not described by the MELD II-3/III HCC is a particular MELD exception that warrants giving extra points to access LT. These points have to be standardized in each country and have to take into account size, number of nodules, AFP levels and recurrence after downstaging therapy II-1 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Management of patients with liver cirrhosis* • Liver cirrhosis has different aetiologies – – HBV-related liver disease HCV-related liver disease Alcoholic liver disease NAFLD and NASH • Thus, collaboration between specialists is required to address all comorbidities • Final decision should be made within each expert centre by a multidisciplinary group of staff – Considering risks and benefits for individual patients *Without HCC EASL CPG LT. J Hepatol 2016; 64: 433– 85
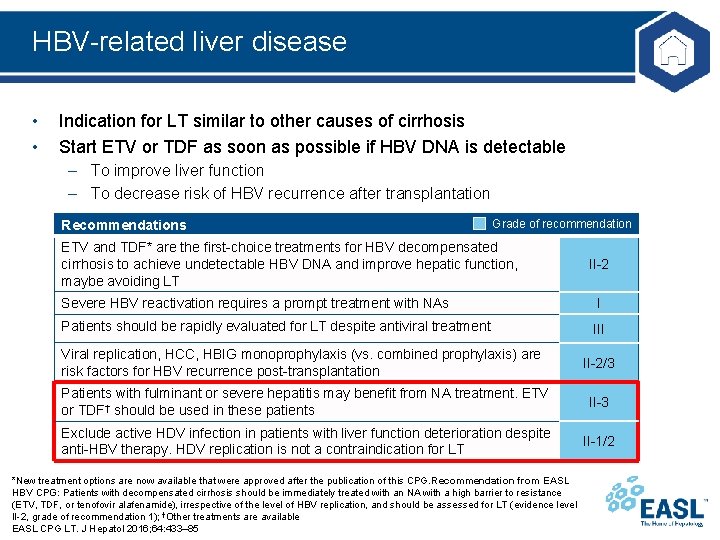
HBV-related liver disease • • Indication for LT similar to other causes of cirrhosis Start ETV or TDF as soon as possible if HBV DNA is detectable – To improve liver function – To decrease risk of HBV recurrence after transplantation Recommendations Grade of recommendation ETV and TDF* are the first-choice treatments for HBV decompensated cirrhosis to achieve undetectable HBV DNA and improve hepatic function, maybe avoiding LT II-2 Severe HBV reactivation requires a prompt treatment with NAs I Patients should be rapidly evaluated for LT despite antiviral treatment III Viral replication, HCC, HBIG monoprophylaxis (vs. combined prophylaxis) are risk factors for HBV recurrence post-transplantation II-2/3 Patients with fulminant or severe hepatitis may benefit from NA treatment. ETV or TDF† should be used in these patients II-3 Exclude active HDV infection in patients with liver function deterioration despite anti-HBV therapy. HDV replication is not a contraindication for LT II-1/2 *New treatment options are now available that were approved after the publication of this CPG. Recommendation from EASL HBV CPG: Patients with decompensated cirrhosis should be immediately treated with an NA with a high barrier to resistance (ETV, TDF, or tenofovir alafenamide), irrespective of the level of HBV replication, and should be assessed for LT (evidence level II-2, grade of recommendation 1); †Other treatments are available EASL CPG LT. J Hepatol 2016; 64: 433– 85
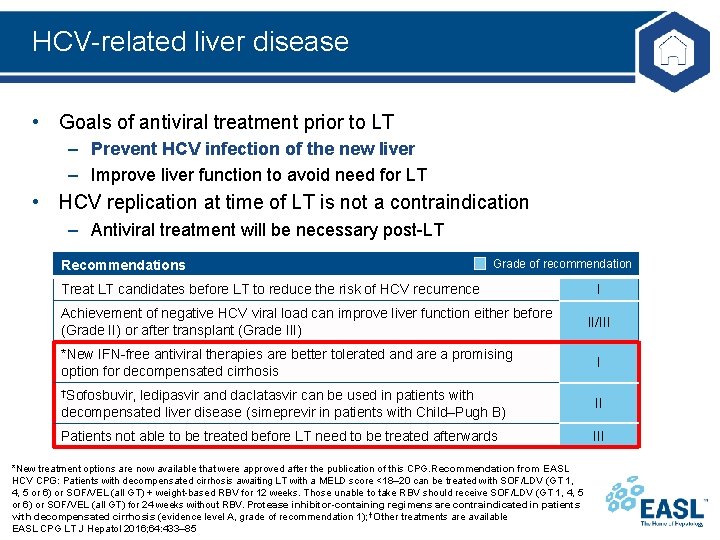
HCV-related liver disease • Goals of antiviral treatment prior to LT – Prevent HCV infection of the new liver – Improve liver function to avoid need for LT • HCV replication at time of LT is not a contraindication – Antiviral treatment will be necessary post-LT Recommendations Grade of recommendation Treat LT candidates before LT to reduce the risk of HCV recurrence Achievement of negative HCV viral load can improve liver function either before (Grade II) or after transplant (Grade III) *New IFN-free antiviral therapies are better tolerated and are a promising option for decompensated cirrhosis †Sofosbuvir, I II/III I ledipasvir and daclatasvir can be used in patients with decompensated liver disease (simeprevir in patients with Child–Pugh B) II Patients not able to be treated before LT need to be treated afterwards III *New treatment options are now available that were approved after the publication of this CPG. Recommendation from EASL HCV CPG: Patients with decompensated cirrhosis awaiting LT with a MELD score <18– 20 can be treated with SOF/LDV (GT 1, 4, 5 or 6) or SOF/VEL (all GT) + weight-based RBV for 12 weeks. Those unable to take RBV should receive SOF/LDV (GT 1, 4, 5 or 6) or SOF/VEL (all GT) for 24 weeks without RBV. Protease inhibitor-containing regimens are contraindicated in patients with decompensated cirrhosis (evidence level A, grade of recommendation 1); †Other treatments are available EASL CPG LT J Hepatol 2016; 64: 433– 85

Alcoholic liver disease • ALD is one of the most common indications for LT in the West • LT for alcoholic cirrhosis has a favourable outcome – Similar to other aetiologies of ESLD • Evaluation based on medical and psychiatric criteria to better determine patients most likely to benefit from LT – Psychosocial management is needed to ensure long-term success Recommendations Grade of recommendation A period of 6 months’ abstinence before the transplant could improve liver function, avoiding unnecessary LT, and could also improve compliance A psychiatric and psycho-sociological evaluation, and support pre- and post-LT is required for patients with alcoholic liver disease in need of LT LT can be offered to patients with acute alcoholic hepatitis non-responsive to steroid therapy. Nevertheless, the procedure should be done only in carefully selected patients EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-3 III II-2

NAFLD and NASH • Prevalence of NAFLD and NASH is increasing – Cirrhosis and liver failure associated with NAFLD and NASH are increasingly recognized as indications for LT • Patients should be monitored carefully for comorbid factors linked to metabolic syndrome – Could increase risk of complications during surgery Recommendations Grade of recommendation Assess and control comorbidities such as obesity, hypertension, diabetes and dyslipidaemia in the pre- and post-transplant setting, as they increase morbidity EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

Primary biliary cholangitis • Availability of UDCA has improved survival for patients with PBC – Number of LT candidates has dramatically decreased • LT is indicated in PBC – – When expected survival is <1 year In patients with decompensated cirrhosis In patients with complicated portal hypertension In patients with refractory and intolerable pruritus Recommendations Grade of recommendation In PBC patients, indication for liver transplantation should be given for decompensated liver disease, complicated portal hypertension, and for uncontrolled and intolerable pruritus refractory to all medical therapies EASL CPG LT J Hepatol 2016; 64: 433– 85 II-3

Primary sclerosing cholangitis • Suspected CCA may be an indication for LT – Can be a contraindication at an advanced stage • Patients transplanted with unsuspected CCA are at a high risk of recurrent CCA and have a poor prognosis • IBD is frequently associated with PSC – Not a contraindication for liver LT if quiescent – Active IBD should be controlled before transplantation Recommendations Grade of recommendation In PSC patients, decompensated liver disease, complicated portal hypertension and repeated episodes of cholangitis should be indications for LT PSC is a risk factor for CCA, thus, CCA should be excluded before LT Patients with PSC and ulcerative colitis should undergo colonoscopy annually before and after LT due to the higher risk of developing colon cancer EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-3 III II-3

