Clinical pharmacology of corticosteroids in horses and doping















































































- Slides: 120

Clinical pharmacology of corticosteroids in horses and doping control P. L. Toutain, National Veterinary School, Toulouse, France ICRAV 2012, September 15 -22, 2012 Philadelphia, USA 1

The European Horse Scientific Liaison Committee The view expressed in this presentation are those of the author and do not commit the official policy of the EHLSC 2

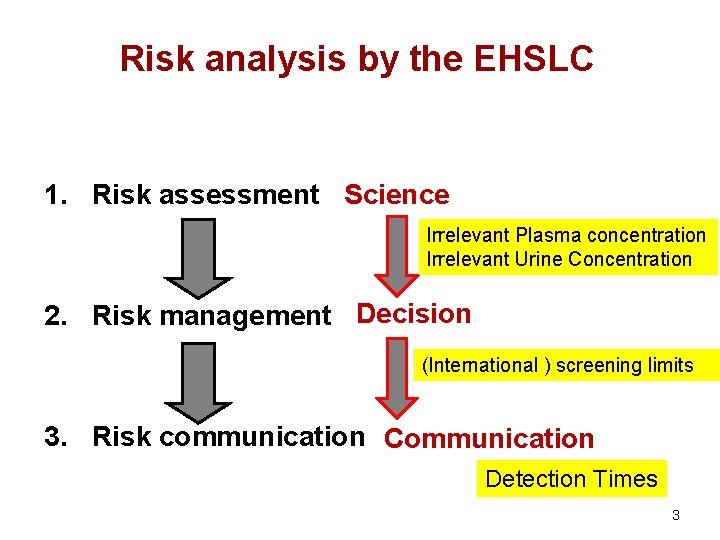
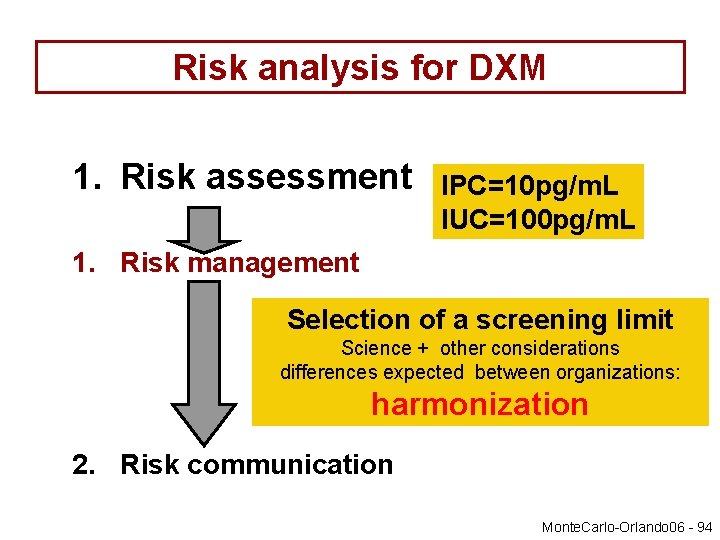
Risk analysis by the EHSLC 1. Risk assessment Science Irrelevant Plasma concentration Irrelevant Urine Concentration 2. Risk management Decision (International ) screening limits 3. Risk communication Communication Detection Times 3

Goals of the presentation I. An overview on PK/PD of corticosteroids – Cortisol as a biomarker of GGs administration – Synthetic GC • Potency, esters, routes of administration etc. II. Principles to establish irrelevant plasma and irrelevant urine concentrations – For systemic action – For local action • Intra-articular administration • inhalation 4

Glucocorticosteroids (GCs) v Glucocorticosteroids (GCs) are broad and potent anti -inflammatory drugs. § They are extensively used in horses to mitigate or suppress inflammation associated with a variety of conditions especially joint and respiratory system inflammation. v GCs are not curative: § GCs are only palliative symptomatic treatments and chronic use of GCs can be, in fine , detrimental to the horse raising a welfare issue. v GCs possess many other pharmacological properties of potential abuse in competing horses 5

Cortisol in horses: an endogenous hormone and a surrogate endpoint of the duration of the GC effects 6

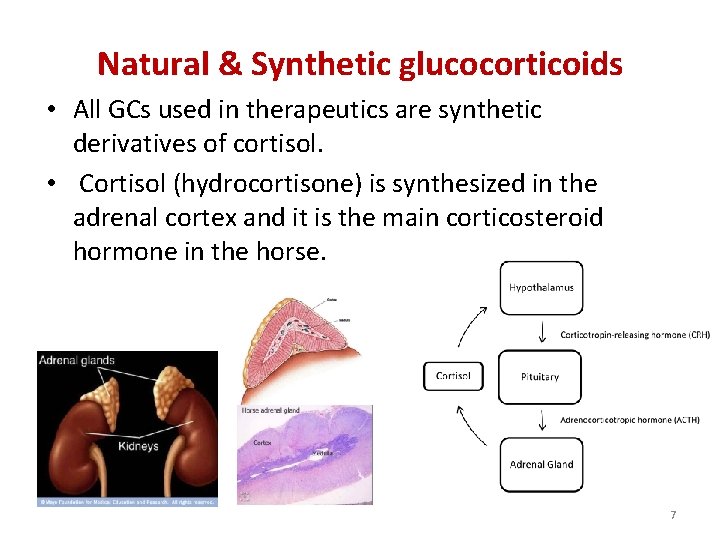
Natural & Synthetic glucocorticoids • All GCs used in therapeutics are synthetic derivatives of cortisol. • Cortisol (hydrocortisone) is synthesized in the adrenal cortex and it is the main corticosteroid hormone in the horse. 7

Cortisol in horses: an endogenous hormone and a surrogate endpoint of the duration of the GC effects 8

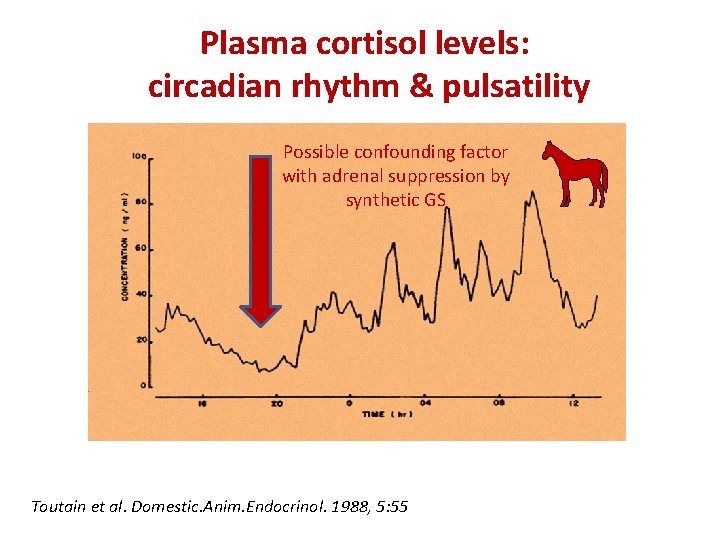
Plasma cortisol levels: circadian rhythm & pulsatility Possible confounding factor with adrenal suppression by synthetic GS Toutain et al. Domestic. Anim. Endocrinol. 1988, 5: 55

Plasma cortisol levels: circadian rhythm & pulsatility • the plasma cortisol profile follows a typical circadian rhythm with an overall 24 h mean plasma concentration of 43. 4± 6. 2 ng/m. L: the minimal concentrations (27. 9± 6. 9 ng/ml) were observed early in the night at 21. 2± 0. 59 h and maximal plasma concentrations (58. 9± 9. 5 ng/m. L at 9. 2± 0. 6 h in the morning) 10

Plasma cortisol levels: circadian rhythm & pulsatility • The cortisol secretion is pulsatile with minuteto-minute variations in the plasma cortisol concentrations making the interpretation of snapshot plasma samples difficult. 11

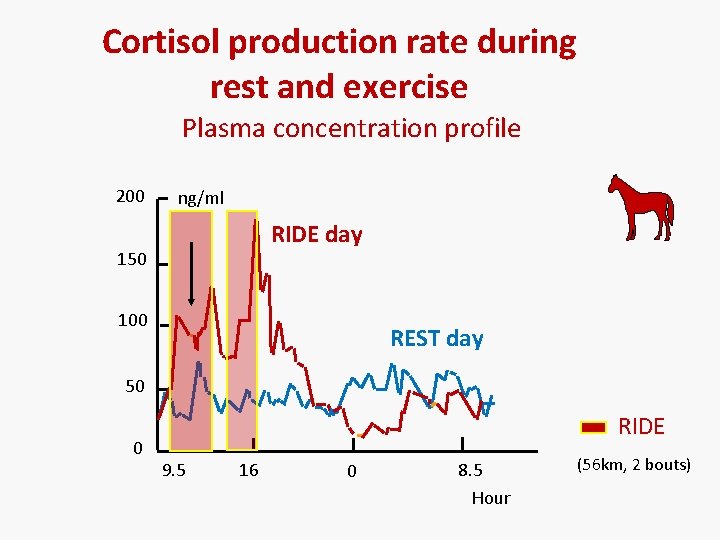
Cortisol production rate during rest and exercise Plasma concentration profile 200 ng/ml RIDE day 150 100 REST day 50 0 RIDE 9. 5 16 0 8. 5 Hour (56 km, 2 bouts)

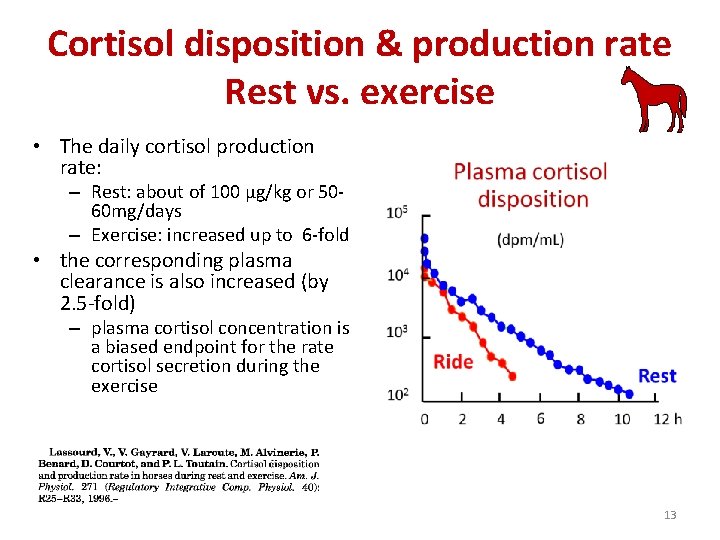
Cortisol disposition & production rate Rest vs. exercise • The daily cortisol production rate: – Rest: about of 100 µg/kg or 5060 mg/days – Exercise: increased up to 6 -fold • the corresponding plasma clearance is also increased (by 2. 5 -fold) – plasma cortisol concentration is a biased endpoint for the rate cortisol secretion during the exercise 13

