Clinical Outcomes At 2 Years of the Absorb

Clinical Outcomes At 2 Years of the Absorb BVS versus Xience EES in Patients Presenting with Acute Coronary Syndrome versus Stable Coronary Disease - AIDA Trial Sub-Study Laura S. M. Kerkmeijer, MD Ruben Y. G. Tijssen, MD Joanna J. Wykrzykowska, MD, Ph. D Amsterdam UMC, Amsterdam, The Netherlands

Laura S. M. Kerkmeijer, MD I have no relevant financial relationships

Background BRS use appears theoretically attractive in patients presenting with Acute Coronary Syndrome: Ø Younger Ø Less frequent previous MI or PCI Ø Soft plaques Ø Greater strut thickness may prevent thrombus dislodgement and acute thrombus prolapse

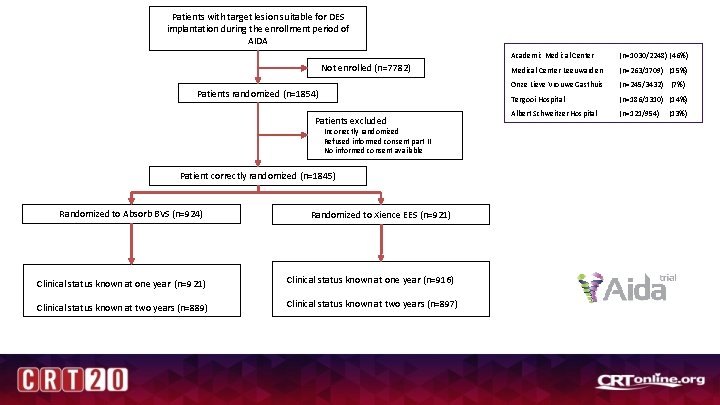
Patients with target lesion suitable for DES implantation during the enrollment period of AIDA Not enrolled (n=7782) Patients randomized (n=1854) Patients excluded Incorrectly randomized Refused informed consent part II No informed consent available Patient correctly randomized (n=1845) Randomized to Absorb BVS (n=924) Randomized to Xience EES (n=921) Clinical status known at one year (n=916) Clinical status known at two years (n=889) Clinical status known at two years (n=897) Academic Medical Center (n=1030/2248) (46%) Medical Center Leeuwarden (n=263/1709) (15%) Onze Lieve Vrouwe Gasthuis (n=245/3432) (7%) Tergooi Hospital (n=186/1310) (14%) Albert Schweitzer Hospital (n=121/954) (13%)
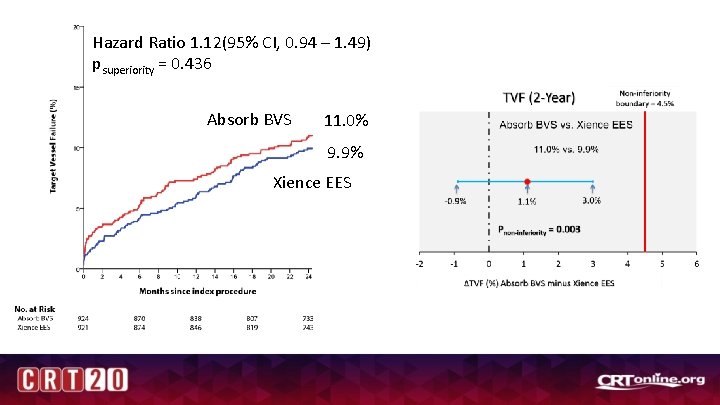
Hazard Ratio 1. 12(95% CI, 0. 94 – 1. 49) psuperiority = 0. 436 Absorb BVS 11. 0% 9. 9% Xience EES
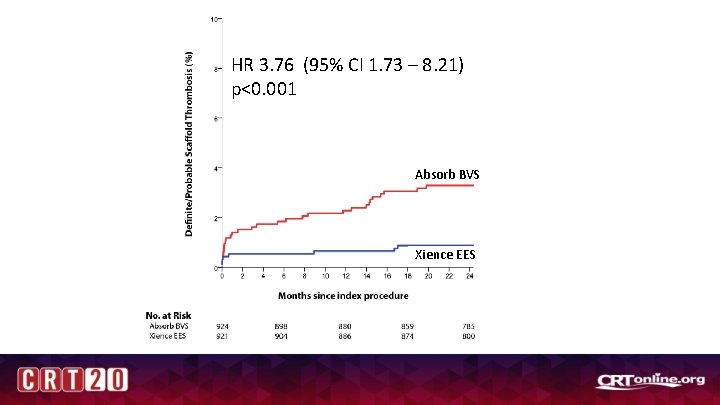
HR 3. 76 (95% CI 1. 73 – 8. 21) p<0. 001 Absorb BVS Xience EES


