Clinical development of biosimilar medicinal agents Sndor KerpelFronius

Clinical development of biosimilar medicinal agents Sándor Kerpel-Fronius, M. D. , D. Sc. Semmelweis University Department of Pharmacology and Pharmacotherapy Budapest, Hungary Email: kerpel-fronius. sandor @med. semmelweis-uni. hu. Kerpel-Fronius S. 1

Grouping of medicinal products EMEA: Annex 1 Directive 2001/83 Annex 1 (as ammended by Directive 2003/63 (Biological medicinal products) v Chemical medicinal products Ø Medicines containing chemical active ingredient(s) Ø Small molecules which can be accurately characterized chemically v Biological medicinal products Ø Medicines containing biological material, macromolecules produced by or extracted from a biological source Ø Cannot be fully characterized due to the structural complexity of the macromolecules Ø There is a need to use a combination of physico-chemical -biological testing, together with process control, to define their quality and characteristics Kerpel-Fronius S. 2
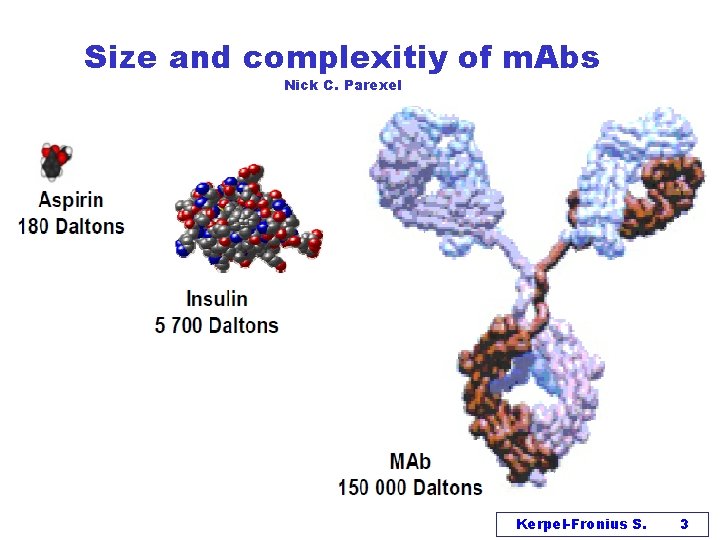
Size and complexitiy of m. Abs Nick C. Parexel Kerpel-Fronius S. 3
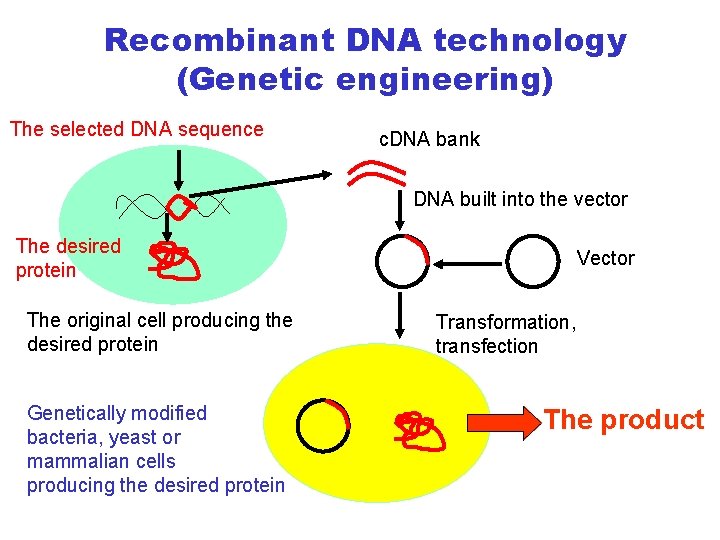
Recombinant DNA technology (Genetic engineering) The selected DNA sequence c. DNA bank DNA built into the vector The desired protein The original cell producing the desired protein Genetically modified bacteria, yeast or mammalian cells producing the desired protein Vector Transformation, transfection The product
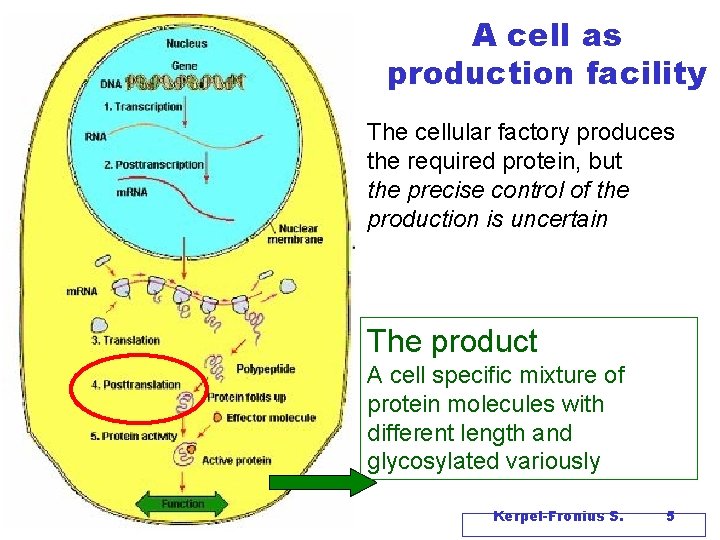
A cell as production facility The cellular factory produces the required protein, but the precise control of the production is uncertain The product A cell specific mixture of protein molecules with different length and glycosylated variously Kerpel-Fronius S. 5
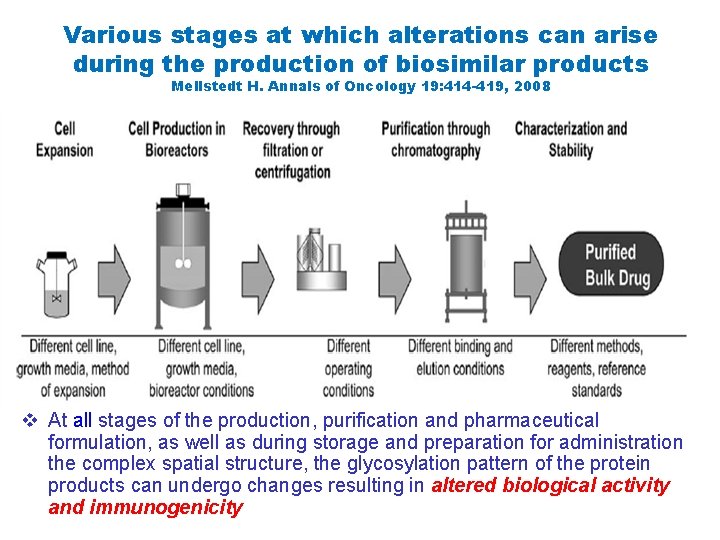