DXM disposition: rest vs. exercise • The 3 -hour test exercise included walking, trotting and canter sequences – the average speed was calculated to be approximately 12 km per hour. – Mean heart rate (about 120/min) for all horses to standardise the test exercise and for comparability of horses. 14

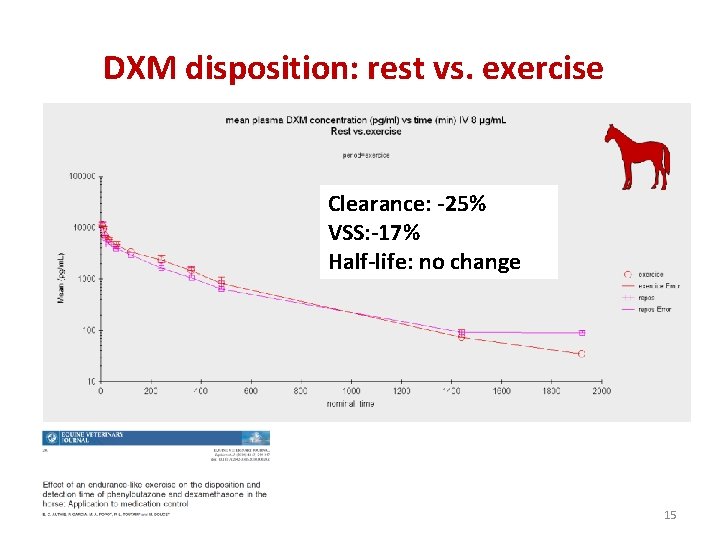
DXM disposition: rest vs. exercise Clearance: -25% VSS: -17% Half-life: no change 15

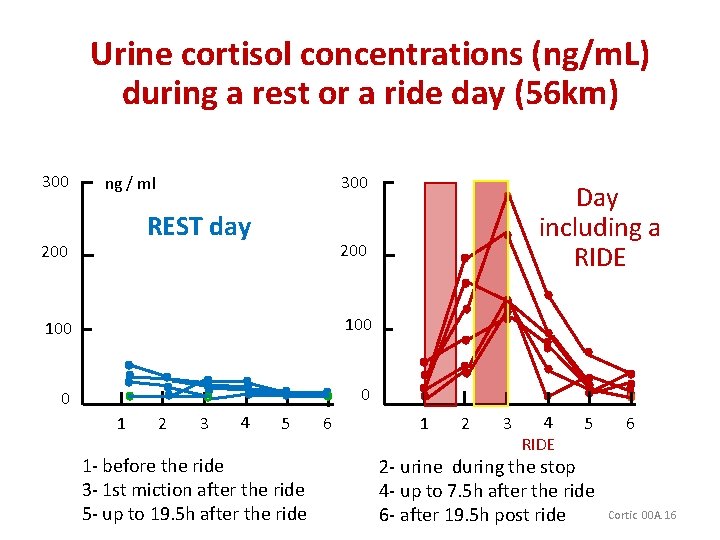
Urine cortisol concentrations (ng/m. L) during a rest or a ride day (56 km) 300 ng / ml REST day 200 100 0 0 1 2 3 4 Day including a RIDE 5 1 - before the ride 3 - 1 st miction after the ride 5 - up to 19. 5 h after the ride 6 1 2 3 4 RIDE 5 2 - urine during the stop 4 - up to 7. 5 h after the ride 6 - after 19. 5 h post ride 6 Cortic 00 A. 16

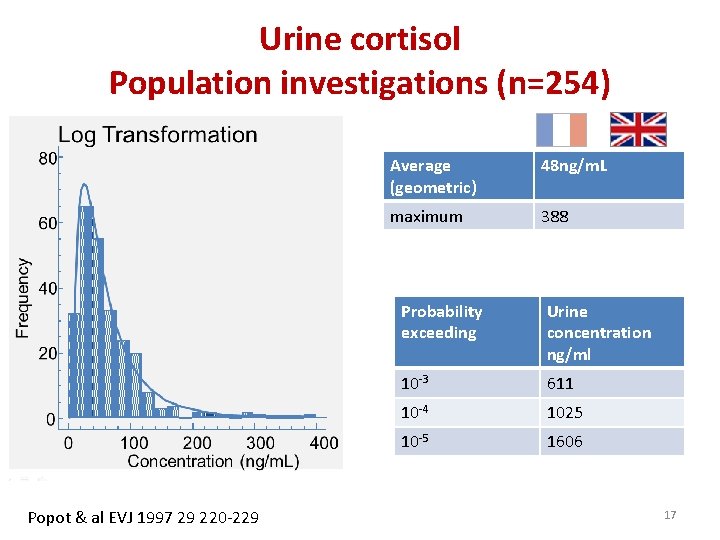
Urine cortisol Population investigations (n=254) Popot & al EVJ 1997 29 220 -229 Average (geometric) 48 ng/m. L maximum 388 Probability exceeding Urine concentration ng/ml 10 -3 611 10 -4 1025 10 -5 1606 17

Urine cortisol • International threshold: 1µg/ml or 1000 ng/m. L 18

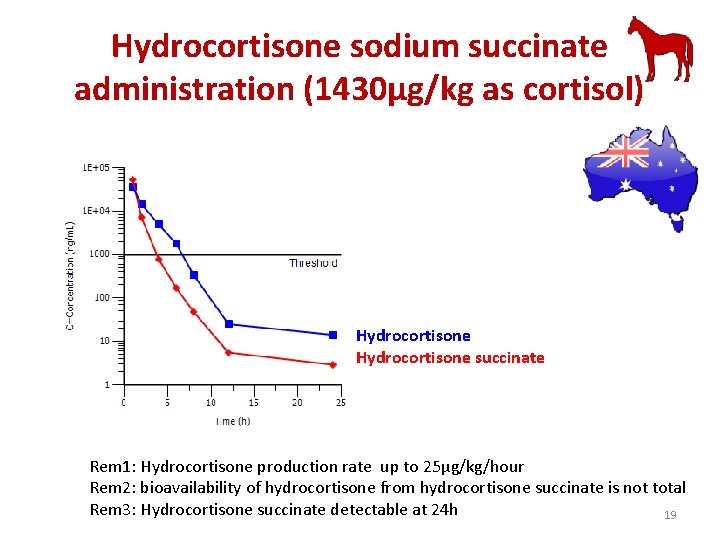
Hydrocortisone sodium succinate administration (1430µg/kg as cortisol) Hydrocortisone succinate Rem 1: Hydrocortisone production rate up to 25µg/kg/hour Rem 2: bioavailability of hydrocortisone from hydrocortisone succinate is not total Rem 3: Hydrocortisone succinate detectable at 24 h 19

Plasma cortisol levels and synthetic corticoid treatment 20

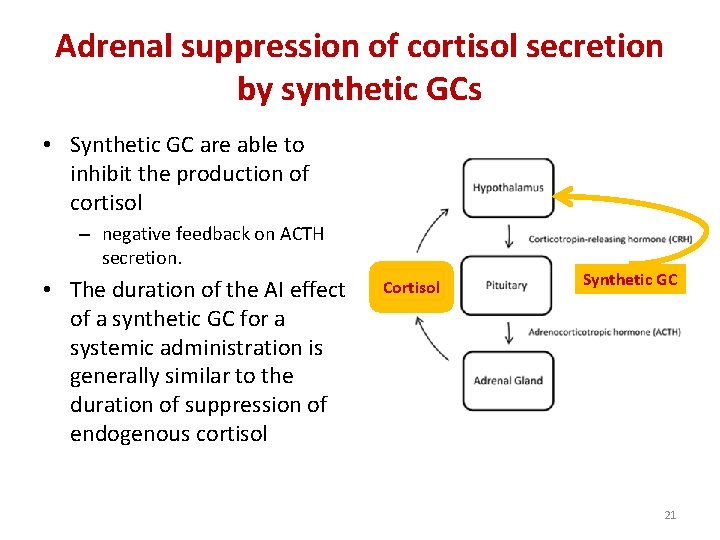
Adrenal suppression of cortisol secretion by synthetic GCs • Synthetic GC are able to inhibit the production of cortisol – negative feedback on ACTH secretion. • The duration of the AI effect of a synthetic GC for a systemic administration is generally similar to the duration of suppression of endogenous cortisol Cortisol Synthetic GC 21

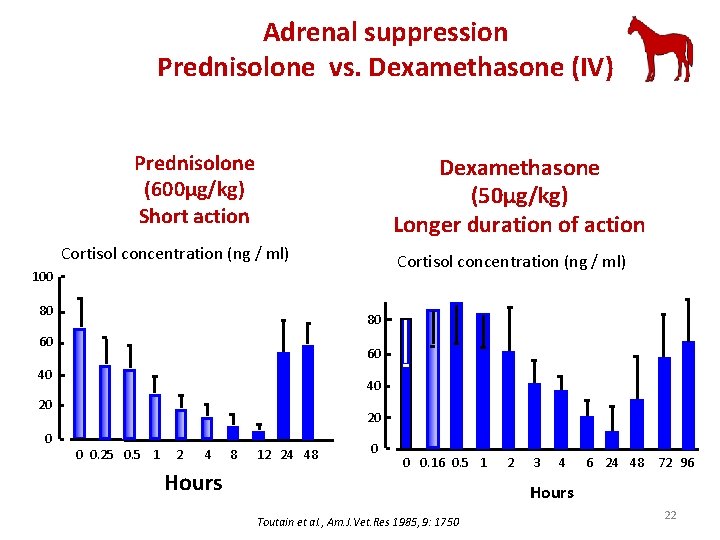
Adrenal suppression Prednisolone vs. Dexamethasone (IV) Prednisolone (600µg/kg) Short action Dexamethasone (50µg/kg) Longer duration of action Cortisol concentration (ng / ml) 100 80 80 60 60 40 40 20 0 0. 25 0. 5 1 2 4 Hours 8 12 24 48 0 0 0. 16 0. 5 1 2 3 4 6 24 48 72 96 Hours Toutain et al. , Am. J. Vet. Res 1985, 9: 1750 22