Definitions • Patients presented with an ACS if they were hospitalized with acute onset of symptoms. • For STEMI a planned primary PCI because of persistent ST-segment elevation was required. • For NSTEMI, both of the following was required: • ST-segment depression or transient elevation ≥ 1 mm in 2 or more leads • Positive biomarker indicating myocardial tissue necrosis
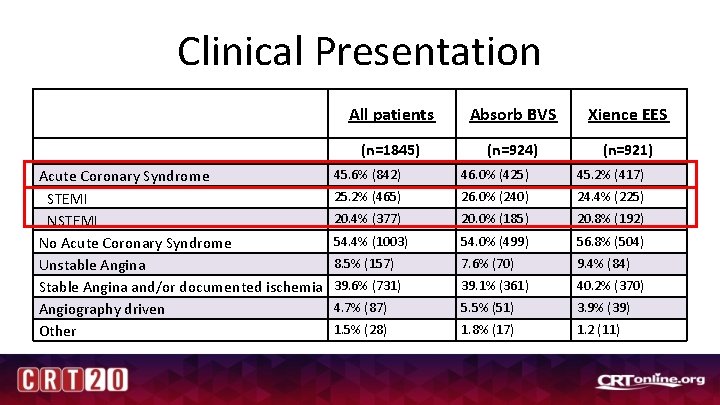
Clinical Presentation Acute Coronary Syndrome STEMI No Acute Coronary Syndrome Unstable Angina Stable Angina and/or documented ischemia Angiography driven Other All patients Absorb BVS Xience EES (n=1845) (n=924) (n=921) 45. 6% (842) 46. 0% (425) 45. 2% (417) 25. 2% (465) 26. 0% (240) 24. 4% (225) 20. 4% (377) 20. 0% (185) 20. 8% (192) 54. 4% (1003) 54. 0% (499) 56. 8% (504) 8. 5% (157) 7. 6% (70) 9. 4% (84) 39. 6% (731) 39. 1% (361) 40. 2% (370) 4. 7% (87) 5. 5% (51) 3. 9% (39) 1. 5% (28) 1. 8% (17) 1. 2 (11)
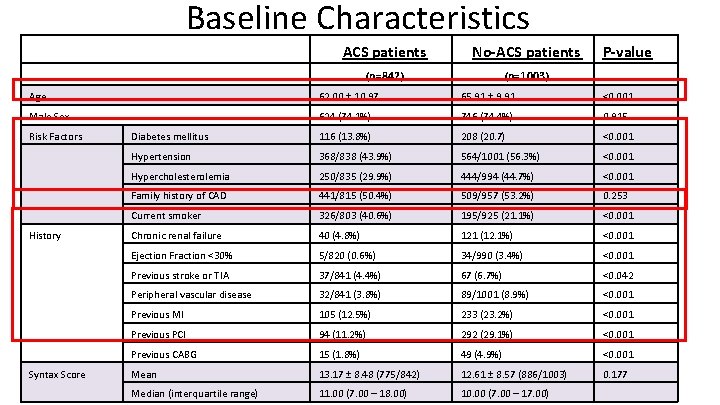
Baseline Characteristics ACS patients No-ACS patients (n=842) (n=1003) P-value Age 62. 00 ± 10. 97 65. 91 ± 9. 91 <0. 001 Male Sex 624 (74. 1%) 746 (74. 4%) 0. 915 Diabetes mellitus 116 (13. 8%) 208 (20. 7) <0. 001 Hypertension 368/838 (43. 9%) 564/1001 (56. 3%) <0. 001 Hypercholesterolemia 250/835 (29. 9%) 444/994 (44. 7%) <0. 001 Family history of CAD 441/815 (50. 4%) 509/957 (53. 2%) 0. 253 Current smoker 326/803 (40. 6%) 195/925 (21. 1%) <0. 001 Chronic renal failure 40 (4. 8%) 121 (12. 1%) <0. 001 Ejection Fraction <30% 5/820 (0. 6%) 34/990 (3. 4%) <0. 001 Previous stroke or TIA 37/841 (4. 4%) 67 (6. 7%) <0. 