Autoimmune hepatitis • AIH is more common in young women • Clinical presentation is variable – Usually presents as active chronic hepatitis – May also present as established cirrhosis – Rarely can present as a fulminant (acute) disease • Response to immunosuppressive treatment including steroids is good Recommendations Grade of recommendation LT is indicated in patients with decompensated cirrhosis due to AIH not responding to medical therapy, and in cases of fulminant AIH EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-3
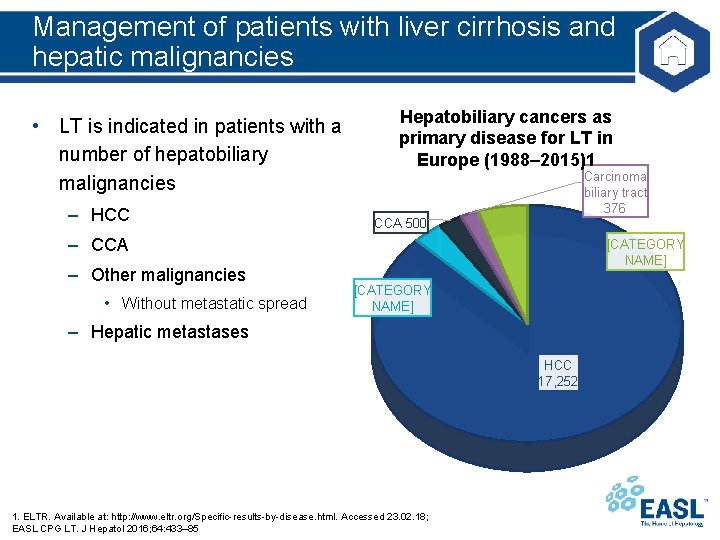
Management of patients with liver cirrhosis and hepatic malignancies • LT is indicated in patients with a number of hepatobiliary malignancies – HCC Hepatobiliary cancers as primary disease for LT in Europe (1988– 2015)1 Carcinoma biliary tract 376 CCA 500 – CCA – Other malignancies • Without metastatic spread [CATEGORY NAME] – Hepatic metastases HCC 17, 252 1. ELTR. Available at: http: //www. eltr. org/Specific-results-by-disease. html. Accessed 23. 02. 18; EASL CPG LT. J Hepatol 2016; 64: 433– 85

Liver transplantation in HCC • HCC is the most common primary tumour of the liver • LT is suitable for early, unresectable HCC – Particularly in the setting of CLD • 5 -year survival exceeds 70% when Milan criteria are used • Expanded criteria have shown similar outcomes – Identify LT candidates at low risk of recurrence otherwise excluded by Milan criteria remain the benchmark Milan criteria Solitary HCC <5 cm diameter OR ≤ 3 nodules with diameter <3 cm UCSF criteria 1 Single lesion ≤ 6. 5 cm in diameter OR 2– 3 lesions ≤ 4. 5 cm; total tumour diameter ≤ 8 cm AFP model 2 Points* Largest diameter, cm ≤ 3 3– 6 >6 0 1 4 Nodules, n 1– 3 ≥ 4 0 2 AFP level, ng/ml ≤ 100– 1, 000 >1, 000 0 2 3 *Total score ≤ 2 associated with low risk of recurrence and 5 -year survival ~70% 1. Yao FY, et al. Hepatology 2001; 33: 1394– 403; 2. Duvoux C, et al. Gastroenterology 2012; 143: 986– 94; EASL CPG LT. J Hepatol 2016; 64: 433– 85

Liver transplantation in HCC and CCA • The dropout rate from LT waiting lists is ~15– 30% due to HCC progression – Downstaging and bridging treatment should be offered to all patients expected to wait >6 months • CCA accounts for 5– 20% of primary liver tumours – LT for CCA remains controversial due to a high risk of recurrence Recommendations Grade of recommendation LT for HCC patients meeting Milan criteria* has an excellent outcome. An expansion of these criteria is acceptable if the recurrence-free survival is comparable. All new models should be compared to the Milan model LT is usually not recommended for cholangiocarcinoma or mixed hepatocellular/cholangiocarcinoma due to poor results. LT for perihilar cholangiocarcinoma could be offered in centres with clinical research protocols employing adjuvant or neoadjuvant therapy *Solitary HCC with diameter <5 cm or up to 3 nodules with diameter <3 cm EASL CPG LT. J Hepatol 2016; 64: 433– 85 I II-3

Comorbidities • All LT candidates require evaluation for comorbidities – – – CV, respiratory, renal Infections Nutrition Anatomy Neoplastic lesions Social assessment, psychiatric and addiction Recommendations • There is no formal age limit – Patients >65 years of age need a multidisciplinary evaluation • LT has been performed successfully in patients >70 years – Increased risk of CV complications Grade of recommendation Patients with an indication for LT should undergo an extensive work-up before their addition to the waiting list III No age limit of potential liver transplantation recipients is established, considering the good outcome of elderly patients. A multidisciplinary evaluation should always be performed in elderly patients to exclude comorbidities III EASL CPG LT. J Hepatol 2016; 64: 433– 85

Organ donation • Consent – Establishment of consent varies across Europe • Most European countries have presumed consent • Deceased and living donation – Also varies between countries • Including donation after brain death (DBD) and donation after circulatory death (DCD) • Waiting lists – Definition of listing criteria – Some bilateral and multilateral agreements exist EASL CPG LT. J Hepatol 2016; 64: 433– 85

Liver allocation in Europe • LT data are collected by the ELTR – Registry of all LT procedures – Link between transplant centres – Scientific exchange and publications • LT rates vary between countries – Organ allocation systems and organisations – Legislation – Organ donation rates – LT – Traditions • System diversity means there are no Europe-wide ‘rules’ 1. Adam R, et al. J Hepatol 2012; 57: 675– 88; EASL CPG LT. J Hepatol 2016; 64: 433– 85 Number of liver transplants performed in European countries (May 1968–December 2009)1

Donor classification • Deceased – DBD or DCD • • Living donor liver transplantation (LDLT) Extended criteria donor (ECD) – Organ with unfavourable characteristics associated with suboptimal post-LT outcomes – Fall into two main risk categories: poor graft function and potential for disease transmission • • Donor age >65 years Protocols to select ECD and DCD ICU stay with ventilation >7 days livers to improve safety BMI >30 and expand donor pool Steatosis of the liver >40% Serum sodium >165 mmol/L Aminotransferases: ALT >105 U/L, AST >90 U/L Serum bilirubin >3 mg/d. L EASL CPG LT. J Hepatol 2016; 64: 433– 85

Donation after circulatory death • DCD is a strategy to increase number of viable grafts – Recipients show mortality rates comparable to other risk predictors* Maastricht Criteria: categories of DCD Category I Dead on arrival. Tissue can be recovered from category I donors or any individuals who die in a hospital in a manner not suitable for solid organ recovery. There is no requirement for a precisely timed approach to tissue recovery Category II Unsuccessful cardiopulmonary resuscitation. These are patients who suffer a witnessed cardiac arrest outside the hospital and undergo unsuccessful CPR. When CPR fails in a medically suitable organ donor, uncontrolled organ donation is an option Category III Awaiting cardiac arrest following withdrawal of care. With appropriate permission, organs may be recovered after death from patients with irreversible brain injury or respiratory failure in whom treatment is withdrawn. Death is declared after a predetermined period Category IV Cardiac arrest after brain death. Rarely, a consented brain-dead donor has a cardiac arrest before scheduled organ recovery. Such category IV donors should either proceed as for a normal multiorgan retrieval – if this has already started – or should be managed as a category III donor as appropriate to the circumstances of cardiac arrest Category V Cardiac arrest in a hospital patient. Newly added in 2000, this category is made up of category II donors that originate in hospital. The distinction allows for improved tracking of the outcomes DCD is based on category III in most countries in Europe† *Such as advanced age, HCV, HCC, and older donor age; †Type II is predominant in France and Spain EASL CPG LT. J Hepatol 2016; 64: 433– 85
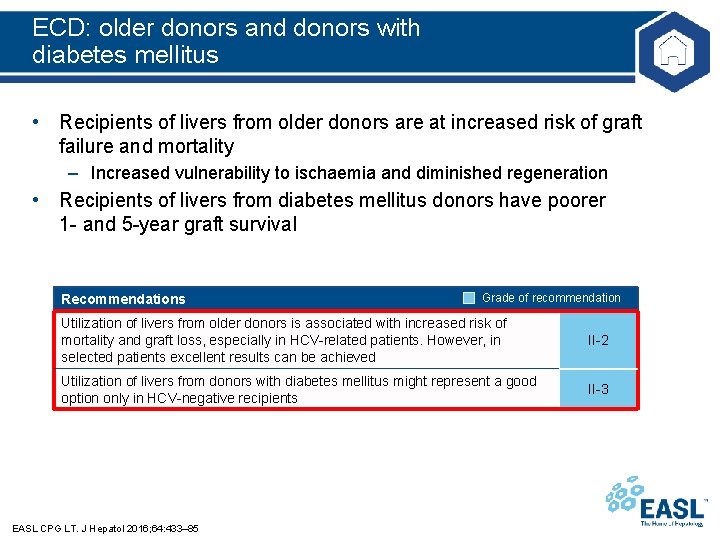
ECD: older donors and donors with diabetes mellitus • Recipients of livers from older donors are at increased risk of graft failure and mortality – Increased vulnerability to ischaemia and diminished regeneration • Recipients of livers from diabetes mellitus donors have poorer 1 - and 5 -year graft survival Recommendations Grade of recommendation Utilization of livers from older donors is associated with increased risk of mortality and graft loss, especially in HCV-related patients. However, in selected patients excellent results can be achieved II-2 Utilization of livers from donors with diabetes mellitus might represent a good option only in HCV-negative recipients II-3 EASL CPG LT. J Hepatol 2016; 64: 433– 85

