Clinical Event Committee CEC C Michael Gibson M

Clinical Event Committee (CEC) C. Michael Gibson, M. S. , M. D. Harvard Medical School Chairman, PERFUSE Study Group Founder and Chairman, Wiki. Doc & Wiki. Patient, The World’s Open Source Textbook of Medicine Viewed 896 Million Times A Year

What is a CEC • Clinical Event Committee (CEC) – A group of medical experts who's purpose is to review predetermined adverse events over the course of a trail • Clinical Events Committees (CECs) are generally recommended for studies that evaluate treatments that prolong life or reduce risk of a major adverse health outcome. • The CEC provides an independent, unbiased and objective adjudication of the protocol-specified events for a trial. – Use of an independent CEC for review and analysis of events will ensure the adjudication process is as free from bias as possible. – Depending on the trial, the CEC may be blinded to the treatment – High quality adjudicated data provides insight into the safety and efficacy of a trail

Member Selection • Physician specialty and background – Selection is based on the types of endpoints and method of adjudication – Physicians should be independent of the trial and the study sponsor • Endpoint adjudication requires more than just a medical understanding of clinical outcomes. – The CEC Charter helps to provide guidance during the adjudication process • Minimum quorum requirement is typically 3 members – The majority of members should be board certified clinicians with previous adjudication experience

Event Adjudication • Clinical endpoint events should be predetermined during the trial design and defined in the study protocol – Standardized definitions improve consistency and quality • Classification of study endpoints – Device, Procedure, Drug, Delivery System – Not related, possibly related, probably related, causal relationship • Outcomes are dependent on the quality of the available subject – High quality relevant source – CEC may favor a conservative approach in the absents of sufficient data • Absents of evidence in not evidence of absents
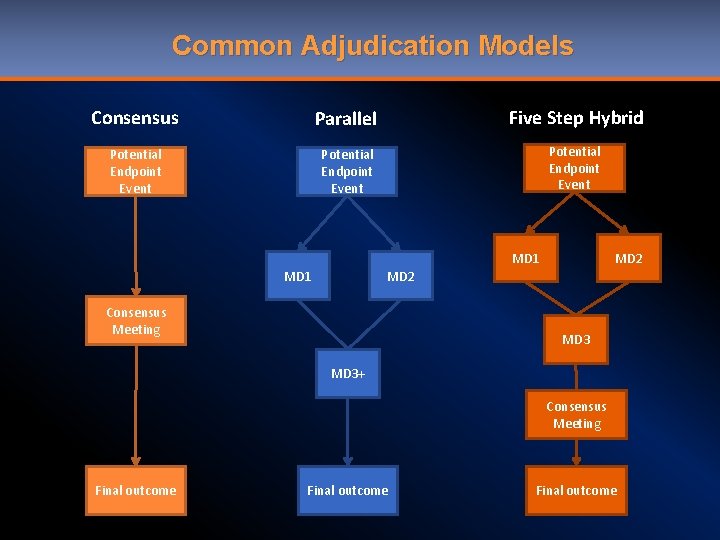
Common Adjudication Models Consensus Parallel Five Step Hybrid Potential Endpoint Event MD 1 MD 2 Consensus Meeting MD 3+ Consensus Meeting Final outcome