042 Peripheral vascular disease 32/841 (3. 8%) 89/1001 (8. 9%) <0. 001 Previous MI 105 (12. 5%) 233 (23. 2%) <0. 001 Previous PCI 94 (11. 2%) 292 (29. 1%) <0. 001 Previous CABG 15 (1. 8%) 49 (4. 9%) <0. 001 Mean 13. 17 ± 8. 48 (775/842) 12. 61 ± 8. 57 (886/1003) 0. 177 Median (interquartile range) 11. 00 (7. 00 – 18. 00) 10. 00 (7. 00 – 17. 00) Risk Factors History Syntax Score
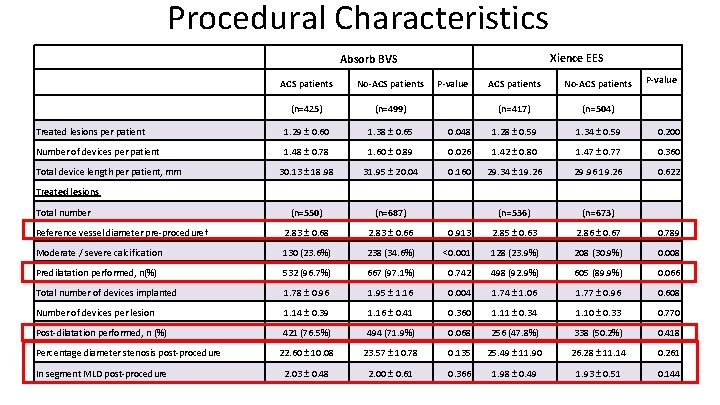
Procedural Characteristics Xience EES Absorb BVS ACS patients No-ACS patients (n=425) (n=499) Treated lesions per patient 1. 29 ± 0. 60 1. 38 ± 0. 65 Number of devices per patient 1. 48 ± 0. 78 P-value ACS patients No-ACS patients (n=417) (n=504) 0. 048 1. 28 ± 0. 59 1. 34 ± 0. 59 0. 200 1. 60 ± 0. 89 0. 026 1. 42 ± 0. 80 1. 47 ± 0. 77 0. 360 30. 13 ± 18. 98 31. 95 ± 20. 04 0. 160 29. 34 ± 19. 26 29. 96 19. 26 0. 622 (n=550) (n=687) (n=536) (n=673) Reference vessel diameter pre-procedure† 2. 83 ± 0. 68 2. 83 ± 0. 66 0. 913 2. 85 ± 0. 63 2. 86 ± 0. 67 0. 789 Moderate / severe calcification 130 (23. 6%) 238 (34. 6%) <0. 001 128 (23. 9%) 208 (30. 9%) 0. 008 Predilatation performed, n(%) 532 (96. 7%) 667 (97. 1%) 0. 742 498 (92. 9%) 605 (89. 9%) 0. 066 Total number of devices implanted 1. 78 ± 0. 96 1. 95 ± 1. 16 0. 004 1. 74 ± 1. 06 1. 77 ± 0. 96 0. 608 Number of devices per lesion 1. 14 ± 0. 39 1. 16 ± 0. 41 0. 360 1. 11 ± 0. 34 1. 10 ± 0. 33 0. 770 Post-dilatation performed, n (%) 421 (76. 5%) 494 (71. 9%) 0. 068 256 (47. 8%) 338 (50. 2%) 0. 418 22. 60 ± 10. 08 23. 57 ± 10. 78 0. 135 25. 49 ± 11. 90 26. 28 ± 11. 14 0. 261 2. 03 ± 0. 48 2. 00 ± 0. 61 0. 366 1. 98 ± 0. 49 1. 93 ± 0. 51 0. 144 Total device length per patient, mm P-value Treated lesions Total number Percentage diameter stenosis post-procedure In segment MLD post-procedure