ECD: donors with graft steatosis • Hepatic steatosis is associated with a range of post-LT complications – Poor graft function in particular • Steatosis is classified as mild, moderate or severe – Mild (10 30% steatosis): suitable for transplantation – Moderate (30 60%): acceptable outcomes in select DR combinations – Severe (>60%): unacceptable risk of graft failure, AKI, biliary complications and mortality Recommendation Grade of recommendation Grafts with microsteatosis or mild macrosteatosis are considered suitable for transplantation. Livers with moderate macrovesicular steatosis may result in acceptable outcomes in select donor-recipient combinations. Grafts with severe macrosteatosis should not be used as they are associated with increased risks of graft loss and mortality EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2
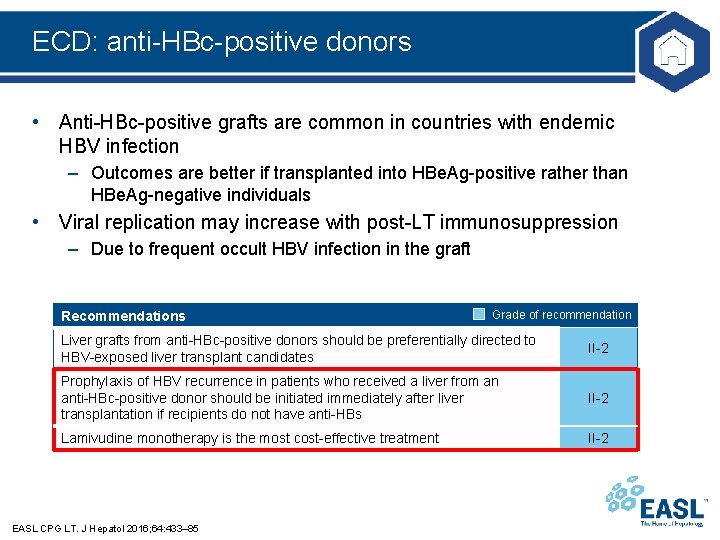
ECD: anti-HBc-positive donors • Anti-HBc-positive grafts are common in countries with endemic HBV infection – Outcomes are better if transplanted into HBe. Ag-positive rather than HBe. Ag-negative individuals • Viral replication may increase with post-LT immunosuppression – Due to frequent occult HBV infection in the graft Recommendations Grade of recommendation Liver grafts from anti-HBc-positive donors should be preferentially directed to HBV-exposed liver transplant candidates II-2 Prophylaxis of HBV recurrence in patients who received a liver from an anti-HBc-positive donor should be initiated immediately after liver transplantation if recipients do not have anti-HBs II-2 Lamivudine monotherapy is the most cost-effective treatment II-2 EASL CPG LT. J Hepatol 2016; 64: 433– 85

ECD: HCV-positive donors • Use of anti-HCV-positive grafts in recipients with HCV infection is commonplace and considered safe – LT of HCV-positive grafts into negative recipients is avoided Recommendation Grade of recommendation The use of anti-HCV-positive grafts in recipients with HCV infection is generally considered safe, whereas it should be avoided in HCV-negative recipients EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2

Liver transplantation • To overcome a shortage of grafts, alternative strategies need to be developed – Europe and USA: most common LT is ‘conventional’ or ‘standard’: Whole liver grafts – Asia: partial grafts from living donors • Whole liver grafts – Techniques differ depending on whether recipient IVC is preserved • Partial graft transplants – Auxiliary LT – Split LT – Living donor LT EASL CPG LT. J Hepatol 2016; 64: 433– 85
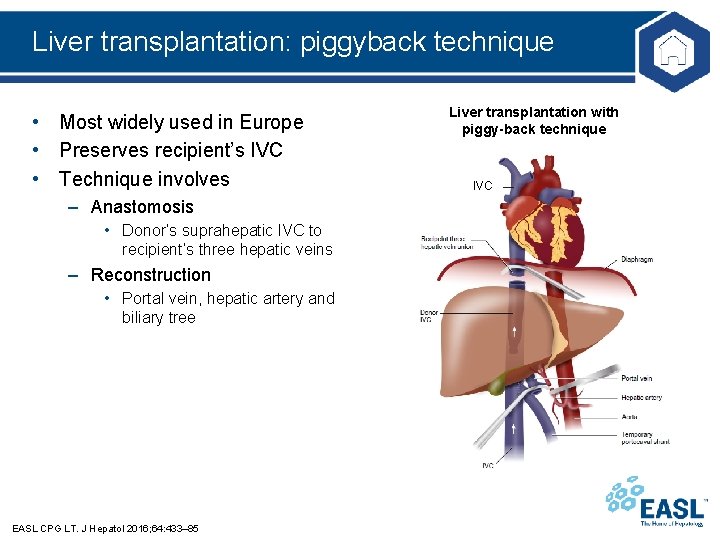
Liver transplantation: piggyback technique • Most widely used in Europe • Preserves recipient’s IVC • Technique involves – Anastomosis • Donor’s suprahepatic IVC to recipient’s three hepatic veins – Reconstruction • Portal vein, hepatic artery and biliary tree EASL CPG LT. J Hepatol 2016; 64: 433– 85 Liver transplantation with piggy-back technique IVC