Various stages at which alterations can arise during the production of biosimilar products Mellstedt H. Annals of Oncology 19: 414 -419, 2008 v At all stages of the production, purification and pharmaceutical formulation, as well as during storage and preparation for administration the complex spatial structure, the glycosylation pattern of the protein products can undergo changes resulting in altered biological activity and immunogenicity

Biosimilar application (FDA) Sherman RE, Associate Director for Medical Policy Center of Drug Evaluation and Research Biologics Price Competition and Innovation (BPCI) Act Biosimilarity means v biological product is highly similar to the reference product notwithstanding minor differences in clinically inactive components v there are no clinically meaningful differences between the biological product and the reference product in terms of the safety, purity, and potency of the product. Specifications of biosimilar applications v biosimilarity to a reference product v same mechanism(s) of action for the proposed condition(s) of use–only to the extent known for the reference product v condition(s) of use proposed in labelling have been previously approved for the reference product v same route of administration, dosage form and strength as the reference product
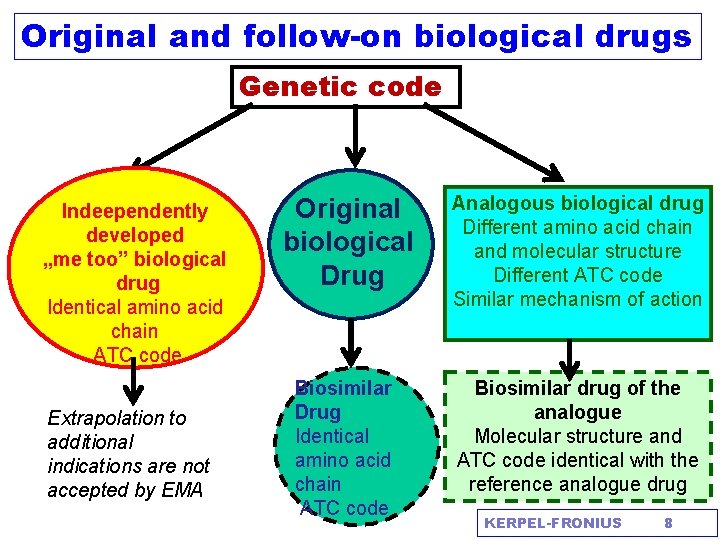
Original and follow-on biological drugs Genetic code Indeependently developed „me too” biological drug Identical amino acid chain ATC code Extrapolation to additional indications are not accepted by EMA Original biological Drug Analogous biological drug Different amino acid chain and molecular structure Different ATC code Similar mechanism of action Biosimilar Drug Identical amino acid chain ATC code Biosimilar drug of the analogue Molecular structure and ATC code identical with the reference analogue drug KERPEL-FRONIUS 8
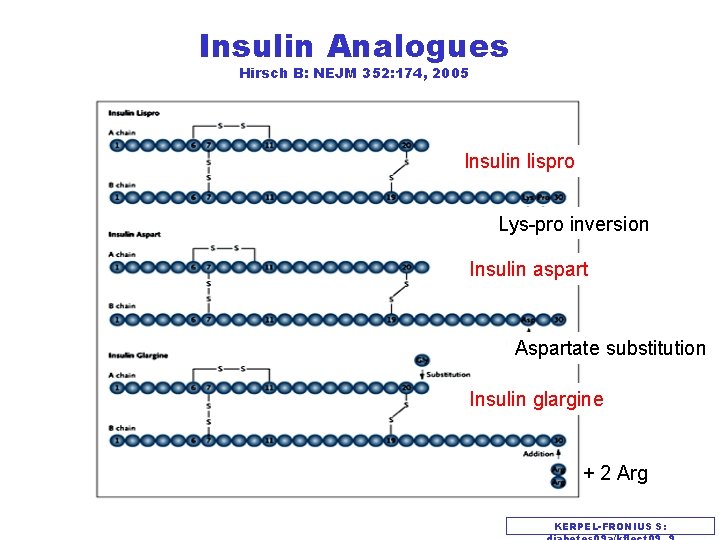
Insulin Analogues Hirsch B: NEJM 352: 174, 2005 Insulin lispro Lys-pro inversion Insulin aspart Aspartate substitution Insulin glargine + 2 Arg KERPEL-FRONIUS S:
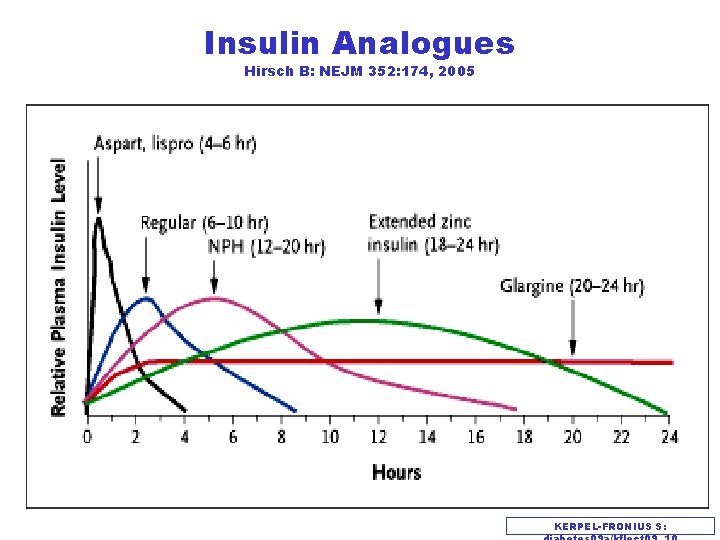