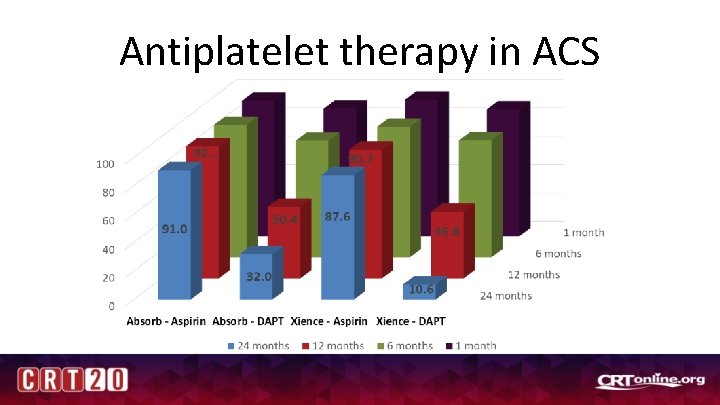
Antiplatelet therapy in ACS
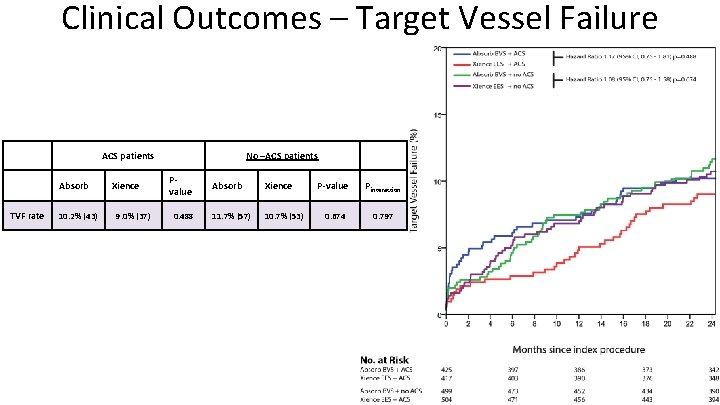
Clinical Outcomes – Target Vessel Failure ACS patients Absorb TVF rate 10. 2% (43) Xience 9. 0% (37) No –ACS patients Pvalue 0. 488 Absorb Xience 11. 7% (57) 10. 7% (53) P-value Pinteraction 0. 674 0. 797
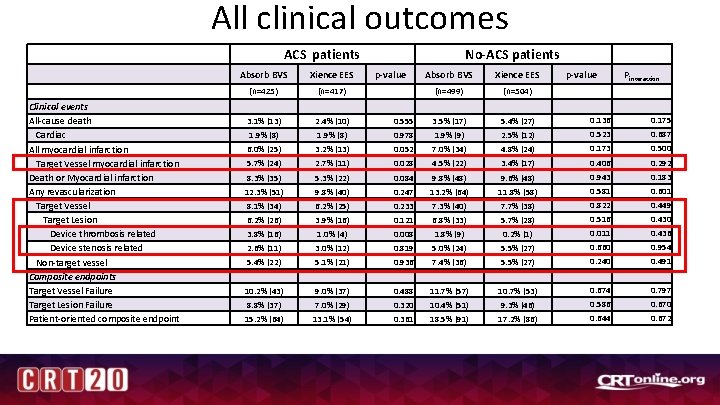
All clinical outcomes ACS patients Clinical events All-cause death Cardiac All myocardial infarction Target Vessel myocardial infarction Death or Myocardial infarction Any revascularization Target Vessel Target Lesion Device thrombosis related Device stenosis related Non-target vessel Composite endpoints Target Vessel Failure Target Lesion Failure Patient-oriented composite endpoint Absorb BVS Xience EES (n=425) (n=417) 3. 1% (13) 2. 4% (10) 1. 9% (8) No-ACS patients p-value Absorb BVS Xience EES p-value Pinteraction (n=499) (n=504) 0. 555 3. 5% (17) 5. 4% (27) 0. 136 0. 175 0. 978 1. 9% (9) 2. 5% (12) 0. 523 0. 687 0. 500 6. 0% (25) 3. 2% (13) 0. 052 7. 0% (34) 4. 8% (24) 0. 173 5. 7% (24) 2. 7% (11) 0. 028 4. 5% (22) 3. 4% (17) 0. 406 0. 292 0. 183 8. 3% (35) 5. 3% (22) 0. 084 9. 8% (48) 9. 6% (48) 0. 943 12. 3% (51) 9. 8% (40) 0. 247 13. 2% (64) 11. 8% (58) 0. 581 0. 601 0. 449 8. 1% (34) 6. 2% (25) 0. 233 7. 3% (40) 7. 7% (38) 0. 822 6. 2% (26) 3. 9% (16) 0. 121 6. 8% (33) 5. 7% (28) 0. 516 0. 430 3. 8% (16) 1. 0% (4) 0. 008 1. 8% (9) 0. 2% (1) 0. 011 0. 436 2. 6% (11) 3. 0% (12) 0. 819 5. 0% (24) 5. 5% (27) 0. 