Partial graft transplant: auxiliary LT • Auxiliary LT may be performed orthotopically or heterotopically and provides an alternative in two situations: – Patients with ALF • Provide support to patient’s diseased liver while it recovers • When native liver functions normally, graft is removed and immunosuppression is withdrawn – Patients with functional congenital or metabolic disorders affecting a normal liver • Allows correction of the metabolic disorder while avoiding a full LT • Poorer outcomes are obtained in Budd–Chiari syndrome and Wilson disease • Acute hepatitis B is a controversial indication – Risk of graft reinfection EASL CPG LT. J Hepatol 2016; 64: 433– 85
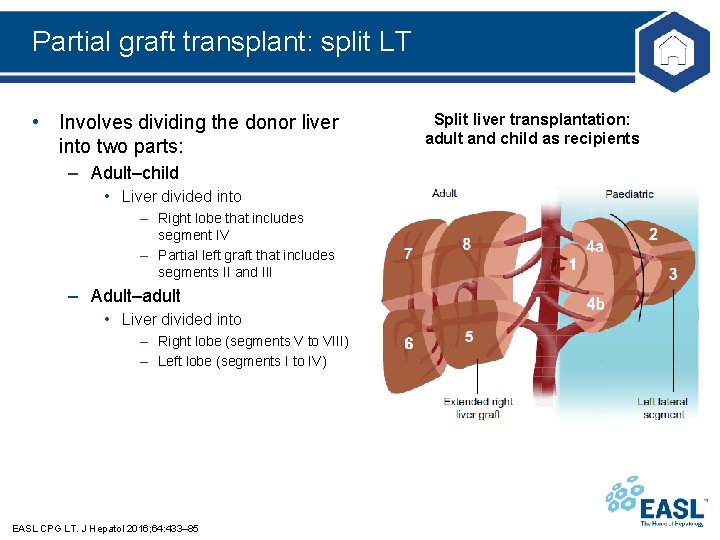
Partial graft transplant: split LT • Involves dividing the donor liver into two parts: – Adult–child • Liver divided into – Right lobe that includes segment IV – Partial left graft that includes segments II and III – Adult–adult • Liver divided into – Right lobe (segments V to VIII) – Left lobe (segments I to IV) EASL CPG LT. J Hepatol 2016; 64: 433– 85 Split liver transplantation: adult and child as recipients
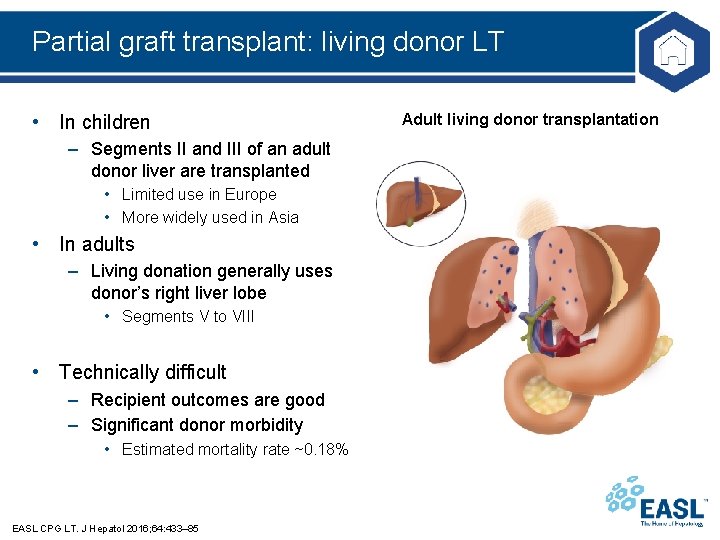
Partial graft transplant: living donor LT • In children – Segments II and III of an adult donor liver are transplanted • Limited use in Europe • More widely used in Asia • In adults – Living donation generally uses donor’s right liver lobe • Segments V to VIII • Technically difficult – Recipient outcomes are good – Significant donor morbidity • Estimated mortality rate ~0. 18% EASL CPG LT. J Hepatol 2016; 64: 433– 85 Adult living donor transplantation
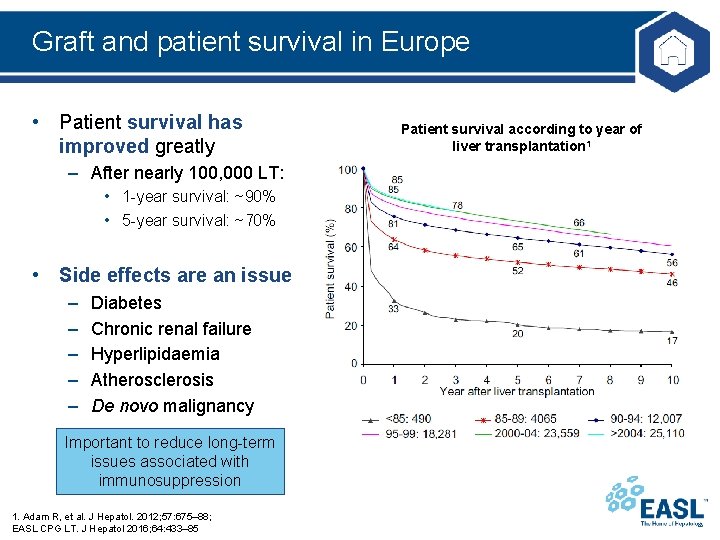
Graft and patient survival in Europe • Patient survival has improved greatly – After nearly 100, 000 LT: • 1 -year survival: ~90% • 5 -year survival: ~70% • Side effects are an issue – – – Diabetes Chronic renal failure Hyperlipidaemia Atherosclerosis De novo malignancy Important to reduce long-term issues associated with immunosuppression 1. Adam R, et al. J Hepatol. 2012; 57: 675– 88; EASL CPG LT. J Hepatol 2016; 64: 433– 85 Patient survival according to year of liver transplantation 1
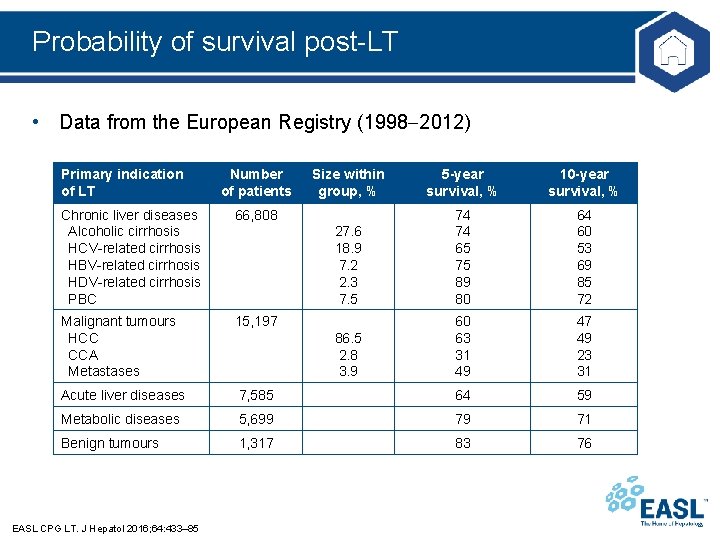
Probability of survival post-LT • Data from the European Registry (1998 2012) Primary indication of LT Number of patients Size within group, % 5 -year survival, % 10 -year survival, % 27. 6 18. 9 7. 2 2. 3 7. 5 74 74 65 75 89 80 64 60 53 69 85 72 86. 5 2. 8 3. 9 60 63 31 49 47 49 23 31 Chronic liver diseases Alcoholic cirrhosis HCV-related cirrhosis HBV-related cirrhosis HDV-related cirrhosis PBC 66, 808 Malignant tumours HCC CCA Metastases 15, 197 Acute liver diseases 7, 585 64 59 Metabolic diseases 5, 699 79 71 Benign tumours 1, 317 83 76 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Surgical complications • Although complications post-LT have been significantly reduced, they still have a major impact on short- and long-term prognoses Type Complication Incidence Vascular Arterial Hepatic artery thrombosis, most commonly presenting as graft dysfunction 1– 7% Venous Outflow obstruction by IVC anastomosis stenosis 1– 6% Portal vein thrombosis 2– 26% Biliary tract Leakage Very rarely embolism or tubules or reoperation are required ~5% Ischaemic bile duct injuries In patients receiving a DCD graft 15– 37% Recurrence of PSC 20– 30% Anastomotic stenosis (overall) 4– 9% Anastomotic stenosis (partial liver grafts) 5– 50% EASL CPG LT. J Hepatol 2016; 64: 433– 85
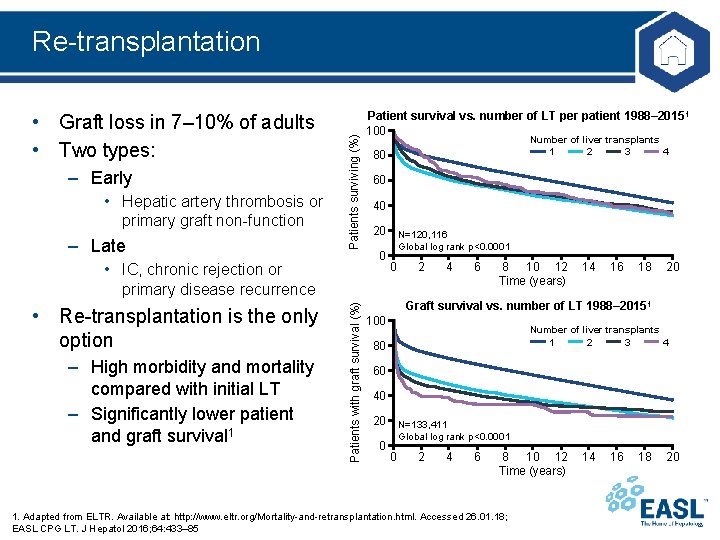
• Graft loss in 7– 10% of adults • Two types: – Early • Hepatic artery thrombosis or primary graft non-function – Late Patients surviving (%) Re-transplantation • Re-transplantation is the only option – High morbidity and mortality compared with initial LT – Significantly lower patient and graft survival 1 Patients with graft survival (%) • IC, chronic rejection or primary disease recurrence Patient survival vs. number of LT per patient 1988– 20151 100 Number of liver transplants 1 2 3 4 80 60 40 20 0 N=120, 116 Global log rank p<0. 0001 0 2 4 6 8 10 12 Time (years) 14 16 18 20 Graft survival vs. number of LT 1988– 20151 100 Number of liver transplants 1 2 3 4 80 60 40 20 0 N=133, 411 Global log rank p<0. 0001 0 2 4 6 8 10 12 Time (years) 1. Adapted from ELTR. Available at: http: //www. eltr. org/Mortality-and-retransplantation. html. Accessed 26. 01. 18; EASL CPG LT. J Hepatol 2016; 64: 433– 85 14 16 18 20