Insulin Analogues Hirsch B: NEJM 352: 174, 2005 KERPEL-FRONIUS S:
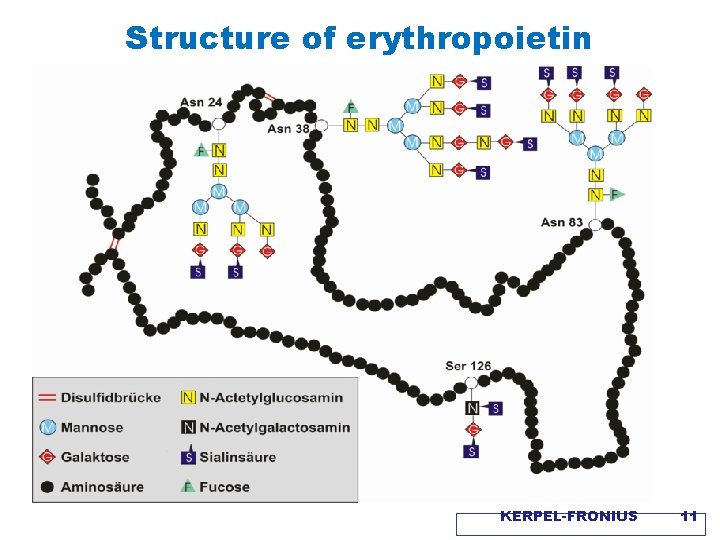
Structure of erythropoietin KERPEL-FRONIUS 11
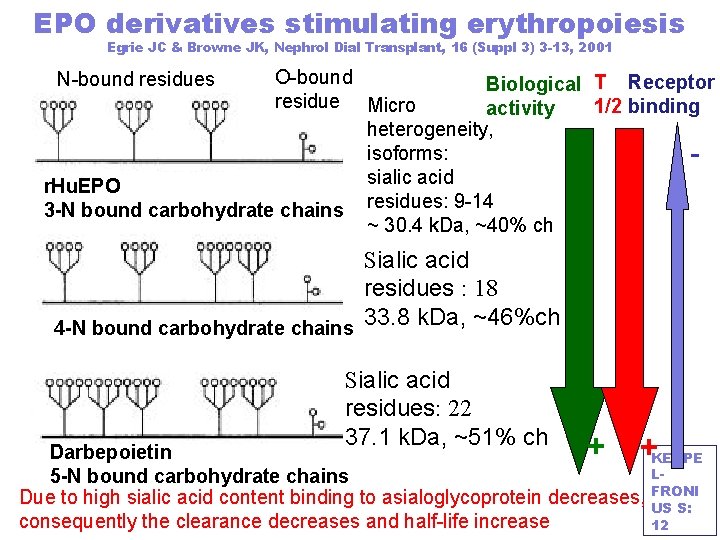
EPO derivatives stimulating erythropoiesis Egrie JC & Browne JK, Nephrol Dial Transplant, 16 (Suppl 3) 3 -13, 2001 O-bound Biological T Receptor residue Micro 1/2 binding activity heterogeneity, isoforms: sialic acid r. Hu. EPO 3 -N bound carbohydrate chains residues: 9 -14 ~ 30. 4 k. Da, ~40% ch N-bound residues - Sialic acid residues : 18 4 -N bound carbohydrate chains 33. 8 k. Da, ~46%ch Sialic acid residues: 22 37. 1 k. Da, ~51% ch + + Darbepoietin KERPE L 5 -N bound carbohydrate chains Due to high sialic acid content binding to asialoglycoprotein decreases, FRONI US S: consequently the clearance decreases and half-life increase 12
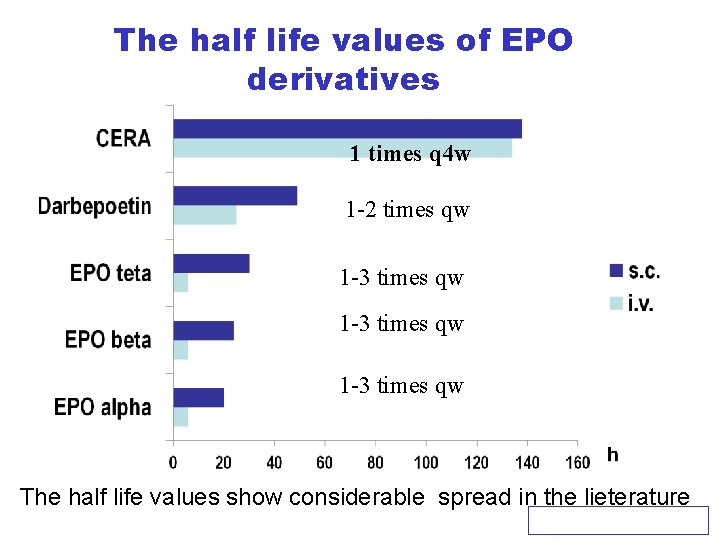
The half life values of EPO derivatives 1 times q 4 w 1 -2 times qw 1 -3 times qw The half life values show considerable spread in the lieterature KERPEL-FRONIUS S: 13
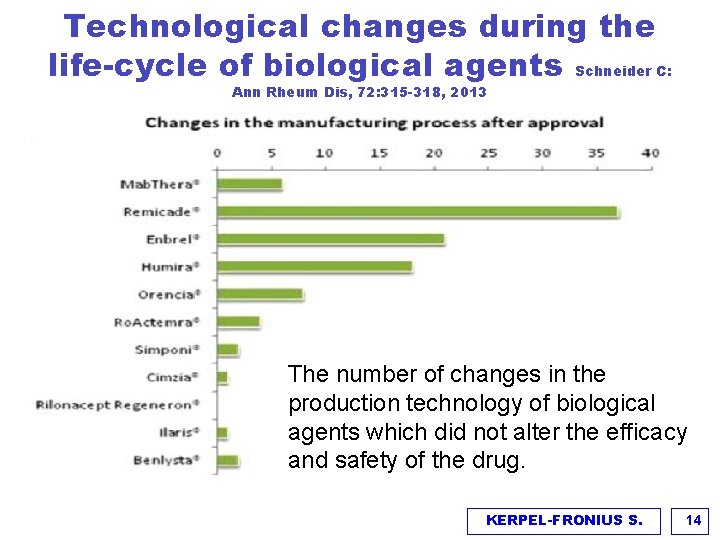
Technological changes during the life-cycle of biological agents Schneider C: Ann Rheum Dis, 72: 315 -318, 2013 The number of changes in the production technology of biological agents which did not alter the efficacy and safety of the drug. KERPEL-FRONIUS S. 14

Generic and Biosimilar medicinal products Guideline on similar biological medicinal products: CHMP/437/04 Innovative medicinal products Chemical medicinal products Biological medicinal products Follow-on medicinal products Generic medicinal products • Chemical identity can be proven • Comparative bioequivalence can be used to prove safety and efficacy • No therapeutic comparison is needed Biosimilar medicinal products • Full chemical identity at the molecular level cannot be proven • Comparability program using the reference drug is needed to prove similarity of quality, clinical efficacy and safety (immunogenicity!)