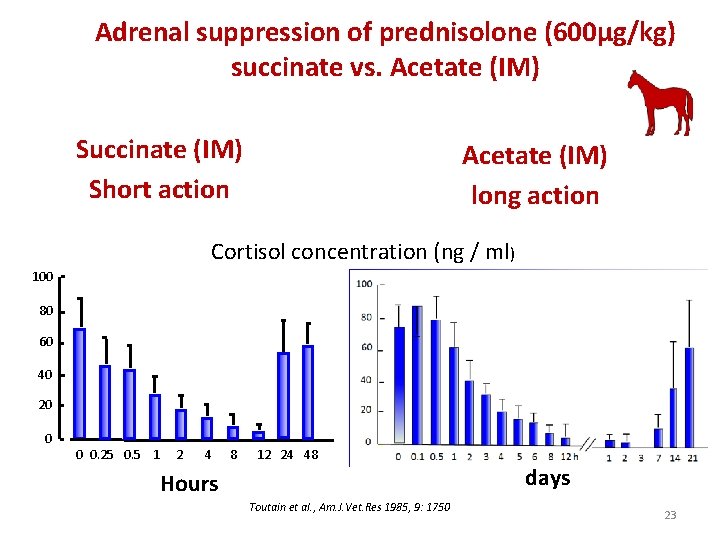
Adrenal suppression of prednisolone (600µg/kg) succinate vs. Acetate (IM) Succinate (IM) Short action Acetate (IM) long action Cortisol concentration (ng / ml) 100 80 60 40 20 0 0 0. 25 0. 5 1 2 4 8 12 24 48 days Hours Toutain et al. , Am. J. Vet. Res 1985, 9: 1750 23

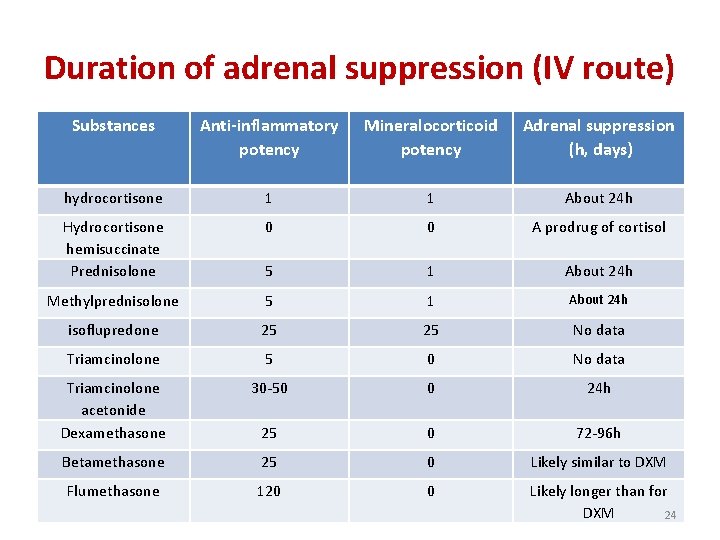
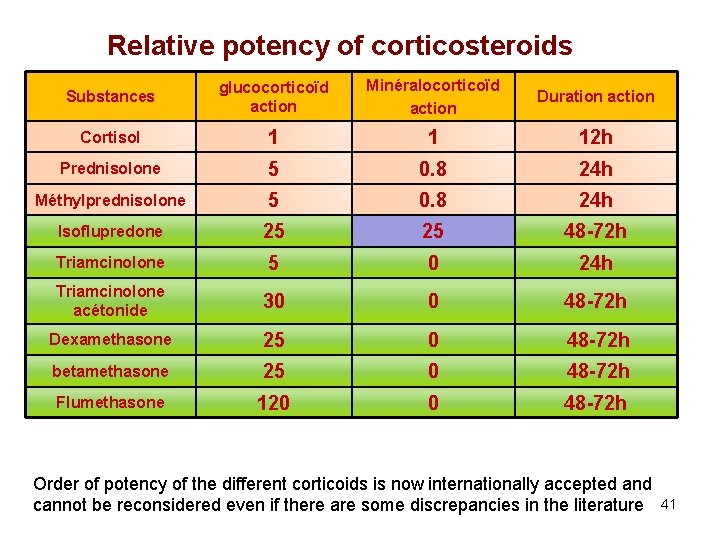
Duration of adrenal suppression (IV route) Substances Anti-inflammatory potency Mineralocorticoid potency Adrenal suppression (h, days) hydrocortisone 1 1 About 24 h Hydrocortisone hemisuccinate Prednisolone 0 0 A prodrug of cortisol 5 1 About 24 h Methylprednisolone 5 1 About 24 h isoflupredone 25 25 No data Triamcinolone 5 0 No data Triamcinolone acetonide Dexamethasone 30 -50 0 24 h 25 0 72 -96 h Betamethasone 25 0 Likely similar to DXM Flumethasone 120 0 Likely longer than for DXM 24

Adrenal suppression of cortisol secretion by synthetic GCs: local administration • For local administrations, the fraction that gains access to the blood can be too low to impact on the adrenal gland function. 25

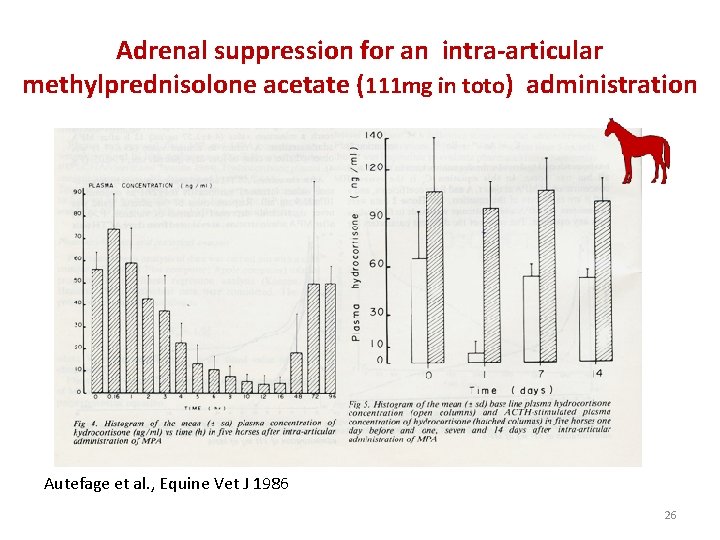
Adrenal suppression for an intra-articular methylprednisolone acetate (111 mg in toto) administration Autefage et al. , Equine Vet J 1986 26

Adrenal suppression for an intra-articular methylprednisolone administration • For the IA route a negative effect on the adrenal gland persisting for about 4 days after an administration of 111 mg in toto of methylprednisolone acetate (100 mg of methylprednisolone in toto) was reported (Autefage et al. , 1986). • In this trial the responsiveness of the adrenal gland as judged by a series of ACTH tests was not impaired showing that adrenal suppression cannot be sensitive enough to be an appropriate biomarker of the duration of GC articular effect 27

Adrenal suppression for an intra-articular triamcinolone acetonide administration • The same approach was used for triamcinolone acetonide (TA) after an IV dose of 40µg/kg (Soma et al. , 2011) with a recovery of adrenal suppression beginning 19 h following administration (corresponding plasma concentration of about 1 ng/ml) and nearly complete at about 96 h (extrapolated corresponding plasma concentration of about 0. 2 pg/m. L). • Using the generic PK/PD model described by Toutain and Lassourd (Toutain & Lassourd, 2002), and considering therapeutic TA dose (0. 011 -0. 022 mg/kg) (Plumb, 2011), and the TA plasma clearance (7. 46 m. L/kg/min), an EPC of 1 ng/m. L and an IPC of 2 -4 pg/m. L can be computed that is consistent with the experimental data of Soma et al. 2011. 28

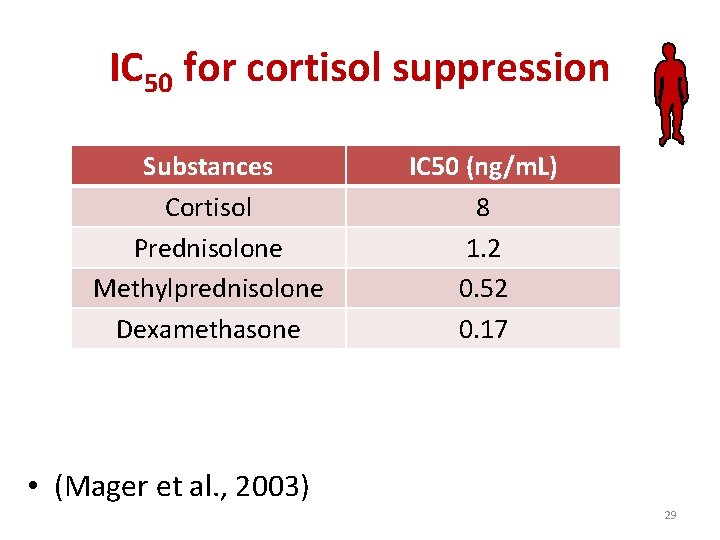
IC 50 for cortisol suppression Substances Cortisol Prednisolone Methylprednisolone Dexamethasone IC 50 (ng/m. L) 8 1. 2 0. 52 0. 17 • (Mager et al. , 2003) 29

Synthetic derivatives of cortisol 30

Synthetic derivatives of cortisol • A variety of GCs have been developed to increase the potency of the antiinflammatory effect and to increase selectivity by decreasing or even suppress the mineralocorticoid effects of cortisol 31

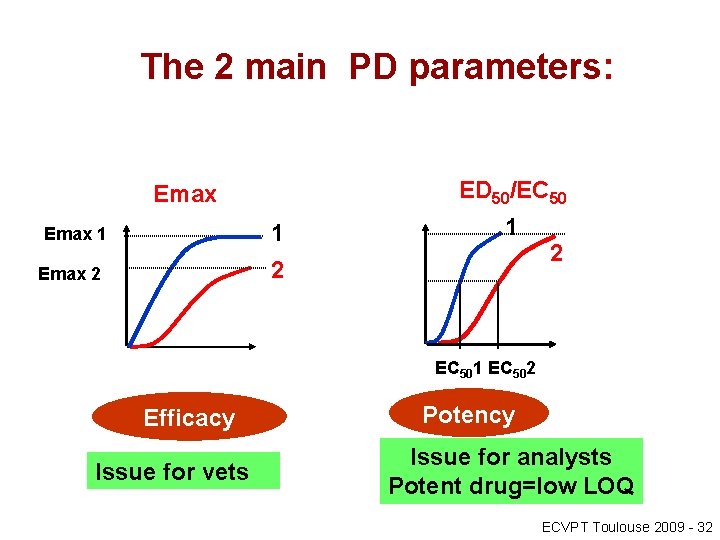
The 2 main PD parameters: ED 50/EC 50 Emax 1 1 2 Emax 2 2 EC 501 EC 502 Efficacy Issue for vets Potency Issue for analysts Potent drug=low LOQ ECVPT Toulouse 2009 - 32