Immunosuppression: standard regimens • Spontaneous resolution of severe acute rejection episodes has been described in patients after LT – Due to the ‘privileged’ immunological status of the liver • Main concern is avoiding significant long-term side effects of immunosuppressive therapy Recommendations Grade of recommendation CNI-based immunosuppression is still the cornerstone of immunosuppressive regimens in LT. Tac results in better long-term graft and patient survival than Cs. A, including in HCV patients I To date there is no evidence that combination of MMF with CNI improves graft or patient survival compared with CNI and steroids or AZA I Induction agents are safe when used together with CNIs, allowing a reduction of CNI dose, especially in patients with pre-transplant renal impairment I Some concern still remains for the high costs of IL-2 R agents and their potential negative influence on tolerance III EASL CPG LT. J Hepatol 2016; 64: 433– 85
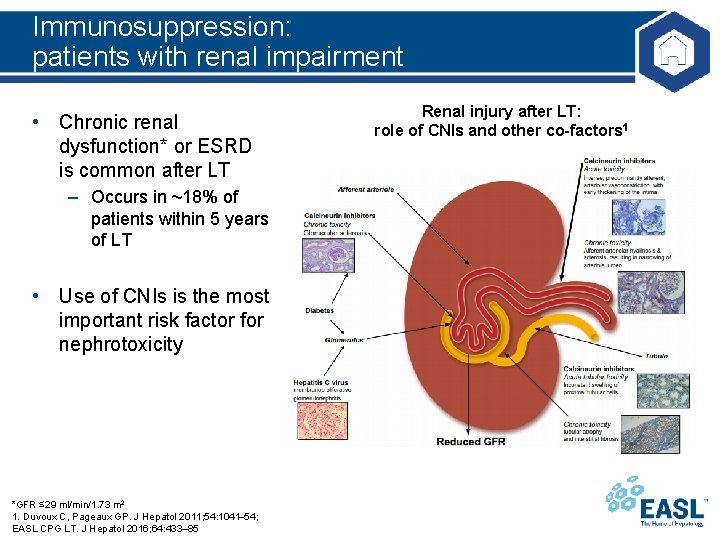
Immunosuppression: patients with renal impairment • Chronic renal dysfunction* or ESRD is common after LT – Occurs in ~18% of patients within 5 years of LT • Use of CNIs is the most important risk factor for nephrotoxicity *GFR ≤ 29 ml/min/1. 73 m 2 1. Duvoux C, Pageaux GP. J Hepatol 2011; 54: 1041 54; EASL CPG LT. J Hepatol 2016; 64: 433– 85 Renal injury after LT: role of CNIs and other co-factors 1

Immunosuppression: patients with renal impairment • Chronic renal dysfunction* or ESRD occurs in ~18% of patients within 5 years of LT • Use of CNIs is the most important risk factor for nephrotoxicity Recommendations Grade of recommendation IL-2 R antibodies with delayed and low-dose Tac plus MMF and steroids is safe and significantly improves renal function after LT I MMF monotherapy should not be used due to the risk of acute cellular rejection I MMF in combination with CNI reduction of at least 50% is associated with significant improvement in renal function and has a low risk of acute rejection I No RCTs have been performed comparing renal function with MMF and AZA III Conversion to SRL can be done safely and provide adequate immunosuppression without increased incidence of rejection, graft loss or infection I Early EVR-based, CNI-free immunosuppression seems to improve renal function after LT; however, this leads to an increased risk of acute rejection I RCTs with longer follow-up are needed. Safety concerns still remain III * GFR ≤ 29 ml/min/1. 73 m 2 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Immunosuppression: HCV liver-transplanted patients • A fine balance between suppressing immunity and maintaining optimal host viral response is required Recommendations Grade of recommendation There are no conclusive data showing a meaningful clinical difference between CNIs with respect to HCV recurrence after LT I A rapid decrease in steroid immunosuppression may determine a worse graft evolution in some patients I The ‘protective role’ of slow steroid withdrawal shown in several studies also requires further investigation III There is still controversy regarding the best antiproliferative agent for HCV recipients. Observational studies suggest that maintenance of AZA is associated with less fibrosis progression compared with MMF II-1 Only properly designed RCTs will confirm if m. TOR inhibitors are useful in HCV transplant recipients. There are very few HCV-specific data on EVR III OKT 3 and alemtuzumab are associated with severe HCV recurrence I Data for IL-2 R antagonists are contradictory I EASL CPG LT. J Hepatol 2016; 64: 433– 85

Immunosuppression: patients with HCC • Immunosuppression plays a central role in the increased risk of cancer after LT – Including the recurrence of HCC • A dose-dependent relationship between CNIs and HCC recurrence post LT has been reported – Studies were retrospective • Comparative data on CNIs are lacking and/or inconclusive Recommendations Grade of recommendation To date there is evidence that SRL does not improve long-term, recurrencefree survival beyond 5 years I Benefit of SRL is evident in 3– 5 years in patients with HCC within Milan criteria I EASL CPG LT. J Hepatol 2016; 64: 433– 85

Immunosuppression: patients with de novo tumours • There are few data on differential risk of immunosuppressive regimens Recommendations Grade of recommendation Risk of de novo malignancy should be considered similar in clinical practice with Tac or Cs. A-based immunosuppressive regimens II-2 The risk of malignancy related to CNIs in clinical practice may come from the dosage rather than the type of CNI used I There is no evidence suggesting a link between the use of MMF and de novo malignancy after liver transplantation III There are no published RCTs evaluating the effect of m. TOR inhibitors in preventing nor treating de novo malignancy after liver transplantation III EASL CPG LT. J Hepatol 2016; 64: 433– 85

Medical complications: management of HCV recurrence • HCV recurrence is universal and progression is accelerated after LT in patients with detectable HCV RNA – ~33% HCV-infected LT recipients experience aggressive HCV recurrence • At risk of clinical decompensation and graft loss • Post-LT graft loss within 1 year is predicted by: – Significant fibrosis (F ≥ 2 METAVIR) – Portal hypertension (HVPG ≥ 6 mm. Hg) – High TE values (>8. 6 k. Pa) Recommendations Grade of recommendation Follow-up of recurrent hepatitis C after LT should include a regular assessment of graft damage. Liver biopsy, HVPG measurement or TE are useful tools to assess graft damage and should be part of the follow-up protocol for these patients EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2

Medical complications: HCV treatment after LT • Early antiviral therapy may be appropriate Recommendations Grade of recommendation Antiviral therapy is recommended for all patients with HCV recurrence; initiate treatment early in those with significant graft damage (F ≥ 2). SVR is associated with improved outcomes in these patients II-1 *Treatment with Peg. IFN + RBV and addition of a first-generation PI for genotype 1 -infected patients are no longer recommended in LT recipients II-2 *Recommendation from EASL CPG HCV: IFN-free regimens are the only options in patients after liver transplantation because of their virological efficacy, ease of use, safety and tolerability. Protease inhibitor-containing regimens are contraindicated in patients with decompensated cirrhosis (evidence level A, grade of recommendation 1). EASL CPG LT. J Hepatol 2016; 64: 433– 85

Medical complications: HCV treatment after LT • Early antiviral therapy may be appropriate Recommendations Grade of recommendation *SOF/LDV + RBV and SOF + SPV (± RBV) are safe and achieve high SVR rates in genotype 1 - and 4 -infected LT recipients, including cirrhotic patients SOF alone or in combination with LDV has also shown to be safe and efficacious in severe forms of recurrence (i. e. fibrosing cholestatic hepatitis) II-1 In naïve patients with mild recurrence, the combination of ABT 450/r, OBV, DSV and RBV has shown high efficacy, but Cs. A and Tac adjustments are necessary due to drug–drug interactions †Other IFN-free regimens are being evaluated in clinical trials More data on PK and drug–drug interaction studies are required *New treatment options are now available that were approved after the publication of this CPG. Recommendation from EASL HCV CPG: Patients with post-transplant HCV recurrence, regardless of cirrhosis status, can be treated with SOF/LDV (GT 1, 4, 5 or 6) or SOF/VEL (all GT (evidence level A, grade of recommendation 1). For precise regimens and need for RBV or pretreatment ISD dose adjustments, please refer to the full guideline. Patients without cirrhosis or with compensated cirrhosis, with an e. GFR <30 ml/min/1. 73 m 2, can be treated with GLE/PIB for 12 weeks. ISD levels need to be monitored and adjusted as needed during and after treatment (evidence level B, grade of recommendation 1); †Other IFN-free regimens are now available. EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