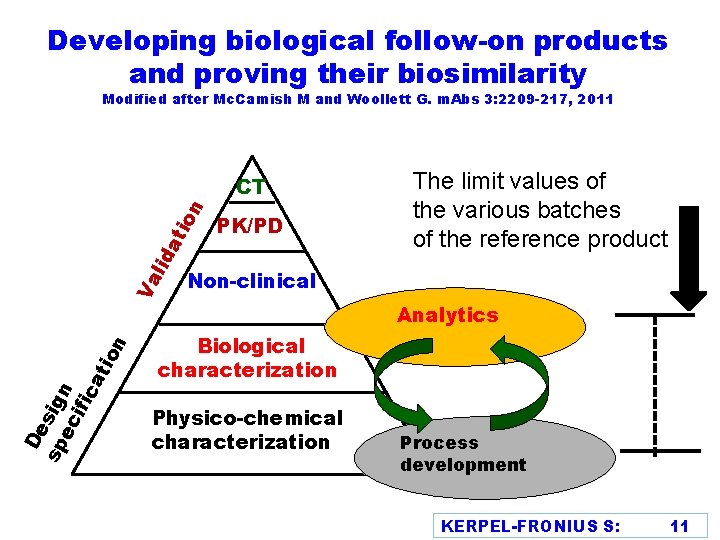
Developing biological follow-on products and proving their biosimilarity Modified after Mc. Camish M and Woollett G. m. Abs 3: 2209 -217, 2011 De sp sign ec ifi ca tio n Va lid at ion CT PK/PD The limit values of the various batches of the reference product Non-clinical Analytics Biological characterization Physico-chemical characterization Process development KERPEL-FRONIUS S: 11
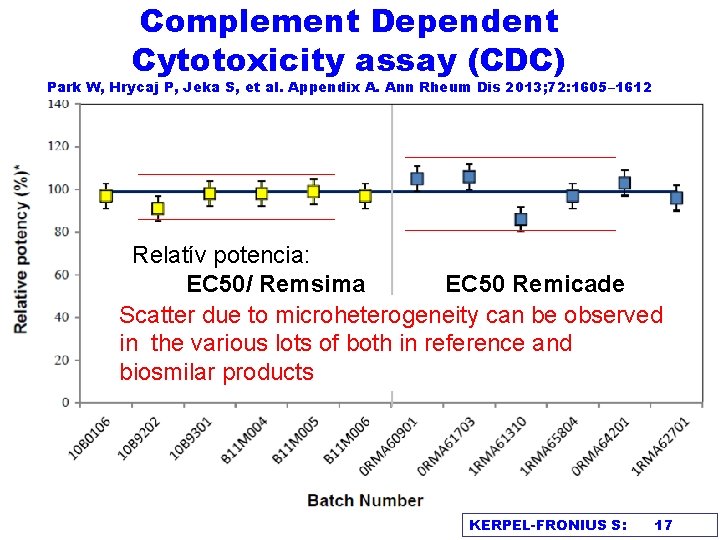
Complement Dependent Cytotoxicity assay (CDC) Park W, Hrycaj P, Jeka S, et al. Appendix A. Ann Rheum Dis 2013; 72: 1605– 1612 Relatív potencia: EC 50/ Remsima EC 50 Remicade Scatter due to microheterogeneity can be observed in the various lots of both in reference and biosmilar products KERPEL-FRONIUS S: 17

Steps for proving biosimilarity of follow-on m. Abs EMA guideline draft (EMA/CHMP/BMWP/403543/2010 Similarity v Identical amino acid composition v Similarity of physical-chemical measurements v Non-clinical comparative experiments Ø In vitro biological experiments Ø In vivo efficacy evaluation in animal models if feasible, comparative safety evaluation v Human comparative studies Ø Clinical pharmacodynamic studies Ø Clinical pharmacokinetic studies Ø Evaluation of dose: pharmacodynamic relationships Ø Therapeutic trials Identity KERPEL-FRONIUS S.

Similar biological medicinal products Clinical advantages and problems Proven identical and/or similar properties: • Identical amino acid composition • Similar pharmacokinetic properties • Similar pharmacologic effects Dissimilar properties: Biosimilar medicines • Post-translational modifications • Immunogenic properties • The clinical significance of the differences are uncertain KERPEL-FRONIUS S. 19