660 0. 954 5. 4% (22) 5. 1% (21) 0. 936 7. 4% (36) 5. 5% (27) 0. 240 0. 491 10. 2% (43) 9. 0% (37) 0. 488 11. 7% (57) 10. 7% (53) 0. 674 0. 797 8. 8% (37) 7. 0% (29) 0. 320 10. 4% (51) 9. 3% (46) 0. 586 0. 670 15. 2% (64) 13. 1% (54) 0. 361 18. 5% (91) 17. 2% (86) 0. 644 0. 672
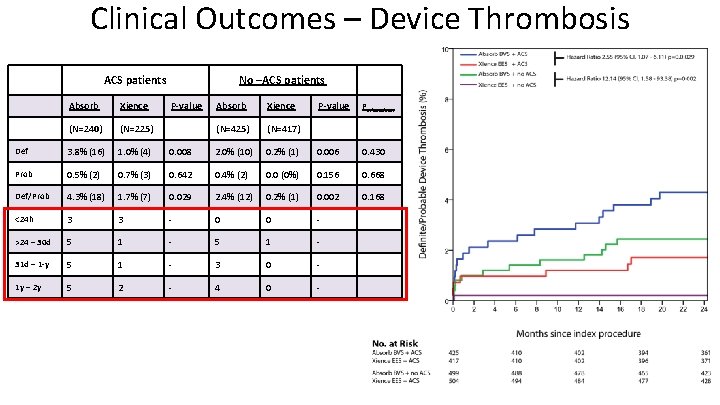
Clinical Outcomes – Device Thrombosis ACS patients Absorb Xience (N=240) (N=225) Def 3. 8% (16) 1. 0% (4) Prob 0. 5% (2) Def/Prob No –ACS patients P-value Absorb Xience P-value Pinteraction (N=425) (N=417) 0. 008 2. 0% (10) 0. 2% (1) 0. 006 0. 430 0. 7% (3) 0. 642 0. 4% (2) 0. 0 (0%) 0. 156 0. 668 4. 3% (18) 1. 7% (7) 0. 029 2. 4% (12) 0. 2% (1) 0. 002 0. 168 <24 h 3 3 - 0 0 - >24 – 30 d 5 1 - 31 d – 1 -y 5 1 - 3 0 - 1 y – 2 y 5 2 - 4 0 -
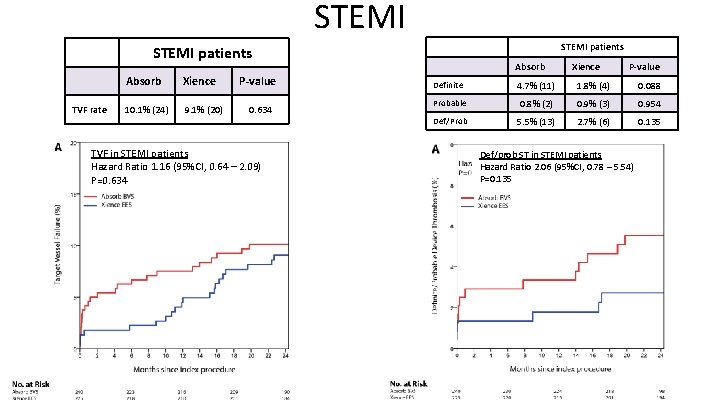
STEMI patients TVF rate Absorb Xience 10. 1% (24) 9. 1% (20) P-value 0. 634 TVF in STEMI patients Hazard Ratio 1. 16 (95%CI, 0. 64 – 2. 09) P=0. 634 Absorb Xience P-value Definite 4. 7% (11) 1. 8% (4) 0. 088 Probable 0. 8% (2) 0. 9% (3) 0. 954 Def/Prob 5. 5% (13) 2. 7% (6) 0. 135 Def/prob ST in STEMI patients Hazard Ratio 2. 06 (95%CI, 0. 78 – 5. 54) P=0. 135

Conclusions • We found no difference in rates of Target Vessel failure between the Absorb BVS and Xience EES groups. • Rates of definite/probable device thrombosis were higher in the Absorb BVS group throughout all clinical presentations. • No significant interaction between ACS and no-ACS patients and device for the occurence of TVF or device thrombosis was found.

Published in Catheter Cardiovasc Interv. 2019

Thank you for your attention!
- Slides: 20