Medical complications: prevention and treatment of HBV recurrence • Recurrence of infection is reduced with HBIG and NAs – Patients undergoing LT for HBV-related cirrhosis have excellent long-term outcomes • 5 -year survival rates ≥ 80% • With HBV recurrence, the aim is to control HBV replication to prevent graft loss Recommendations Grade of recommendation Combination of HBIG and NAs is an effective strategy to prevent HBV recurrence in most HBV-infected patients undergoing liver transplantation I Patients with undetectable HBV DNA at the time of liver transplantation and no history of resistance to NAs are the best candidates for low-dose HBIG or a short course of HBIG (1– 3 months) followed by NA monotherapy I Monotherapy with ETV or TDF* appears to be efficacious in controlling infection recurrence, but is probably not sufficient to prevent HBV graft infection II-2 Treatment of HBV recurrence should be initiated promptly with ETV or TDF* II-3 *Other treatments are available EASL CPG LT. J Hepatol 2016; 64: 433– 85
Medical complications: recipients of livers from anti-HBc-positive donors • Post-LT prophylaxis significantly reduces the probability of de novo HBV infection in patients receiving livers from anti-HBc-positive donors Recommendations Grade of recommendation Prophylaxis of HBV recurrence in patients who received a liver from an anti-HBc-positive donor should be initiated immediately after liver transplantation if recipients do not have anti-HBs II-2 Lamivudine monotherapy is the most cost-effective treatment. HBIG should not be used in HBs. Ag-negative patients who have received a liver from an anti-HBc-positive donor II-2 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Medical complications: patients transplanted for alcoholic liver disease • Post-LT outcomes in patients with ALD are good – Similar to individuals transplanted for other forms of liver disease • Moderately heavy drinking does not impact graft function or patient survival – Harmful drinking after LT is associated with a decreased survival Recommendations Grade of recommendation All patients with a prior diagnosis of alcoholic liver disease should be encouraged to remain abstinent from alcohol after liver transplantation II-2 In the case of relapse into regular alcohol consumption, patients should enter psychiatric treatment or counselling II-3 Specialist follow-up is relevant to assess alcohol abuse after liver transplantation, since harmful drinking, though not very frequent, is associated with decreased patient survival II-2 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Medical complications: recurrence of NAFLD • NAFLD and NASH are common after LT – Both de novo and recurrent • Several major risk factors for post-LT NAFLD/NASH – – BMI prior to and following LT Diabetes mellitus Arterial hypertension Hyperlipidaemia Recommendations Grade of recommendation Liver biopsy may be required to confirm recurrent or de novo NAFLD/NASH and to exclude other causes of elevated biochemical liver tests III No specific recommendation regarding prevention and treatment of NAFLD and NASH in liver transplantation recipients can be made, except to avoid excessive weight gain and to control diabetes, dyslipidaemia and arterial hypertension III EASL CPG LT. J Hepatol 2016; 64: 433– 85
Medical complications: recurrence of cholestatic liver disease • Recurrent AIH, PBC and PSC occurs in 10– 50% – Impact on graft function and patient survival is minimal Recommendations Grade of recommendation Recurrent autoimmune and cholestatic liver disease should be confirmed by liver biopsy and/or cholangiography (PSC) II-3 There is no evidence for prophylactic use of ursodeoxycholic acid in patients transplanted for PBC and PSC III EASL CPG LT. J Hepatol 2016; 64: 433– 85
Medical complications: management of HCC recurrence • HCC recurs in 8– 20% of recipients and is usually seen within 2 years of LT – Median survival is <1 year • De novo HCC may arise following progression to liver cirrhosis – Algorithms used for immunocompetent patients are appropriate: • Liver resection, RFA or transarterial chemoembolization* – Re-transplantation may be indicated in selected cases Recommendations Grade of recommendation There is evidence that SRL does not improve long-term recurrence-free survival beyond 5 years I Benefit of SRL is evident in 3– 5 years in patients with HCC within Milan criteria I Treatment of HCC recurrence after LT should be individualized. There are no data supporting the use of sorafenib in cases of disseminated recurrence III *When technically possible EASL CPG LT. J Hepatol 2016; 64: 433– 85

Medical management: additional morbidities • • • Renal dysfunction Prevention and treatment of infections Metabolic syndrome Bone disease De novo malignancies EASL CPG LT. J Hepatol 2016; 64: 433– 85

Long-term follow-up: lifestyle • LT should allow patients to enjoy the same state of health as they did prior to the disease – Goal should be to balance graft functioning, and patient psychological and physical well-being Recommendation Qo. L after LT should always be considered as an outcome measure • Also need to consider: – – Medication adherence Employment Sexual functioning and pregnancy Physical activity and weight control EASL CPG LT. J Hepatol 2016; 64: 433– 85 Grade of recommendation II-2

Appendix Additional recommendations

Genetic diseases • LT is indicated for: – Genetic diseases with parenchymal liver damage – Liver-based genetic disorders with prevalent extrahepatic manifestations Recommendations Grade of recommendation If the genetic defect affects other organs, the indication for LT is less evident and should be discussed in an expert centre III LT in patients with Wilson disease should be in cases of ALF or ESLD. LT can improve neurological symptoms but they can also worsen after the procedure. Neurological assessment before the transplant is mandatory III Hereditary haemochromatosis can be an indication for LT, especially if complicated by HCC. Cardiac evaluation before LT needs to be accurate* III Timing and approach to LT for primary hyperoxaluria type 1 are still controversial. In kidney transplant disease can recur; combined liver-kidney or LT prior to kidney failure is a possibility III LT† for a patient with familial amyloid polyneuropathy should be proposed as soon as symptoms appear. LT outcome is good in patients with no advanced disease manifestations. FAP liver recipients can develop polyneuropathy symptoms in a shorter time compared to FAP patients. Symptoms can be reversed with 2 nd LT III *Considering the cardiomyopathy associated with iron overload; †LT is often done with a domino technique EASL CPG LT. J Hepatol 2016; 64: 433– 85

LT in hepatic malignancies • Indications for LT in hepatic malignancies Recommendations Grade of recommendation LT for HCC patients meeting Milan criteria* has an excellent outcome. An expansion of these criteria is acceptable if the recurrence-free survival is comparable. All new models should be compared to the Milan model I LT is usually not recommended for cholangiocarcinoma or mixed hepatocellular/cholangiocarcinoma due to poor results. LT for perihilar cholangiocarcinoma could be offered in centres with clinical research protocols employing adjuvant or neoadjuvant therapy II-3 LT can be offered to patients with fibrolamellar carcinoma and epithelioid haemangioendothelioma II-3 Liver metastasis from non-liver tumours, such as neuroendocrine, might be considered for LT in very selected patients and only in trained liver transplant centres with experience in such indication for LT II-3 Liver metastasis from colorectal cancer is usually a contraindication for LT and might be proposed in very selected patients within research trials, and only in trained liver transplant centres with experience in such indication to LT II-3 *Solitary HCC with diameter <5 cm or up to 3 nodules with diameter <3 cm EASL CPG LT. J Hepatol 2016; 64: 433– 85
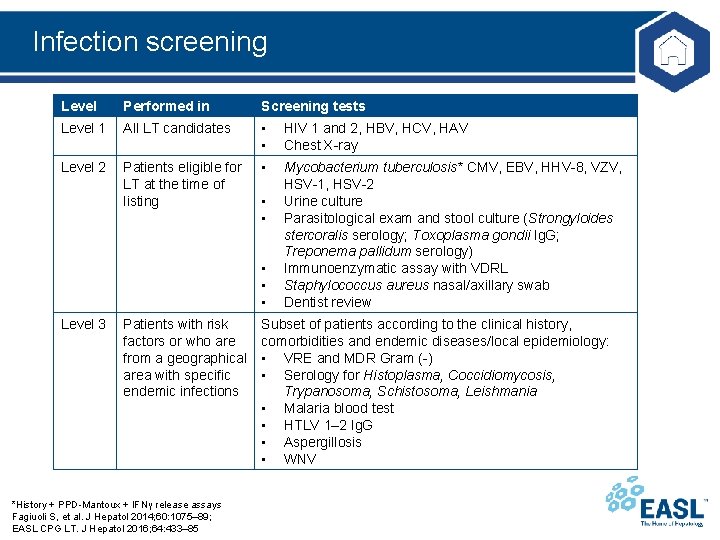
Infection screening Level Performed in Screening tests Level 1 All LT candidates • • HIV 1 and 2, HBV, HCV, HAV Chest X-ray Level 2 Patients eligible for LT at the time of listing • Mycobacterium tuberculosis* CMV, EBV, HHV-8, VZV, HSV-1, HSV-2 Urine culture Parasitological exam and stool culture (Strongyloides stercoralis serology; Toxoplasma gondii Ig. G; Treponema pallidum serology) Immunoenzymatic assay with VDRL Staphylococcus aureus nasal/axillary swab Dentist review • • • Level 3 Patients with risk factors or who are from a geographical area with specific endemic infections *History + PPD-Mantoux + IFNγ release assays Fagiuoli S, et al. J Hepatol 2014; 60: 1075– 89; EASL CPG LT. J Hepatol 2016; 64: 433– 85 Subset of patients according to the clinical history, comorbidities and endemic diseases/local epidemiology: • VRE and MDR Gram (-) • Serology for Histoplasma, Coccidiomycosis, Trypanosoma, Schistosoma, Leishmania • Malaria blood test • HTLV 1– 2 Ig. G • Aspergillosis • WNV