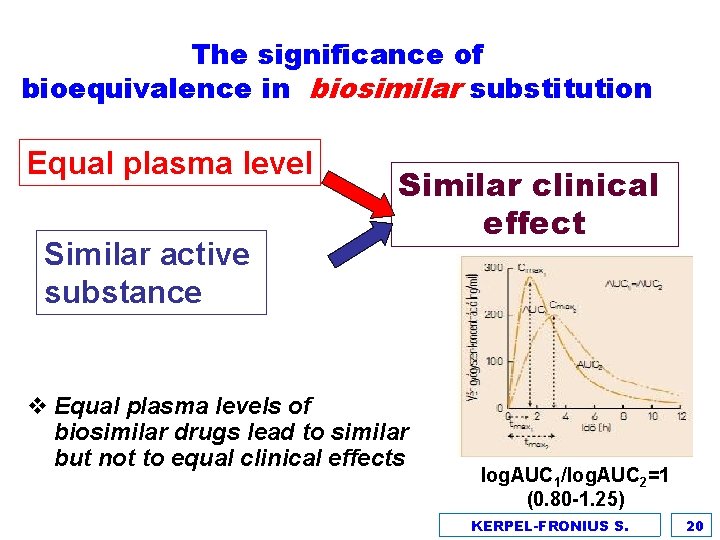
The significance of bioequivalence in biosimilar substitution Equal plasma level Similar active substance Similar clinical effect v Equal plasma levels of biosimilar drugs lead to similar but not to equal clinical effects log. AUC 1/log. AUC 2=1 (0. 80 -1. 25) KERPEL-FRONIUS S. 20
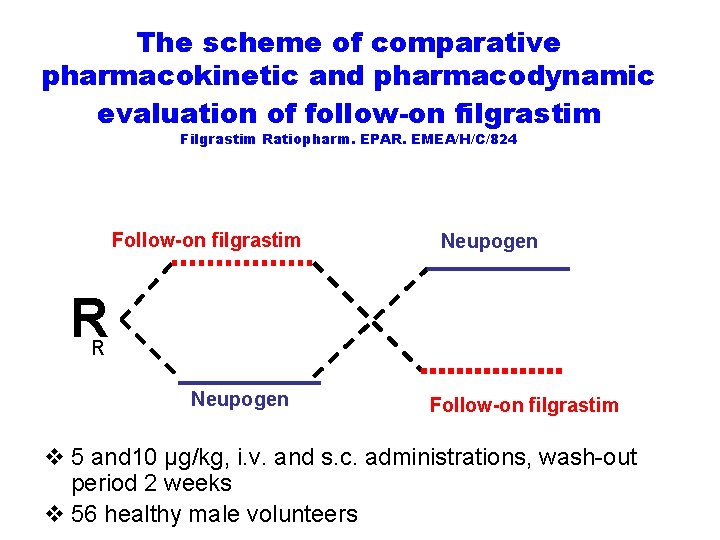
The scheme of comparative pharmacokinetic and pharmacodynamic evaluation of follow-on filgrastim Filgrastim Ratiopharm. EPAR. EMEA/H/C/824 Follow-on filgrastim Neupogen R R Neupogen Follow-on filgrastim v 5 and 10 μg/kg, i. v. and s. c. administrations, wash-out period 2 weeks v 56 healthy male volunteers
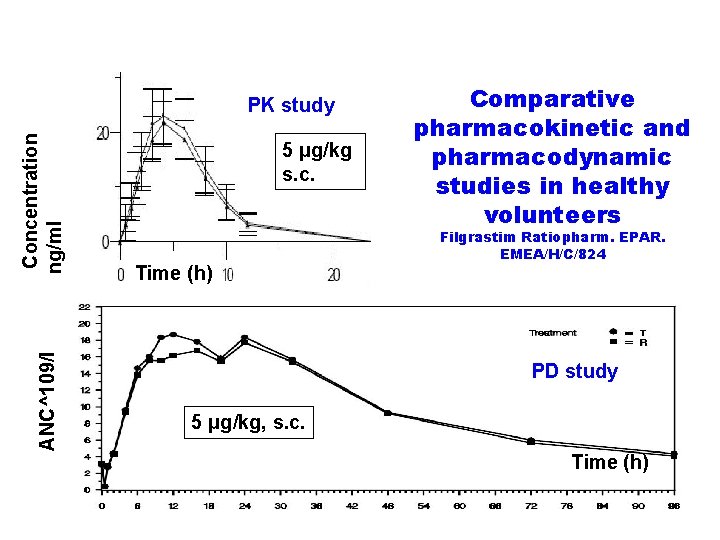
ANC^109/l Concentration ng/ml PK study 5 μg/kg s. c. Time (h) Comparative pharmacokinetic and pharmacodynamic studies in healthy volunteers Filgrastim Ratiopharm. EPAR. EMEA/H/C/824 PD study 5 μg/kg, s. c. Time (h)
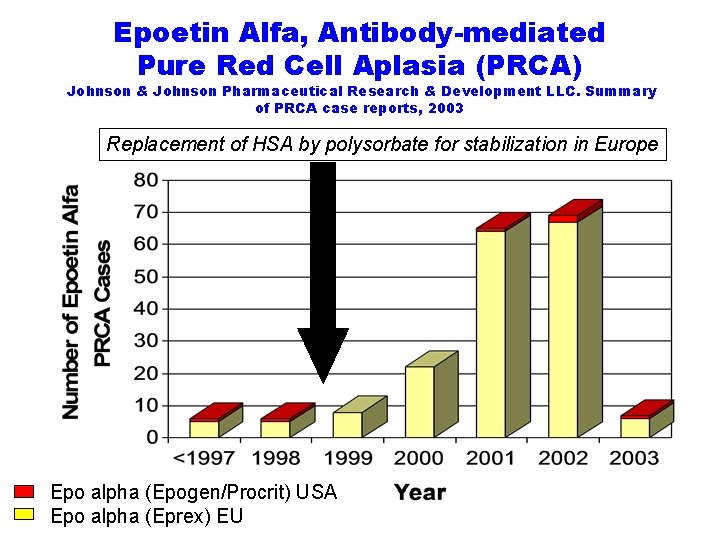
Epoetin Alfa, Antibody-mediated Pure Red Cell Aplasia (PRCA) Johnson & Johnson Pharmaceutical Research & Development LLC. Summary of PRCA case reports, 2003 Replacement of HSA by polysorbate for stabilization in Europe Epo alpha (Epogen/Procrit) USA Epo alpha (Eprex) EU
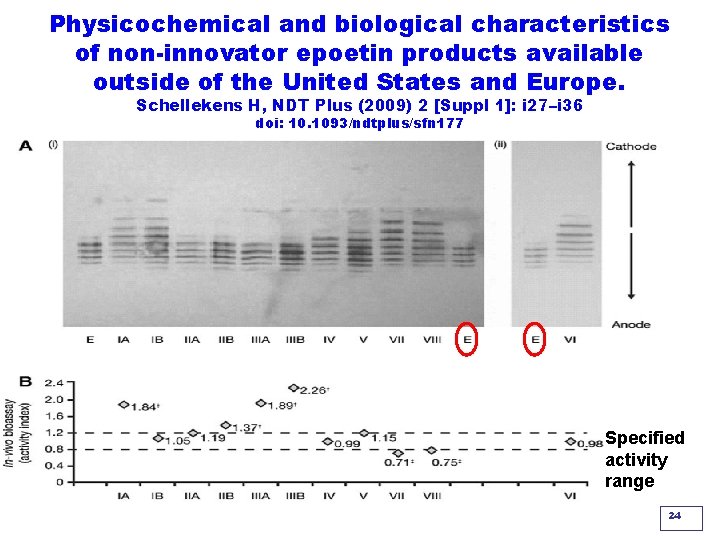
Physicochemical and biological characteristics of non-innovator epoetin products available outside of the United States and Europe. Schellekens H, NDT Plus (2009) 2 [Suppl 1]: i 27–i 36 doi: 10. 1093/ndtplus/sfn 177 Isoelectric focusing Western blot In vivo bioassay in mice Specified activity range 24
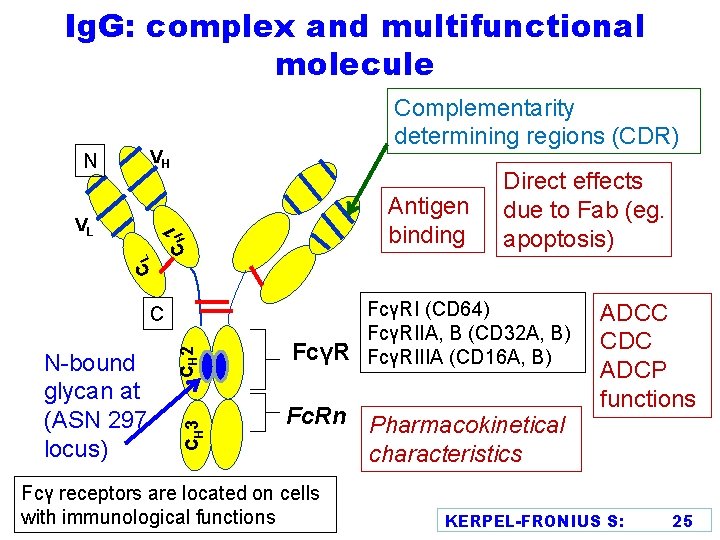