Efficay vs. Potency • Potency and efficacy are the two most relevant PD properties of any drugs including GC and these two terms should not be confused: a more potent drug is not necessarily a more efficacious drug. • Efficacy is a PD parameter corresponding to a maximal possible effect that can be obtained in a given test system: it is qualified by a parameter termed Emax (or Imax for an inhibitory effect). • It is the most important parameter for clinicians when the measured dependent variable has a clinical meaning (lameness score, tidal volume for respiratory function, lack of mineralocorticoid activity for a GC, etc. ). 33

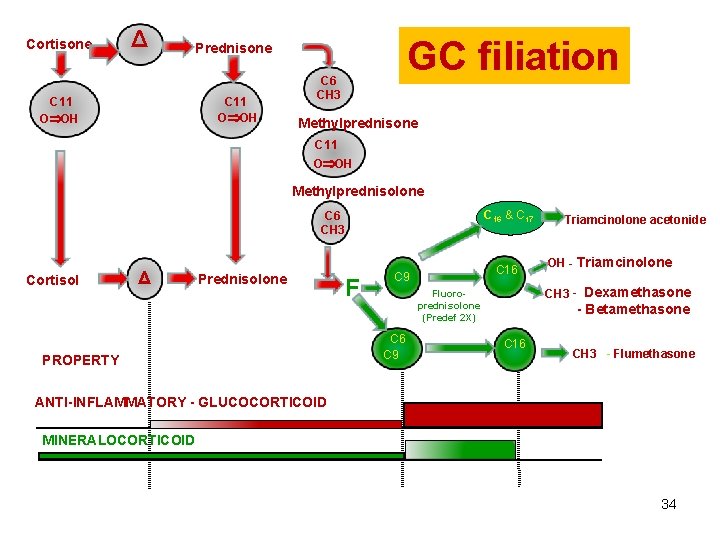
Cortisone Δ GC filiation Prednisone C 11 O OH C 6 CH 3 Methylprednisone C 11 O OH Methylprednisolone C 16 & C 17 C 6 CH 3 Cortisol Δ Prednisolone PROPERTY F C 16 C 9 OH - Triamcinolone CH 3 - Dexamethasone Fluoroprednisolone (Predef 2 X) C 6 C 9 Triamcinolone acetonide - Betamethasone C 16 CH 3 - Flumethasone ANTI-INFLAMMATORY - GLUCOCORTICOID MINERALOCORTICOID 34

Structure activity relationship • The introduction of an additional double bound between C-1 and C-2 of cortisol in all synthetic corticosteroids selectively increases glucocorticoid anti-inflammatory activity that are always parallel. • This single modification, as in prednisolone, does not alter mineralocorticoid activity, resulting in an enhanced glucocorticoid/mineralocorticoid potency ratio (see Table 1). • Substances having a ketone radical at C-11 in lieu of a -OH radical (cortisone, prednisone and methylprednisone) are prodrugs (or metabolites) of cortisol, prednisolone and methylprednisolone respectively 35

Structure activity relationship • • All halogenated GC are derivatives of prednisolone i. e of the 1 -2 dehydrocortisol. The addition of a halogen at the C-9 position (a fluorine for all GC but a chloride for beclomethasone 17 -monopropionate) enhances both glucocorticoid and mineralocorticoid activity as in 9α-fluoroprednisolone or isoflupredone. Isoflupredone lacks selectivity regarding mineralocorticoid effects when isoflupredone is substituted at the C-16 by an -OH radical (triamcinolone) or a -CH 3 group (dexamethasone, betamethasone), the new C-16 substituted compounds are practically devoid of any mineralocorticoid activity while retaining their anti-inflammatory potency. Tiamcinolone acetonide is a more lipophilic substance without mineralocorticoid effect obtained by the introduction of an acetonide between C-16 and C-17. Flumethasone is a derative of dexamethasone with a supplementary fluorine in position 6 and fluticasone can be viewed as a derivative of dexamethasone with two supplementary fluorine in position 6 and 36

Prednisone • Prednisone is used in horses because it is inexpensive and tablet formulations are convenient to administer. • Prednisone is poorly active in horses to treat lung conditions as recurrent airway obstruction (RAO) by the oral route (Robinson et al. , 2002). • This is likely to be due to either poor oral absorption of prednisone or a failure of the liver to convert prednisone to the active prednisolone as no prednisolone is detected in the plasma after an oral administration of prednisone at 2. 2 mg/kg (Peroni et al. , 2002). 37

Isoflupredone (Predef 2 X®) • The addition of a fluorine at the C-9 position considerably enhances both glucocorticoid and mineralocorticoid activity as in 9αfluoroprednisolone (also named isoflupredone). • Isoflupredone is marketed as an anti-inflammatory drug (Predef 2 X®) but it lacks selectivity regarding mineralocorticoid effects and it was shown in horses that isoflupredone acetate (30µg/kg/day for 14 days by the intramuscular route) was associated with hypokaliema in the treatment of RAO (Picandet et al. , 2003). 38

Triamcinolone acetonide • Tiamcinolone acetonide (TA) is a more lipophilic substance obtained by the introduction of an acetonide between C-16 and C-17. • TA is not a prodrug of triamcinolone and can be used for local administration as for IA administration in horses to treat osteoarthritis or as inhalation formulation. 39

Triamcinolone acetonide is 5 -20 folds more potent than triamcinolone 40

Relative potency of corticosteroids Substances glucocorticoïd action Minéralocorticoïd action Duration action Cortisol 1 1 12 h Prednisolone 5 0. 8 24 h Méthylprednisolone 5 0. 8 24 h Isoflupredone 25 25 48 -72 h Triamcinolone 5 0 24 h Triamcinolone acétonide 30 0 48 -72 h Dexamethasone 25 0 48 -72 h betamethasone 25 0 48 -72 h Flumethasone 120 0 48 -72 h Order of potency of the different corticoids is now internationally accepted and cannot be reconsidered even if there are some discrepancies in the literature 41

Most GCs are administered as esters substance Hydrosoluble (IV) Non hydrosoluble (depot) Phosphate Acetate Succinate Butyrate ……… 42

Most GCs are administered as esters • Most GC are administered as esters, • Esterification considerably alters the disposition and duration of the GC action. – Esterification with a monoacid (like acetic acid) at C 21 gives non-hydrosoluble drugs that can be used as long-acting formulations when administered by the intramuscular, subcutaneous or IA routes. – Esterification of the same parental drug by a diacid (such as succinic acid) can give a hydrosoluble ester enabling a salt to be formed (e. g. a sodium succinate). Phosphate esters are also hydrosoluble. 43

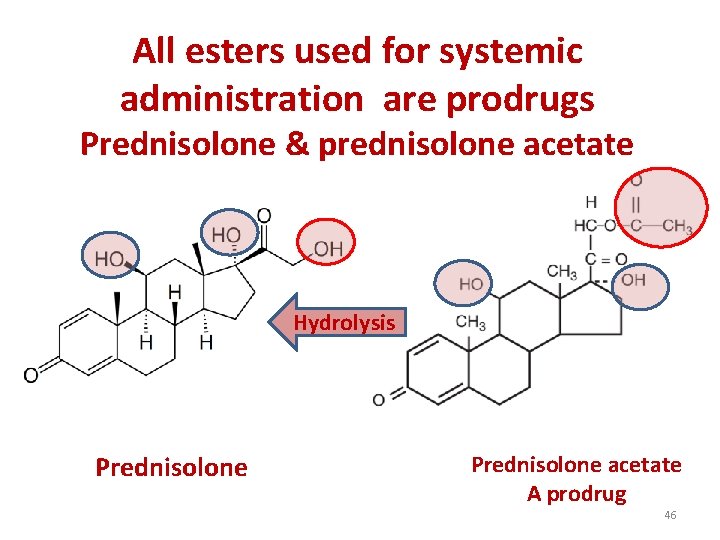
Ester are prodrugs • Practically all esters except beclomethasone 17 -monoprpionate and fluticasone propionate are inactive prodrugs and have to be hydrolyzed to release their active moiety because an OH radical at C-21 is necessary for the binding of corticoids to their cellular receptors. 44

Hydrolysis by esterases • Hydrolysis by esterases may occur either in different body fluids such as blood or synovial fluid (as for methylprednisolone acetate for which the half-life is about 1 h in synovial fluid) or mainly in liver (as for succinate), meaning that for a local administration, the judicious selection of an appropriate ester is in order 45

All esters used for systemic administration are prodrugs Prednisolone & prednisolone acetate Hydrolysis Prednisolone acetate A prodrug 46

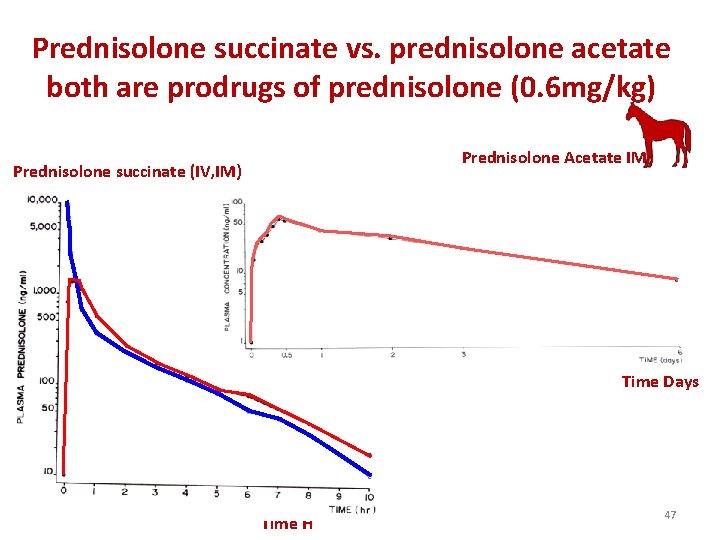
Prednisolone succinate vs. prednisolone acetate both are prodrugs of prednisolone (0. 6 mg/kg) Prednisolone Acetate IM Prednisolone succinate (IV, IM) Time Days Time H 47