Cardiovascular function • Cardiac abnormalities have been noted in patients with cirrhosis – Increased cardiac output – Cirrhotic cardiomyopathy • Latent cardiac dysfunction, including reduced cardiac contractility with systolic and diastolic dysfunction, and electrophysiological abnormalities • Underlying heart disease should be ruled out in all patients Recommendations Grade of recommendation Electrocardiogram and transthoracic echocardiography should be performed in all liver transplant candidates II-3 In patients with multiple cardiovascular risk factors, and in patients older than 50 years, a cardiopulmonary exercise test should be done. If the target heart rate is not achieved during a standard exercise test a pharmacological stress test is the test of choice II-3 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Respiratory function • Lung function tests and a chest X-ray are recommended in all candidates for LT – HPS is found in 10– 17% of patients with cirrhosis • LT is the only curative treatment • PPHTN occurs in 2– 8% of patients with cirrhosis – Pre-LT management requires early diagnosis and therapy with pulmonary vasodilators Recommendations Grade of recommendation Respiratory function needs to be assessed; in particular the presence and stage of HPS and PPHTN should be evaluated II-3 HPS is an indication for liver transplantation II-2/3 Liver transplantation should be considered in patients with PPHTN responding to medical therapy with pulmonary vasodilators and with MPAP ≤ 35 mm. Hg II-2/3 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Renal function • Cirrhotic patients with renal failure have a 7 -fold increased risk of death – Hepatorenal syndrome must be differentiated from other causes of AKI • Combined liver–kidney transplantation may benefit patients with: – ESLD and with GFR <30 ml/min – Hepatorenal syndrome requiring renal replacement therapy for more than 8– 12 weeks – >30% fibrosis and glomerulosclerosis on renal biopsy Recommendations Grade of recommendation Hepatorenal syndrome is not a contraindication for liver transplantation II-2 Chronic kidney disease might be severe and irreversible, requiring combined liver–kidney transplant II-2 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Nutritional assessment • Cirrhosis is associated with malnutrition – Cachexia is present in nearly 70% of patients with ESLD • Malnutrition is associated with lower survival rate post-LT – Patients with BMI <18. 5 are at greatest risk • Nutrition intervention prior to LT may play an important role – Difficult to achieve Recommendations Grade of recommendation Nutritional status is hard to assess in a cirrhotic patient. The thickness and the area of psoas muscle have been correlated with worse outcome Improvement of nutritional status is indicated but no protocols have been approved so far EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2 III

Evaluation of bone abnormalities • Osteoporosis is common among patients with cirrhosis – Particularly in those with chronic cholestatic disease • Major risk factors are: – Female sex – Lower BMI – Tobacco use Recommendations Grade of recommendation As osteoporosis is associated with cirrhosis, densitometry should be part of liver transplant work-up EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

Immunological evaluation Recommendations Grade of recommendation The presence of donor-specific alloantibodies has been associated with acute and chronic antibody-mediated rejection and with several histological damages. The best test and use of anti-DSAs is still under study EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

Infection screening • Patients with cirrhosis are prone to infections – Can result in multiple organ failure and death • Screening of latent infections is required – To treat a potentially lethal infection before LT – To prevent an exacerbation after LT under immunosuppressive regimens • Correct evaluation of acute or chronic infections in the recipient is crucial Recommendations Grade of recommendation Screening for bacterial, fungal and viral infections is mandatory before LT. The presence of an active infection contraindicates the procedure CMV donor/recipient status determines time of prophylaxis EASL CPG LT. J Hepatol 2016; 64: 433– 85 III II-3

Anatomical evaluation • Assessment of the hepatic artery and the main portal system is essential • Evaluation of the biliary tree anatomy is particularly important for living donor transplantation • Comprehensive surgical and anaesthesia consultations are mandatory at the end of the evaluation process – To assess surgical and post-LT risks Recommendations Grade of recommendation Recipient anatomical evaluation is mandatory with a three-phase intravenous contrast CT scan II-3 The presence of portal vein thrombosis is not a contraindication for LT; nevertheless if the thrombosis extends to the whole porto-mesenteric system (Yerdel Stage IV), LT might not be feasible II-3 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Screening for neoplastic lesions in LT candidates • Screening for neoplastic lesions should always be performed • Evaluation should consider: – – Age Sex Alcohol consumption Smoking status • CRC screening is mandatory for candidates older than 50 years Recommendations Grade of recommendation Screening for neoplastic lesions should be part of LT work-up III Evaluation for pulmonary neoplasia, ear-nose-throat, stomatology, oesophageal and bladder is indicated in cases of alcohol or smoking addiction II-3 History of a treated cancer is not an absolute contraindication to LT. A 5 -year interval seems to be a reasonable time between curative cancer treatment and LT, depending on type and stage of previously treated cancer III EASL CPG LT. J Hepatol 2016; 64: 433– 85

Social assessment, psychiatric illness and addiction • Evaluation of a patient’s social network, psychological status and addiction – To assess likelihood of adherence in the recipient • Neuropsychological testing, CT brain scan or NMR and electroencephalography may help to determine reversibility of neuropsychiatric conditions in patients with hepatic encephalopathy • Active drug or alcohol abuse is a contraindication to LT Recommendations Grade of recommendation Social, psychological and, when indicated, psychiatric evaluation should be performed to evaluate adherence of the recipient, and potential risk factors for non-adherence after LT III Stably abstinent, methadone-maintained, opiate-dependent patients should not be excluded from evaluation for LT II-2 Smoking cessation should be mandatory in all transplant candidates EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

ECD: donors with previous malignancy or infection Recommendations Grade of recommendation Livers from a donor with previous history of malignancy can be used in selected situations according to tumour site and its stage II-3 Donors with select bacterial infections can be safely used as long as appropriate therapy is provided to both the donor before procurement and the recipient after transplantation. Livers from donors with isolated fungal infections should be routinely used. Grafts from donors with viral or parasitic disease should be used according to the type of infection and to the severity of recipient liver disease II-3 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Different types of LT: summary Recommendations Grade of recommendation Preservation of the inferior vena cava by piggyback technique is recommended during LT whenever it is possible. The use of this technique is associated with greater haemodynamic stability during surgery II-3 Domino transplant can be used for patients with familial amyloid polyneuropathy, as long as recipients are older than 55 years, in order to reduce the risk of developing the disease II-3 Auxiliary transplantation may be indicated in patients with ALF or functional, congenital or metabolic disorders affecting a normal liver. The advantage of this type of transplantation is the possibility of removing the graft and withdrawing immunosuppression once the native liver returns to its normal function II-3 Because of the low number of available organs in paediatric LT, the use of split LT is an acceptable option, as long as the liver graft volume is sufficient. In this case the child receives a graft that includes segments II and III II-2 In adult LT, the use of the split LT may be an alternative given the organ shortage, but the left liver graft recipients must have a low weight. The use of the left lobe of the graft is associated with worse outcomes II-2 Given the organ shortage, adult LDLT is recommended if there is an available donor. Estimated volume of the graft must be 0. 8% of the recipient’s weight III EASL CPG LT. J Hepatol 2016; 64: 433– 85