Ig. G: complex and multifunctional molecule Complementarity determining regions (CDR) VH CL 1 CH N VL Antigen binding CH 3 N-bound glycan at (ASN 297 locus) CH 2 C FcγR Direct effects due to Fab (eg. apoptosis) FcγRI (CD 64) FcγRIIA, B (CD 32 A, B) FcγRIIIA (CD 16 A, B) Fc. Rn Pharmacokinetical characteristics Fcγ receptors are located on cells with immunological functions ADCC CDC ADCP functions KERPEL-FRONIUS S: 25
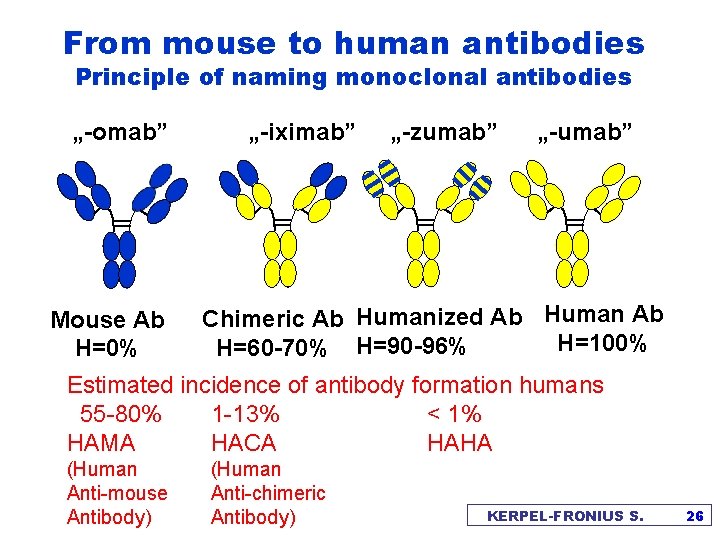
From mouse to human antibodies Principle of naming monoclonal antibodies „-omab” Mouse Ab H=0% „-iximab” „-zumab” „-umab” Chimeric Ab Humanized Ab Human Ab H=100% H=60 -70% H=90 -96% Estimated incidence of antibody formation humans 55 -80% 1 -13% < 1% HAMA HACA HAHA (Human Anti-mouse Antibody) (Human Anti-chimeric Antibody) KERPEL-FRONIUS S. 26
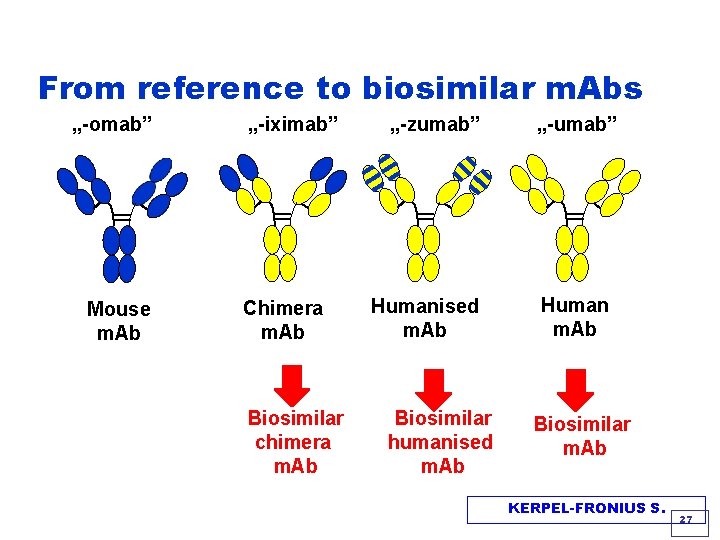
From reference to biosimilar m. Abs „-omab” Mouse m. Ab „-iximab” Chimera m. Ab Biosimilar chimera m. Ab „-zumab” Humanised m. Ab Biosimilar humanised m. Ab „-umab” Human m. Ab Biosimilar m. Ab KERPEL-FRONIUS S. 27
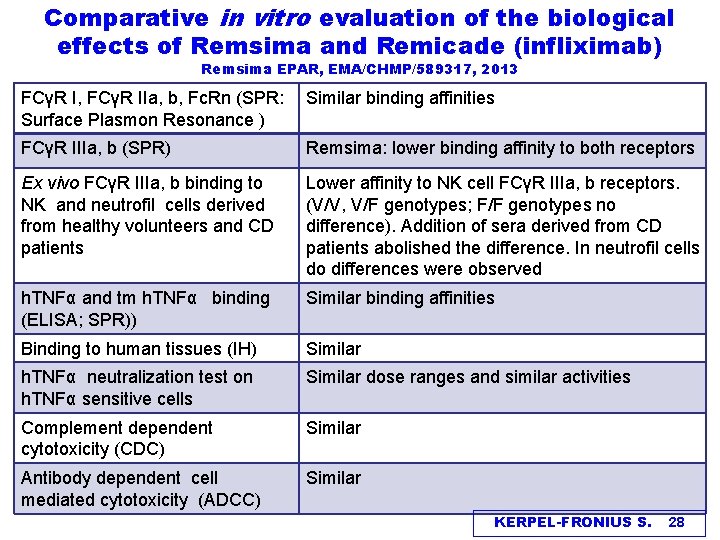
Comparative in vitro evaluation of the biological effects of Remsima and Remicade (infliximab) Remsima EPAR, EMA/CHMP/589317, 2013 FCγR I, FCγR IIa, b, Fc. Rn (SPR: Surface Plasmon Resonance ) Similar binding affinities FCγR IIIa, b (SPR) Remsima: lower binding affinity to both receptors Ex vivo FCγR IIIa, b binding to NK and neutrofil cells derived from healthy volunteers and CD patients Lower affinity to NK cell FCγR IIIa, b receptors. (V/V, V/F genotypes; F/F genotypes no difference). Addition of sera derived from CD patients abolished the difference. In neutrofil cells do differences were observed h. TNFα and tm h. TNFα binding (ELISA; SPR)) Similar binding affinities Binding to human tissues (IH) Similar h. TNFα neutralization test on h. TNFα sensitive cells Similar dose ranges and similar activities Complement dependent cytotoxicity (CDC) Similar Antibody dependent cell mediated cytotoxicity (ADCC) Similar KERPEL-FRONIUS S. 28