Prednisolone succinate vs. prednisolone acetate • illustrates the differences between the disposition of prednisolone after administration of prednisolone succinate by the IV and IM routes and after prednisolone acetate administration by the IM route. 48

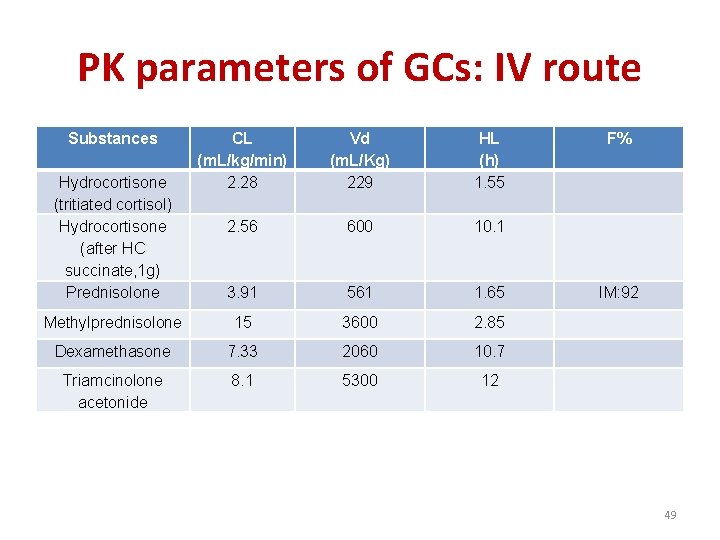
PK parameters of GCs: IV route Substances CL (m. L/kg/min) 2. 28 2. 56 Vd (m. L/Kg) 229 HL (h) 1. 55 F% 600 10. 1 3. 91 561 1. 65 IM: 92 Methylprednisolone 15 3600 2. 85 Dexamethasone 7. 33 2060 10. 7 Triamcinolone acetonide 8. 1 5300 12 Hydrocortisone (tritiated cortisol) Hydrocortisone (after HC succinate, 1 g) Prednisolone 49

Corticosteroid esters and local routes of administration 50

Intra-articular administration 51

Heathrow EHLSC 2008

Musculoskeletal injury following local corticosteroid injection in Thoroughbred racehorses • Veterinary records for 1911 thoroughbred • Thoroughbred racehorses receiving local corticosteroid injection (LCI) suffer musculoskeletal injury MSIs at approximately 4. 5 times the rate of horses not receiving treatment, and for horses receiving multiple LCI the rate is approximately twice that of horses receiving single LCIs. Whitton, C. , et al 2012 Faculty of Veterinary Science, University of Melbourne, Victoria, 3030, Australia. 53

What joint? • It seems impossible to investigate all joints • The largest and the most reproducible joint (in term of administration accuracy) should be selected? 54

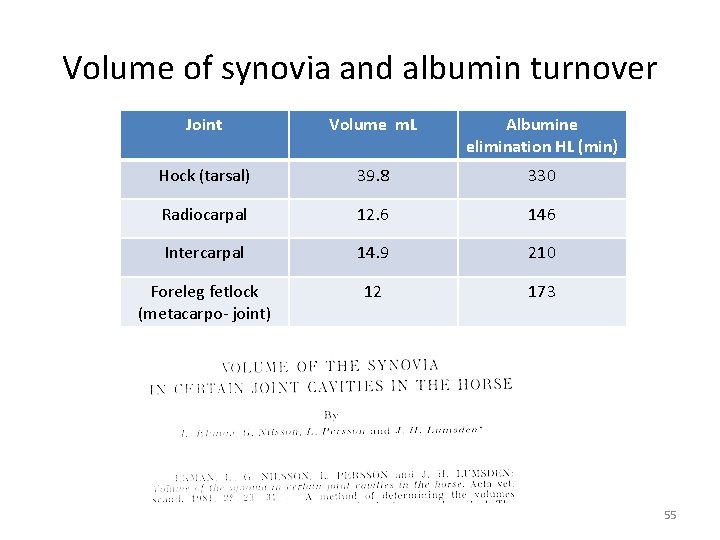
Volume of synovia and albumin turnover Joint Volume m. L Albumine elimination HL (min) Hock (tarsal) 39. 8 330 Radiocarpal 12. 6 146 Intercarpal 14. 9 210 Foreleg fetlock (metacarpo- joint) 12 173 55

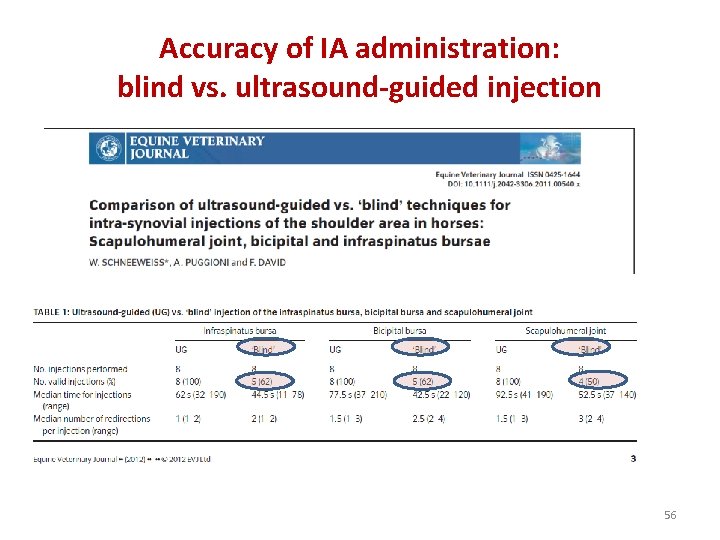
Accuracy of IA administration: blind vs. ultrasound-guided injection 56

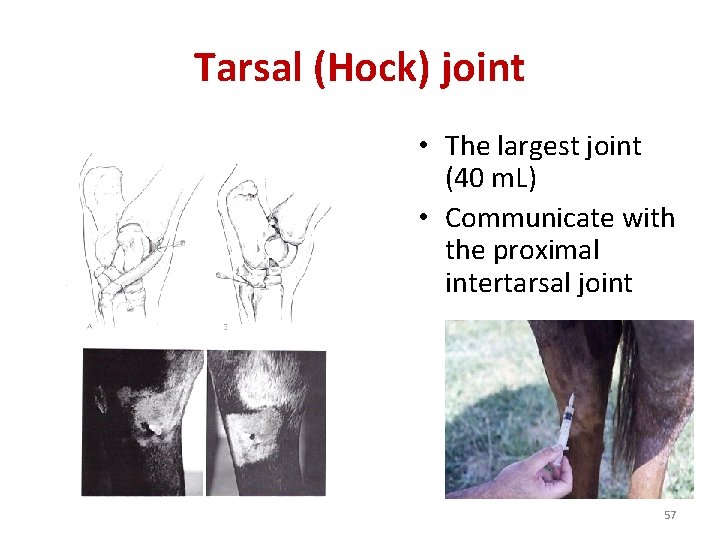
Tarsal (Hock) joint • The largest joint (40 m. L) • Communicate with the proximal intertarsal joint 57

Disposition of methylprednisolone in the synovial fluid • Disappearance of MPA within 6 days but a persistence of its active moiety for 5 to 39 days depending on the horse. The terminal half-life of MP was between 81 and 261 h: 58

Methylprednisolone acetate (MPA): IA • Disappearance of MPA within 6 days but a persistence of its active moiety for 5 to 39 days depending on the horse. • The terminal half-life of MP was between 81 and 261 h: • the reason for the persistence of MP was most likely due to a precipitation of MPA that adhers to the synovial membrane • This likely explains why MPA is not well tolerated in horses (Mc. Ilwraith, 2010) and that other GC are now preferred for IA administration, namely betamethasone esters and triamcinolone acetonide 59

Origin of the sustained release of MP after a MPA administration • the reason for the persistence of MP was most likely due to a precipitation of MPA that adhers to the synovial membrane and most likely acts as a foreign body. • This likely explains why MPA is not well tolerated in horses (Mc. Ilwraith, 2010) and that other GC are now preferred for IA administration, namely betamethasone esters and triamcinolone acetonide 60

Triamcinolone acetonide • TA is available as a sterile crystalline suspension. • This formulation is practically insoluble in water and provides a depot effect with constant release of the active agent from the injection site over a long period of time. • Apparently TA is well tolerated by the joint 61

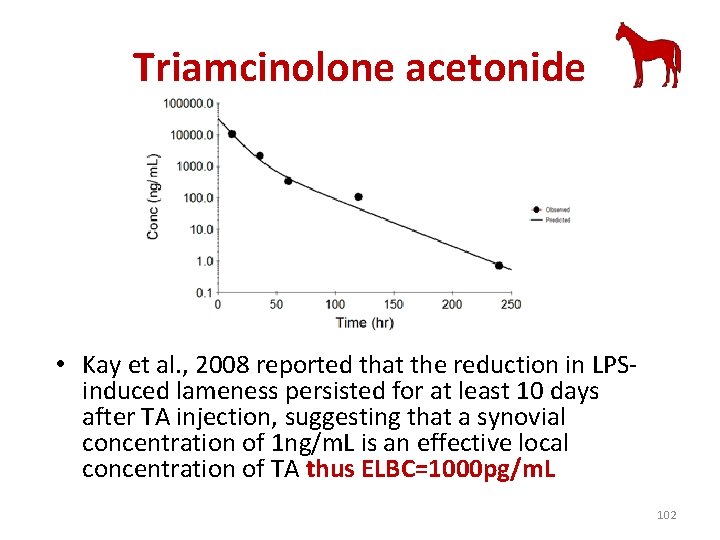
Triamcinolone acetonide • after 10 days and Kay et al (Kay et al. , 2008) reported that the reduction in LPS-induced lameness persisted for at least 10 days after TA injection, confirming that a synovial concentration of 1 ng/m. L is an effective local concentration of TA 62