Different types of LT: summary Recommendations Grade of recommendation It is necessary to prevent hepatic artery thrombosis during LT and the postoperative period. This complication requires re-transplantation in 50% of cases III PVT prior to LT does not generally represent an absolute contraindication. In cases of extensive thrombosis a non-anatomical portal revascularization technique such as a renoportal anastomosis can be performed II-3 If a biliary anastomosis leakage in the post-transplantation period is diagnosed, initial ERCP with sphincterotomy is recommended. If the leakage persists, a temporary biliary stent can be used II-3 In patients with impaired coagulation, temporary packing of 48 hours may be necessary III In advanced cases of ischaemic cholangiopathy, the final treatment is re-transplantation II-3 In cases of stenosis of the biliary anastomosis without improvement after conservative treatment, it is recommended to perform a hepatico-jejunostomy II-3 In partial graft recipients with bile duct anastomosis stenosis or leakage, interventional radiology plays an important role (dilatation, stent insertion), but 50% of patients eventually require a hepatico-jejunostomy III EASL CPG LT. J Hepatol 2016; 64: 433– 85

Re-transplantation • Selection of recipients should consider – Disease severity – Interval since the primary LT – Graft quality • This is more important than the cause of re-transplantation Recommendations Grade of recommendation Re-transplantation has inferior outcome compared with the first transplant, nevertheless it should be considered in cases of acute or chronic graft failure A patient candidate for re-transplantation should undergo a liver work-up as for the first transplant HCV recurrence is not a contraindication for re-transplantation EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2 III II-3

Total withdrawal of immunosuppression • Ultimate aim is for graft acceptance without the need for any long-term pharmacological assistance – Long-term survivors following LT are often systematically and excessively immunosuppressed • Drug weaning is a strategy that should be considered – Must be undertaken gradually under careful physician surveillance Recommendations Grade of recommendation Intended immunosuppression withdrawal is still experimental and can only be considered in the setting of rigorous clinical trials under strict conditions and with intensive follow-up EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

Management of renal dysfunction • Majority of patients who survive the first 6 months after LT present with impaired kidney function – 30– 80% of patients develop CKD stage 3– 4 – Cumulative risk of ESRD requiring maintenance dialysis or renal transplantation is 5– 9% within the first 10 years post-LT • CNIs are considered responsible for >70% of cases of ESRD after LT • AKI and CKD are associated with a significantly increased risk of mortality in early and late post-LT course Recommendations Grade of recommendation Continuous monitoring of renal function in LT recipients is mandatory and should be started immediately after LT Reduction or withdrawal of CNI-associated immunosuppression or alternative CNI-free protocols should be considered as soon as possible in patients with impaired renal function Kidney transplantation is the optimal treatment for LT patients with ESRD EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2 I II-3

Prevention and treatment of infections • Infectious complications are a major cause of morbidity and mortality following transplantation Recommendations Grade of recommendation Use CMV prophylaxis for 3 months in patients at a higher risk of CMV infection PTLD should always be suspected post-LT, especially those at high risk, with fever, weight loss, night sweats, even in the absence of lymphoadenopathy II-2 III Oral prophylaxis against Candida species is recommended during the first months, as it reduces mortality due to fungal infection II-3 Prophylaxis against Aspergillus is only recommended in high-risk situations II-3 Prophylaxis against P. jirovecii with trimethoprim-sulphamethoxazole should be given to all liver transplanted patients for 6– 12 months II-2 Treatment of P. jirovecii infection consists of trimethoprim-sulphamethoxazole. Corticosteroids are useful as adjunctive therapy to reduce both pulmonary inflammation and post-infection fibrosis II-3 Patients undergoing treatment for tuberculosis should be monitored for potential hepatotoxicity and for acute rejection II-3 EASL CPG LT. J Hepatol 2016; 64: 433– 85
Prevention and treatment of metabolic syndrome • Prevalence of Met. S is 50– 60% in the LT population – – • Diabetes mellitus 10– 64% Obesity 24– 64% Dyslipidaemia 40– 66% Arterial hypertension 40– 85% Clinical features of Met. S alone or in combination contribute to late post-operative morbidity and mortality – LT recipients are at increased risk of cardiovascular events – CVD accounts for ~25% of deaths in the long-term follow-up after transplant Recommendations Grade of recommendation LT recipients are at increased risk of CVD. Effective and prompt treatment of modifiable risk factors is imperative Pharmacological therapies must be initiated as soon as possible to control arterial hypertension, hyperlipidaemia, diabetes and obesity Adopting a healthy lifestyle is an additional effective management option EASL CPG LT. J Hepatol 2016; 64: 433– 85 III II-3 III

Prevention and treatment of bone disease • Bone loss accelerates in the first 6 months after LT • Factors associated with increased fracture risk – – – Post-LT immunosuppression regimen Female sex Older age Lower BMI Renal dysfunction Recommendations Grade of recommendation Screen BMD yearly in patients with pre-existing osteoporosis and osteopenia, and every 2– 3 years in patients with normal BMD. Thereafter, screening depends on the progression of changes in BMD and on risk factors II-3 LT patients with osteopenia should perform regular weight-bearing exercise, and receive calcium and vitamin D supplementation II-3 Bisphonate therapy should be considered in patients with osteoporosis or recurrent fractures II-2 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Prevention and treatment of de novo malignancies • Leading cause of mortality after CVD in first year post-LT • Reported incidence of de novo cancers ranges from 3– 26%, dependent on duration of follow-up – Continuous increase in risk post-LT • Up to 19% at 10 years • Up to 34% 15 years Recommendations Grade of recommendation Cancer screening is warranted after LT, especially in populations at increased risk, to detect de novo tumours at an early and potentially curative stage II-2 Patients transplanted for alcoholic liver disease should undergo a more intensive surveillance protocol for the detection of upper gastrointestinal, oropharyngeal-laryngeal, as well as lung cancers II-3 Patients transplanted for PSC with associated inflammatory bowel disease should undergo annual colonoscopy II-3 EASL CPG LT. J Hepatol 2016; 64: 433– 85

Follow-up: adherence • Poor adherence is an issue almost 50% of LT patients – Coincides with substantial increases in rates of graft loss and death • Particularly affects young LT recipients Recommendations Grade of recommendation Physical and psychosocial functions after LT should be properly assessed in adolescent LT recipients II-2 Adherence to medical prescriptions, particularly immunosuppressive therapy, should always be evaluated after LT. Special attention should be given to immunosuppression-related side effects as they represent the major reason for non-adherence among adolescent recipients II-2 Specific, structured support should be planned in transplanted children and adolescents concerning schooling II-2 Multidisciplinary measures developed by professional educators, supported by psychologists, and coordinated by physicians are warranted to improve adherence before and after LT EASL CPG LT. J Hepatol 2016; 64: 433– 85 III

Employment • Proportion of LT recipients who return to work ranges from 26% to 57% – Dependent on the follow-up period • Employed patients have a significantly better Qo. L Recommendations Grade of recommendation Even though no clear correlation has been found between aetiology of liver disease and returning to work after liver transplantation, special attention should be devoted to patients transplanted for alcoholic liver disease, as they seem to be at higher risk of unemployment EASL CPG LT. J Hepatol 2016; 64: 433– 85 II-2

Sexual functioning and pregnancy • Successful LT improves sex hormone disturbances – Immunosuppressive drugs may interfere with hormone metabolism Recommendations Grade of recommendation Liver transplantation patients of reproductive age should always be counselled about the possibility of pregnancy and the use of contraception III Pregnancy should be avoided for the first 12 months after liver transplantation, although some centres advocate waiting 24 months II-3 Immunosuppression should be maintained during pregnancy. Steroids, CNIs and azathioprine have not been reported to be teratogenic II-3 Mycophenolate mofetil and azathioprine are usually not recommended II-3 m. TOR inhibitors may affect spermatogenesis in male recipients II-2 More studies should be designed to investigate the role of immunosuppression on sexual dysfunction, in both male and female recipients EASL CPG LT. J Hepatol 2016; 64: 433– 85 III
- Slides: 87