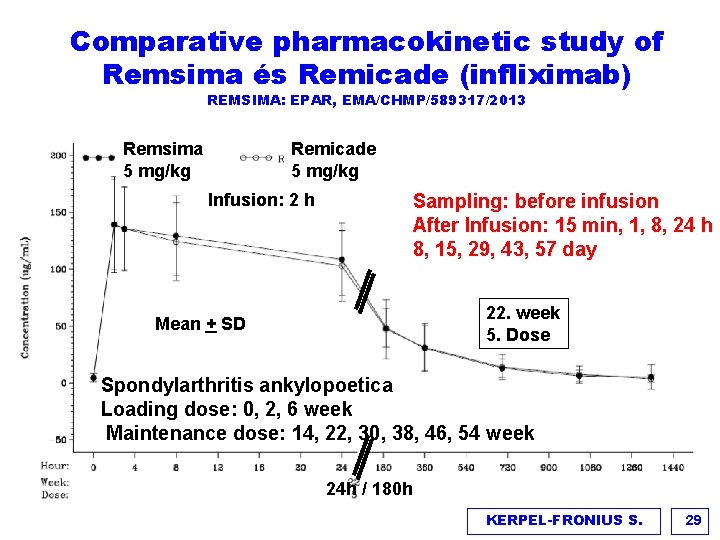
Comparative pharmacokinetic study of Remsima és Remicade (infliximab) REMSIMA: EPAR, EMA/CHMP/589317/2013 Remsima 5 mg/kg Remicade 5 mg/kg Infusion: 2 h Sampling: before infusion After Infusion: 15 min, 1, 8, 24 h 8, 15, 29, 43, 57 day 22. week 5. Dose Mean + SD Spondylarthritis ankylopoetica Loading dose: 0, 2, 6 week Maintenance dose: 14, 22, 30, 38, 46, 54 week 24 h / 180 h KERPEL-FRONIUS S. 29
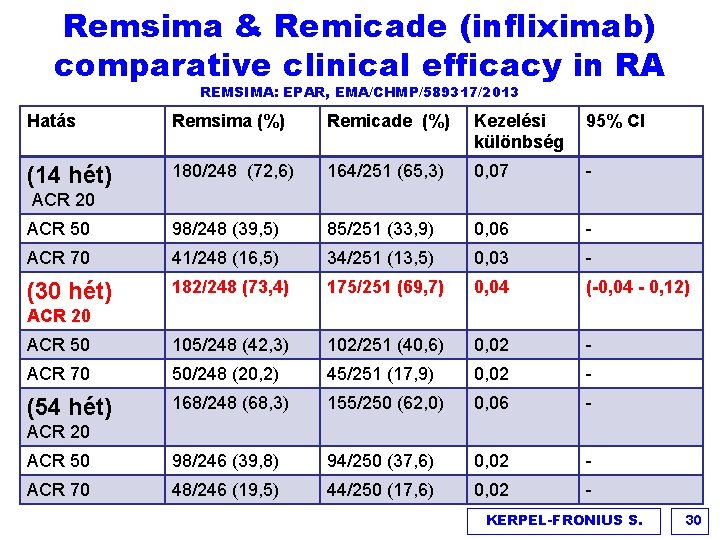
Remsima & Remicade (infliximab) comparative clinical efficacy in RA REMSIMA: EPAR, EMA/CHMP/589317/2013 Hatás Remsima (%) Remicade (%) Kezelési különbség 95% CI (14 hét) 180/248 (72, 6) 164/251 (65, 3) 0, 07 - ACR 50 98/248 (39, 5) 85/251 (33, 9) 0, 06 - ACR 70 41/248 (16, 5) 34/251 (13, 5) 0, 03 - (30 hét) 182/248 (73, 4) 175/251 (69, 7) 0, 04 (-0, 04 - 0, 12) ACR 50 105/248 (42, 3) 102/251 (40, 6) 0, 02 - ACR 70 50/248 (20, 2) 45/251 (17, 9) 0, 02 - (54 hét) 168/248 (68, 3) 155/250 (62, 0) 0, 06 - ACR 50 98/246 (39, 8) 94/250 (37, 6) 0, 02 - ACR 70 48/246 (19, 5) 44/250 (17, 6) 0, 02 - ACR 20 KERPEL-FRONIUS S. 30
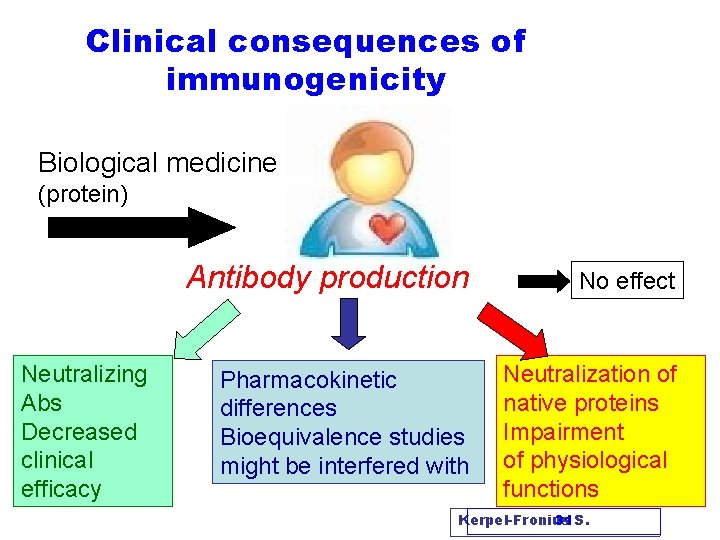
Clinical consequences of immunogenicity Biological medicine (protein) Neutralizing Abs Decreased clinical efficacy Antibody production No effect Pharmacokinetic differences Bioequivalence studies might be interfered with Neutralization of native proteins Impairment of physiological functions Kerpel-Fronius 31 S.