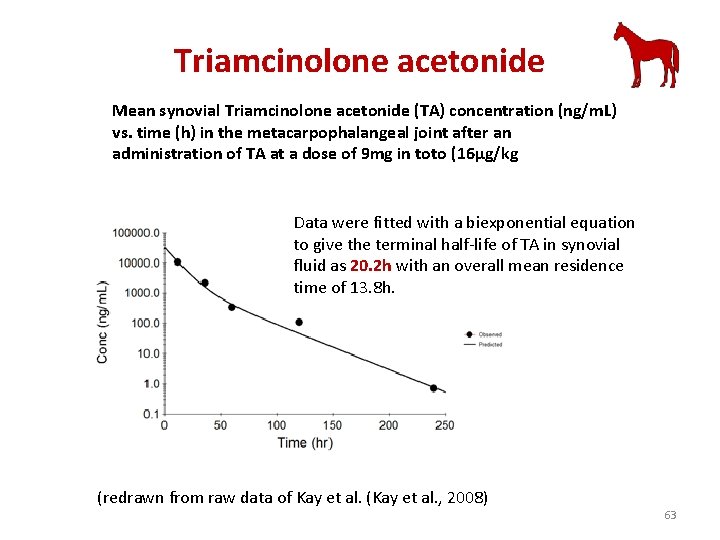
Triamcinolone acetonide Mean synovial Triamcinolone acetonide (TA) concentration (ng/m. L) vs. time (h) in the metacarpophalangeal joint after an administration of TA at a dose of 9 mg in toto (16µg/kg Data were fitted with a biexponential equation to give the terminal half-life of TA in synovial fluid as 20. 2 h with an overall mean residence time of 13. 8 h. (redrawn from raw data of Kay et al. (Kay et al. , 2008) 63

GC Inhalation 64

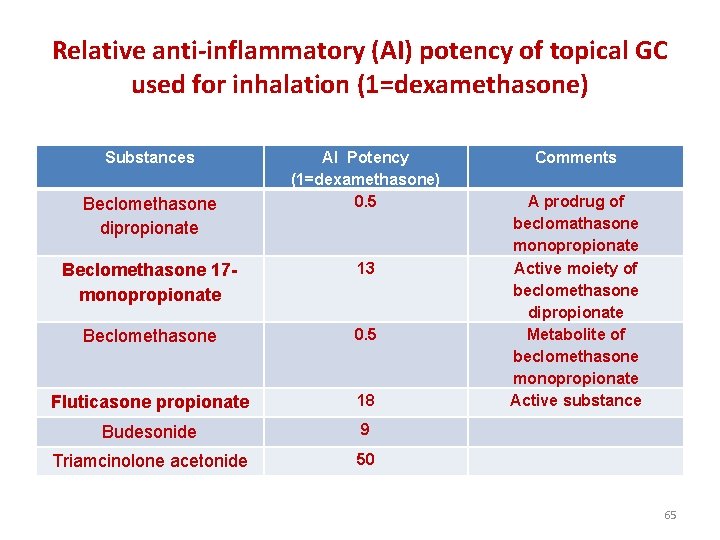
Relative anti-inflammatory (AI) potency of topical GC used for inhalation (1=dexamethasone) Substances AI Potency (1=dexamethasone) 0. 5 Comments Beclomethasone 17 monopropionate 13 Beclomethasone 0. 5 Fluticasone propionate 18 A prodrug of beclomathasone monopropionate Active moiety of beclomethasone dipropionate Metabolite of beclomethasone monopropionate Active substance Budesonide 9 Triamcinolone acetonide 50 Beclomethasone dipropionate 65

Many devices: are they equivalent? In horses, the pulmonary bioavailability is rather a function of the delivery device used for inhalation than a property of the drug. Cortic 00 A. 66

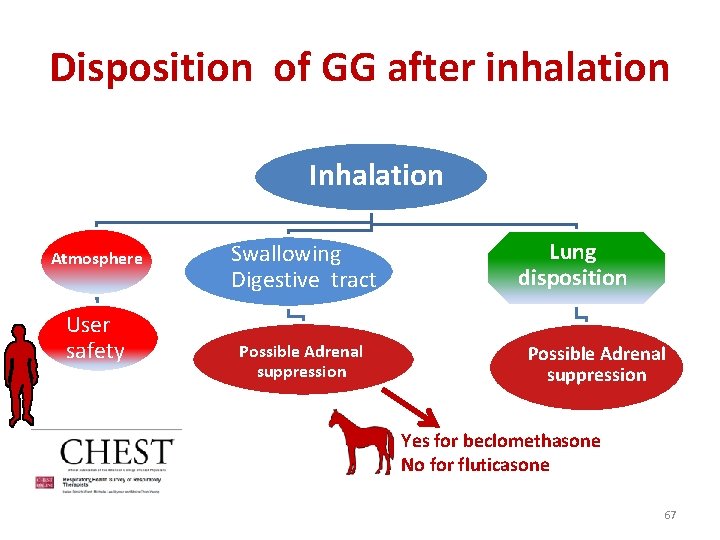
Disposition of GG after inhalation Inhalation Atmosphere User safety Swallowing Digestive tract Possible Adrenal suppression Lung disposition Possible Adrenal suppression Yes for beclomethasone No for fluticasone 67

Inhalation treatment: an user safety issue? • Nebulized medication that is released into the atmosphere from the nebulizer or exhaled by the patient becomes a form of "secondhand" exposure that may affect health-care providers and others in the vicinity of the treatment 68

Systemic disposition of inhaled GS • Beclomethasone dipropionate (a prodrug of beclomethasone 17 -monopropionate) and fluticasone propionate are the two local GC used most in the horse. • A variety of inhaler devices are used and they deposit GC in both the lungs and the oropharynx. • A critical issue for this modality of administration is to accurately know the delivered dose and also the particle size distribution. • The pulmonary bioavailability is rather a function of the delivery device used for inhalation than a property of the drug. In clinical investigations, it is important to provide precise details about the inhaler device and how it was used because not all devices are equivalent. 69

Fluticasone propionate • For example swallowed fluticasone propionate is extensively metabolized by a hepatic first-pass effect and the plasma profile after inhalation reflects only the deposition in, and absorption through the lungs with a bioavailability of about 17% in man (Winkler et al. , 2004). • This also explains that fluticasone propionate in horse (2 mg, q 12 h) can decrease neutrophilia in the airways and respiratory effort without suppressing adrenal gland function (Giguere et al. , 2002). • For a 4 week fluticasone treatment, with a dose of 1980µg q 12 h for the first two weeks, followed by a dose of 1100µg per 24 h for 1 week and finally 1100µg q for 48 h, there was no adrenal suppression in horse (Couetil et al. , 2005). • The lack of adrenal suppression indicates that this endpoint will be not be useable to screen horses under fluticasone propionate treatment. 70

Beclomathasone 17 -monopropionate • Beclomethasone propionate (0. 5 -1. 5 mg twice daily): after inhalation, the fraction of beclomethasone dipropionate (BDP) gaining access to lung is immediately metabolized by lung esterases to its active metabolite, beclomethasone 17 -monopropionate (BMP). • The fraction of BDP that is swallowed is also transformed into BMP in the small intestine and the bioavailability of BMP (about 20% in man) is sufficient to partially suppress adrenal function in horses by 3550% within 24 h for a BDP dose >1 mg, BID)(Rush et al. , 1999). 71

Budesonide • For budesonide, the first-pass inactivation is 89% in man i. e. between fluticasone propionate (99%) and BDP (80%). • The mean absorption times which reflect the duration of lung retention in man are 5 -7 h for fluticasone propionate, 2. 9 h for TA and 1 h for budesonide 72

Inhaled Corticosteroids (ICS) • Inhaled corticosteroids (ICS) are now considered the first-line therapy in treating asthma and are approved for chronic use in children as young as 12 months of age. 73

Pharmacodynamic of inhaled GCs • DXM (0. 1 mg/kg s. i. d) reduces airway obstruction by day 3 of administration and the maximal response is obtained by day 7 (Rush et al. , 2000). • • After the discontinuation of BDP administration, the return to pretreatment values was also slowly observed over 3 to 7 days (Rush et al. , 2000) and after fluticasone propionate, the improvement persisted for 2 weeks when the horses were placed in a low-dust environment (Couetil et al. , 2005) 74

Pharmacodynamic of inhaled GS • Such a progressive establishment of clinical effects and return to pretreatment status makes it difficult or even impossible to establish any PK/PD relationship useful for drug monitoring. • In addition, the dose-effect relationship is flat and difficult to establish. 75

Inhalation • A systemic route is used for severe conditions and a TA dose of 90µg/kg can relieve airway obstruction for 4 weeks (Rush 2001) (Rush, 2001). • The pulmonary route offers the advantage of delivering the drug selectively where it is needed allowing relatively low doses to be used thus minimizing adverse systemic effects. • In addition specific GC have been developed for inhalation in order to be very potent (to reduce the administered volume), and to have a long pulmonary residence time while the systemic elimination remains rapid. 76

Beclomethasone dipropionate • Beclomethasone dipropionate is partly hydrolyzed at position C-21 in the lungs, resulting in its activation to beclomethasone 17 -monopropionate thus beclomethasone dipropionate is a prodrugs that require activation to exert its anti-inflammatory activity by binding to pulmonary glucocorticoid receptors. 77

Determination of a relevant and an irrelevant plasma concentration for GC: the use of PK/PD concepts and their limits 78

Fluticasone propionate • Environmental management is the most important factor in the treatment of RAO but early GC treatment can help accelerate the recovery of a horse with severe RAO (Couetil et al. , 2005). • GCs are only a symptomatic treatment and RAO-affected horses cannot be cured if they are maintained in a dusty environment 79

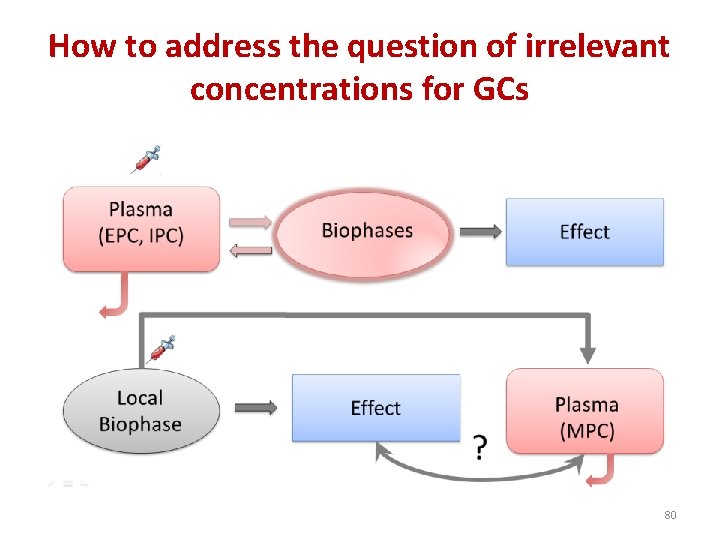
How to address the question of irrelevant concentrations for GCs 80