Adjudication Model Comparison Consensus Pros Cons • • Great for complex endpoints Increases collaboration Diverse physician specialties Long term consistency • Power Gradient • Dependent on common physician availability • More administration • Can be more time consuming Parallel • Independent • No power gradient • High volume • Remote • Gold standard for certain therapeutic areas • Challenging for complex adjudications • Must be highly defined • Can be more expensive • May need to retrain outliers
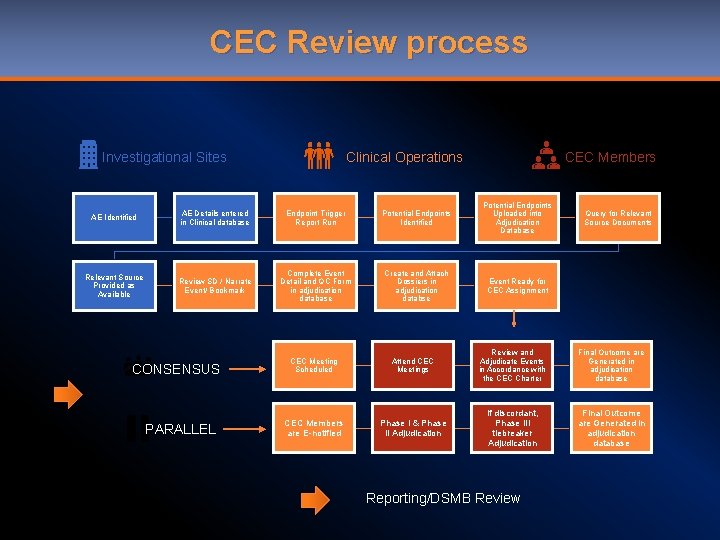
CEC Review process CEC Members Clinical Operations Investigational Sites AE Identified AE Details entered in Clinical database Endpoint Trigger Report Run Potential Endpoints Identified Potential Endpoints Uploaded into Adjudication Database Relevant Source Provided as Available Review SD / Narrate Event/ Bookmark Complete Event Detail and QC Form in adjudication database Create and Attach Dossiers in adjudication databse Event Ready for CEC Assignment CONSENSUS PARALLEL Query for Relevant Source Documents CEC Meeting Scheduled Attend CEC Meetings Review and Adjudicate Events in Accordance with the CEC Charter Final Outcome are Generated in adjudication database CEC Members are E-notified Phase I & Phase II Adjudication If discordant, Phase III tiebreaker Adjudication Final Outcome are Generated in adjudication database Reporting/DSMB Review
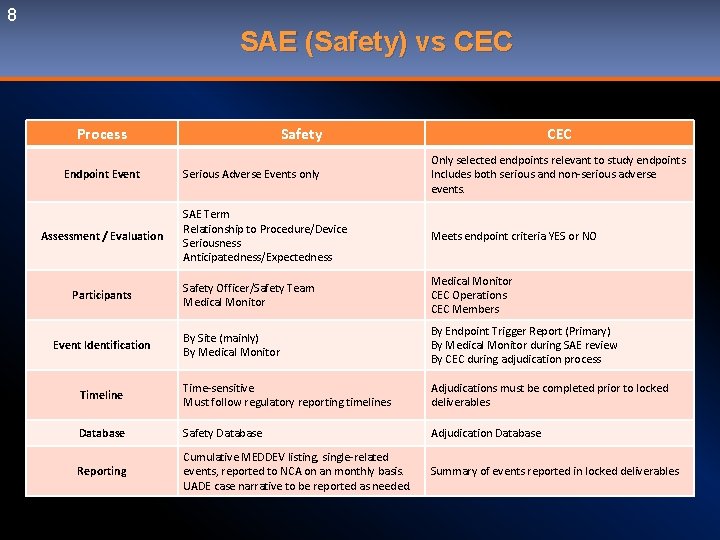
8 SAE (Safety) vs CEC Process Safety CEC Serious Adverse Events only Only selected endpoints relevant to study endpoints Includes both serious and non-serious adverse events. SAE Term Relationship to Procedure/Device Seriousness Anticipatedness/Expectedness Meets endpoint criteria YES or NO Safety Officer/Safety Team Medical Monitor CEC Operations CEC Members By Site (mainly) By Medical Monitor By Endpoint Trigger Report (Primary) By Medical Monitor during SAE review By CEC during adjudication process Timeline Time-sensitive Must follow regulatory reporting timelines Adjudications must be completed prior to locked deliverables Database Safety Database Adjudication Database Reporting Cumulative MEDDEV listing, single-related events, reported to NCA on an monthly basis. UADE case narrative to be reported as needed. Summary of events reported in locked deliverables Endpoint Event Assessment / Evaluation Participants Event Identification