An example: The dexamethasone systemic effect 81

The PK/PD approach to determine irrelevant plasma or urine drug concentrations Steps : 1: effective plasma concentration (EPC) 2: Irrelevant plasma concentration (IPC) 3: Irrelevant urine concentration (IUC) 82

Risk assessment: determination of IPC and IUC 83

Computation of an Effective Plasma Concentration (EPC) 84

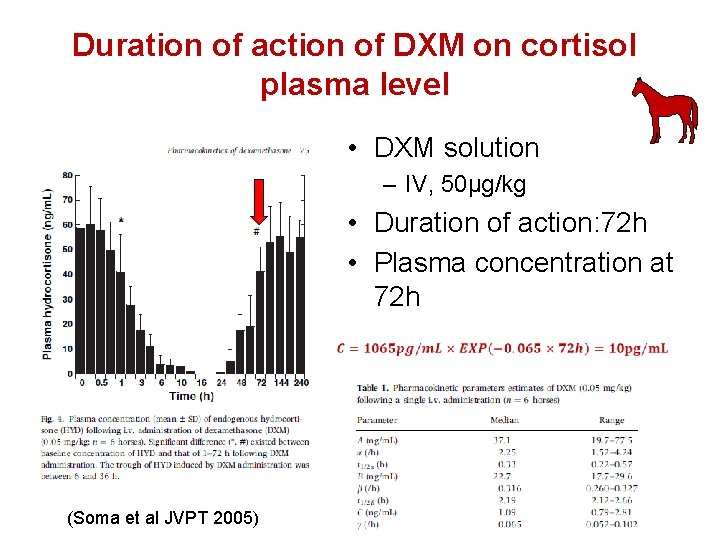
Step 1 : example of Dexamethasone • Standard dose: equivalent to 40µg/kg/24 h • Plasma clearance: 440 m. L/kg/h or about 10000 m. L/kg/24 h (Soma et al JVPT 2005) 40 000 ng. kg-1. 24 h-1 = 4 ng/m. L EPC = -1 -1 1000 m. L. kg. 24 h Note 1: from PK/PD experiment, 1 to 4 ng/m. L 85

Step 2: computation of irrelevant plasma concentrations • An IPC can be deduced from EPC by applying a safety factor (SF) to EPC: IPC = ! EPC SF How to select SF? 86

Safety factor: default value=500 • I proposed a default SF = 500 = 50 x 10 Interindividual horse variability Transform an EPC into an IPC for a given horse PK variability PD variability 3. 3 87

DXM: computation of an irrelevant plasma concentration (IPC) 50 X 10 by rounding up : 10 pg/m. L 88

Relationship between the CV% and the validity of the default uncertainty factor (Log-normal distribution) For man and a variety of substances, the number of subjects not covered by a factor of 3. 16 is about of 1/117 for PK and 1/52 for PD The probability of the same individual falling outside the range for both PK and PD is about 1/6172 Probability of false positive for our thresholds (as cortisol): 1/10000 ECVPT Toulouse 2009 - 89

Duration of action of DXM on cortisol plasma level • DXM solution – IV, 50µg/kg • Duration of action: 72 h • Plasma concentration at 72 h (Soma et al JVPT 2005)

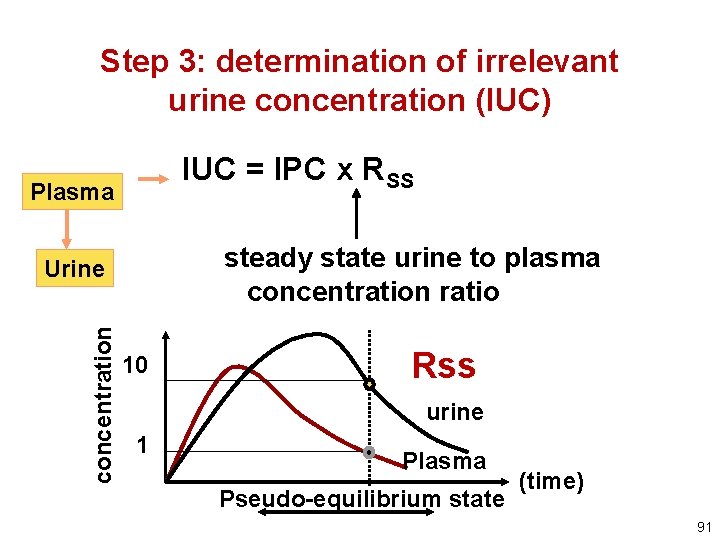
Step 3: determination of irrelevant urine concentration (IUC) IUC = IPC x RSS Plasma steady state urine to plasma concentration ratio concentration Urine 10 Rss urine 1 Plasma Pseudo-equilibrium state (time) 91

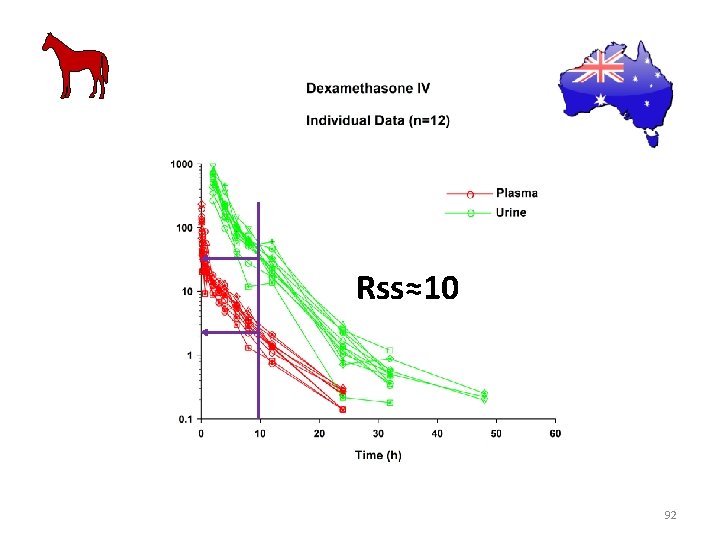
Rss≈10 92

Step 3: determination of irrelevant urine concentration (IUC)

Risk analysis for DXM 1. Risk assessment IPC=10 pg/m. L IUC=100 pg/m. L 1. Risk management Selection of a screening limit Science + other considerations differences expected between organizations: harmonization 2. Risk communication Monte. Carlo-Orlando 06 - 94

The 3 main steps of a Risk analysis 1. Risk assessment 2. Risk management 3. Risk communication Large differences expected between organization with many possible options to communicate with stakeholders Exchanges of information Monte. Carlo-Orlando 06 - 95

Determination of a relevant and an irrelevant plasma concentration for GC for a local route of administration and a local effect

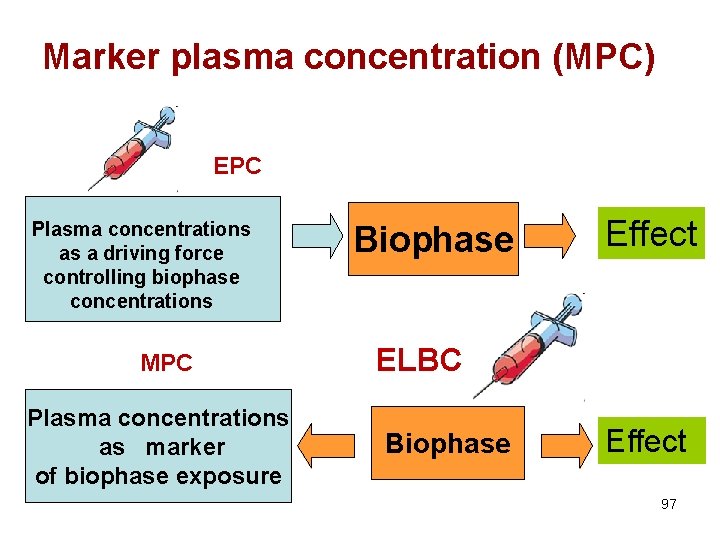
Marker plasma concentration (MPC) EPC Plasma concentrations as a driving force controlling biophase concentrations MPC Plasma concentrations as marker of biophase exposure Biophase Effect ELBC Biophase Effect 97

The case where plasma concentration is not the driving force controlling effect • For any drug administered locally to develop a local effect, plasma drug concentrations is not the driving force controlling drug concentrations at the biophase (i. e. the site of action). • Plasma concentration It is only a marker of a local exposure 98

Two preliminary definitions 1. Effective local biophase concentration (ELBC) • is the biophase drug concentration for which effects are observed (typically reported as a EC 50) 2. Marker Plasma Concentration (MPC) • MPC is the plasma concentration corresponding to the ELBC 99

The MPC vs. EPC • At variance of EPC, MPC is not a pharmacodynamic parameter expressing substance potency • It is an exposure variable determined by the administered pharmaceutical form and route of administration – A single EPC vs. several possible MPCs – With EPC , we can control drug effect – With MPC , we can help vets to ensure good veterinary practices 100

Step 1: define an ELBC • To select an ELBC, it is reasonable to assume that the ELBC is of the same order of magnitude as the plasma EPC or an EC 50 obtained from some relevant in vitro test system, –. 101

Triamcinolone acetonide • Kay et al. , 2008 reported that the reduction in LPSinduced lameness persisted for at least 10 days after TA injection, suggesting that a synovial concentration of 1 ng/m. L is an effective local concentration of TA thus ELBC=1000 pg/m. L 102

Step 2: Select an articulation and a dose fitting good veterinary practices • Very different veterinary practices • Require harmonisation • For triamcinolone acetonide, dose is around 10 mg per joint 103