END

Reference Slides • Source Document Transmittal Form • Adjudication form
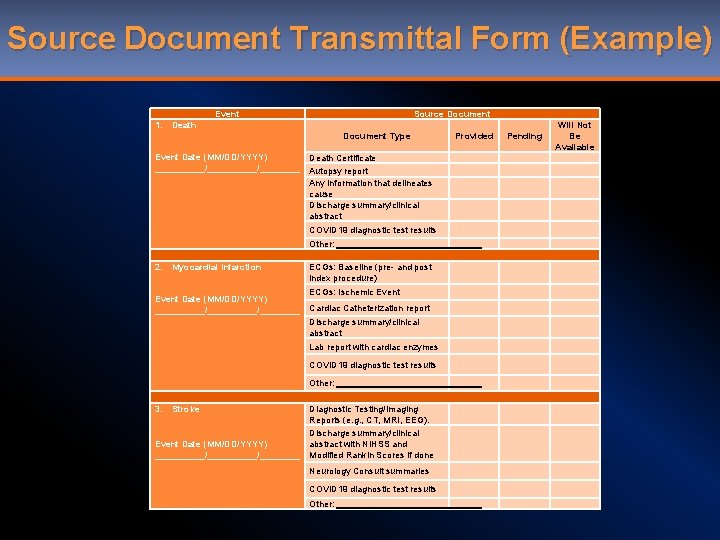
Source Document Transmittal Form (Example) Event 1. Source Document Death Document Type Event Date (MM/DD/YYYY) __________/____ Death Certificate Autopsy report Any information that delineates cause Discharge summary/clinical abstract COVID 19 diagnostic test results Other: 2. Myocardial Infarction Event Date (MM/DD/YYYY) __________/____ ECGs: Baseline (pre- and post index procedure) ECGs: Ischemic Event Cardiac Catheterization report Discharge summary/clinical abstract Lab report with cardiac enzymes COVID 19 diagnostic test results Other: 3. Stroke Event Date (MM/DD/YYYY) __________/____ Diagnostic Testing/Imaging Reports (e. g. , CT, MRI, EEG). Discharge summary/clinical abstract with NIHSS and Modified Rankin Scores if done Neurology Consult summaries COVID 19 diagnostic test results Other: Provided Pending Will Not Be Available
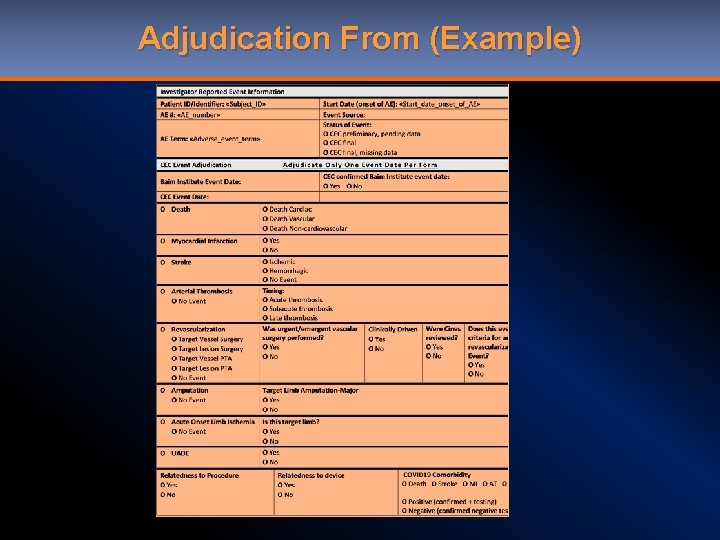
Adjudication From (Example)
- Slides: 12