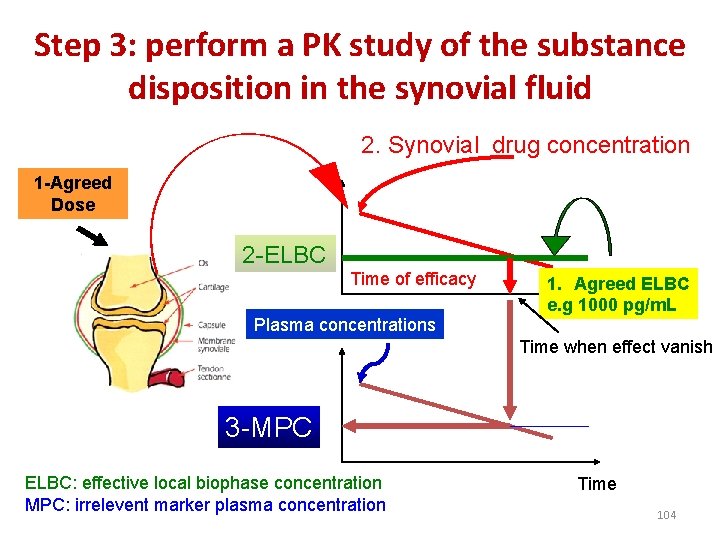
Step 3: perform a PK study of the substance disposition in the synovial fluid 2. Synovial drug concentration 1 -Agreed Dose 2 -ELBC Time of efficacy Plasma concentrations 1. Agreed ELBC e. g 1000 pg/m. L Time when effect vanish 3 -MPC ELBC: effective local biophase concentration MPC: irrelevent marker plasma concentration Time 104

Step 4: computation of an MPC for triamcinolone acetonide • Such a low plasma concentration is not detectable with the current analytical techniques (LOD of about 10 pg/ml) and the plasma concentration cannot be used to monitor an IA administration of TA This is due to the high TA plasma clearance (8 m. L/kg/min) 105

Step 5: Transformation of an MPC to an irrelevant MPC (IMPC) for all horses To take into account interanimal variability 106

Step 5: computation of an IMPC for triamcinolone acetonide • Such a low plasma concentration is not detectable with the current analytical techniques (LOD of about 10 pg/ml) and the plasma concentration cannot be used to monitor an IA administration of TA 107

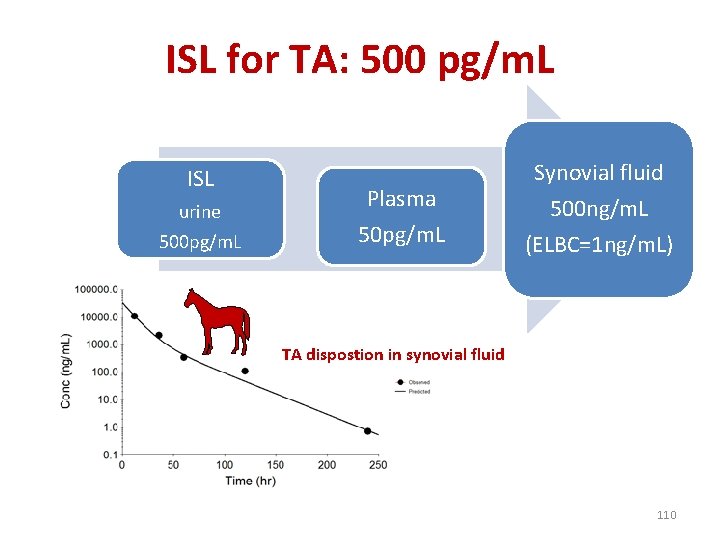
Step 6: Irrelevant urine concentration (IUC) for triamcinolone acetonide • Even for urine, it is impossible to control properly an IA of TA i. e. to guarantee to actually be able to detect urine concentrations corresponding to the irrelevant synovial TA concentrations. ISL=500 pg/m. L 108

What is the synovial concentration of TA that can be controlled with a LOD of 10 pg/m. L in urine? • A: about 8. 8 ng/ml i. e. at day 7 i. e. much above the effective synovial concentration • It would be necessary to follow TA up to 13 days after a IA administration of 9 mg in toto of TA to guarantee a lack of effect 109

ISL for TA: 500 pg/m. L ISL urine 500 pg/m. L Plasma 50 pg/m. L Synovial fluid 500 ng/m. L (ELBC=1 ng/m. L) TA dispostion in synovial fluid 110

MPC vs. EPC & IPC • An MPC is not a PD parameter but a variable associated with a given formulation, for a given dose, a given joint etc. • An IMPC cannot be used to derive a universal SL to monitor drug efficacy; • A given MPC/IMPC can only be used to determine a relevant detection time for that formulation in order to promote good veterinary practices but not to surely select an SL 111

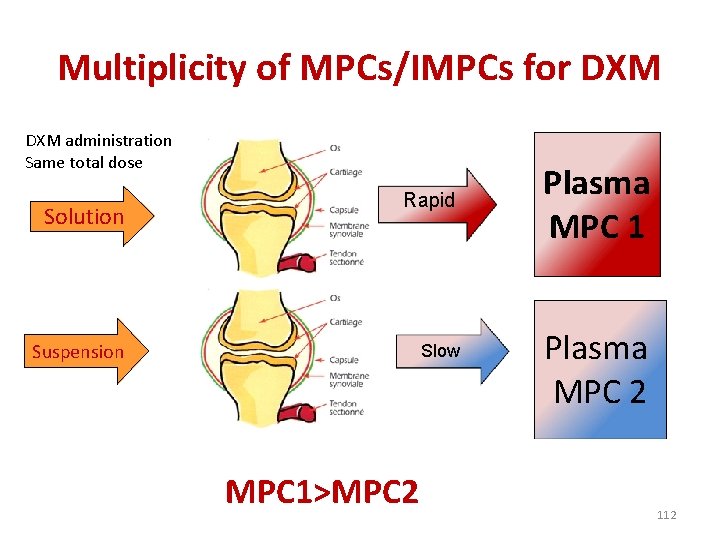
Multiplicity of MPCs/IMPCs for DXM administration Same total dose Solution Rapid Suspension Slow MPC 1>MPC 2 Plasma MPC 1 Plasma MPC 2 112

EPC vs MPC • For example, the EPC of dexamethasone is about 1 -4 ng/m. L whatever the ester and whatever the route of administration as long as we are looking at some systemic effect. • By contrast, the dexamethasone MPC for an intra -articular administration will be different for a phosphate and an acetate formulation, for a solution or a suspension of the same isonicotinate ester or for an intra-articular vs. inhalation of the same TA formulation, etc. 113

Step 6: risk management • As in the transformation of an EPC into an IPC, it is necessary to apply a safety (uncertainty) factor to the MPC. • It should be realized here that the MPC determined is a non-effective marker plasma concentration (or alternatively, as the lower limit of efficacious concentrations). • Therefore it can be suggested that a default uncertainty (safety) factor of 10 is appropriate and not 500 as in the transformation of an EPC into an IPC, because only the variability of the PK and the PD has to be considered in this case. • As for the IPC, we may coin the name of Irrelevant MPC or IMPC for this value that will be given to the risk manager to finally select a screening limit. 114

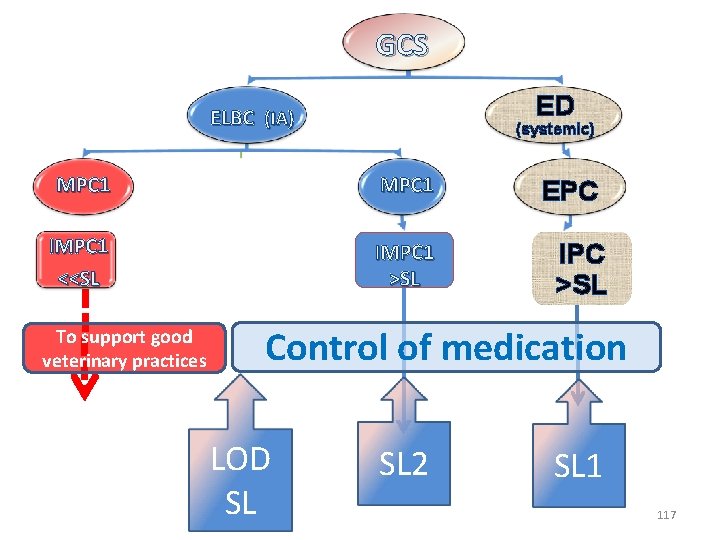
GC ELBC (IA) ED (systemic) EPC IPC >SL Control of medication LOD SL 115

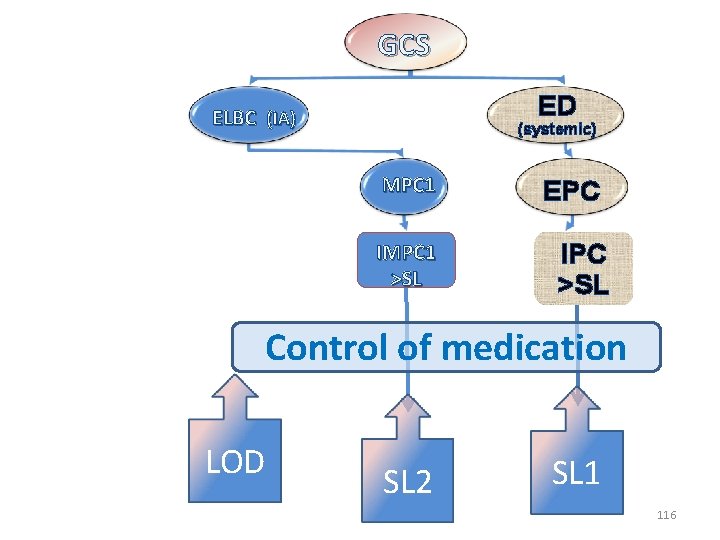
GCS ED ELBC (IA) (systemic) MPC 1 IMPC 1 >SL EPC IPC >SL Control of medication LOD SL 2 SL 1 116

GCS ED ELBC (IA) (systemic) MPC 1 IMPC 1 <<SL IMPC 1 >SL To support good veterinary practices EPC IPC >SL Control of medication LOD SL SL 2 SL 1 117

Multiplicity of MPCs and a single SL: • For glucocorticoids (GCS) administrable by both local and systemic routes, the selection of a single screening limit (SL) for medication control will be ineluctably a compromise between different possible values derived either from an irrelevant plasma concentration (IPC) or one of the several known irrelevant marker plasma concentrations (IMPC). 118

MPC for inhalation • Use a pragmatic approach • Selection by consensus of a DT • Select as SL plasma/urine concentration corresponding to the DT (HK approach) 119

Conclusions 1. GCs are well known for their systemic effect 2. GCs are not very well documented (PK & PD) after a local administration (IA, Inhalation) 3. Computation of IPC and IUC for systemic effect is easy 4. Computation of IPC and IUC is more challenging for local administration(concept of MPC) 5. It exist a single IPC for systemic effect but a multiplicity of possible IMPCs for local administration 6. Selection of a single screening limit will be a compromise 7. Analytical technics are unlikely able to control all kind of local administration 120