Antigenecity - immunogenicity v Antigenicity: is the ability to combine with the final products of the humoral and/or cell mediated immune response. It is not activating the immune response rather it combines with the final products of the immune response. v Immunogenicity: is antigenicity in an inflammatory milieu resulting in a successful humoral response. v All immunogens are antigenic but all antigens are not immunogenic KERPEL-FRONIUS S. 32
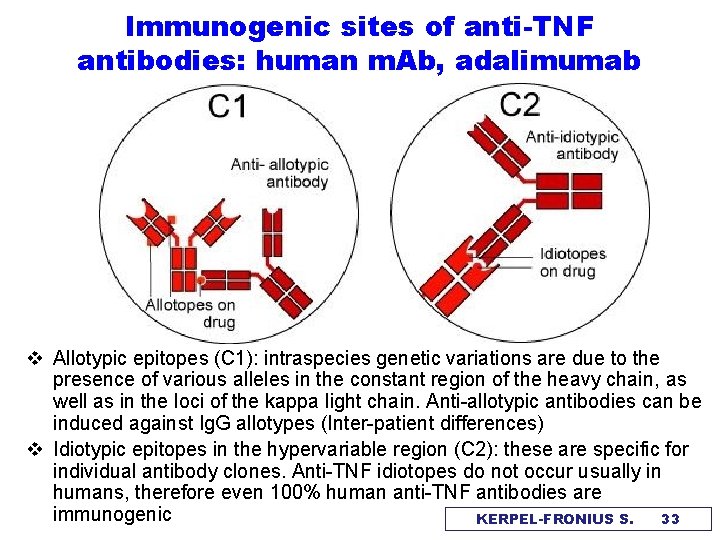
Immunogenic sites of anti-TNF antibodies: human m. Ab, adalimumab v Allotypic epitopes (C 1): intraspecies genetic variations are due to the presence of various alleles in the constant region of the heavy chain, as well as in the loci of the kappa light chain. Anti-allotypic antibodies can be induced against Ig. G allotypes (Inter-patient differences) v Idiotypic epitopes in the hypervariable region (C 2): these are specific for individual antibody clones. Anti-TNF idiotopes do not occur usually in humans, therefore even 100% human anti-TNF antibodies are immunogenic KERPEL-FRONIUS S. 33
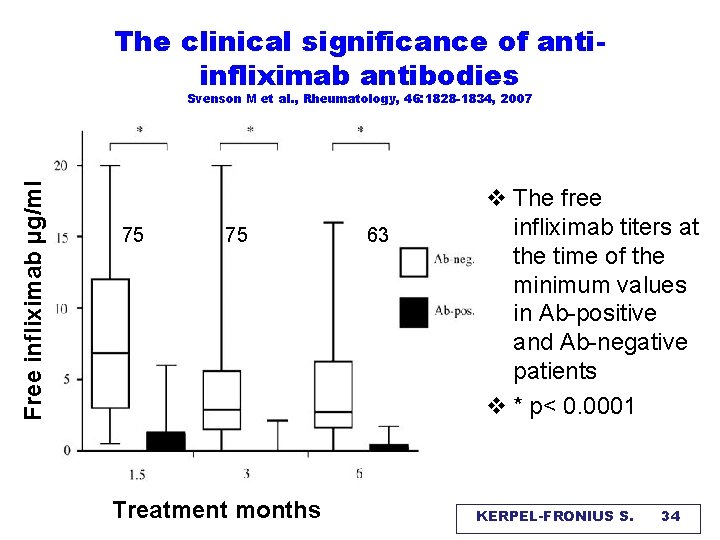
The clinical significance of antiinfliximab antibodies Free infliximab μg/ml Svenson M et al. , Rheumatology, 46: 1828 -1834, 2007 75 75 Treatment months 63 v The free infliximab titers at the time of the minimum values in Ab-positive and Ab-negative patients v * p< 0. 0001 KERPEL-FRONIUS S. 34
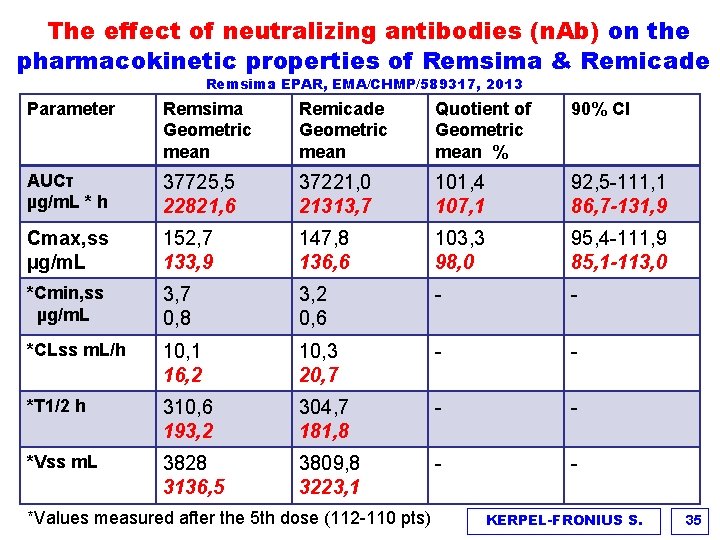
The effect of neutralizing antibodies (n. Ab) on the pharmacokinetic properties of Remsima & Remicade Remsima EPAR, EMA/CHMP/589317, 2013 Parameter Remsima Geometric mean Remicade Geometric mean Quotient of Geometric mean % 90% CI AUCτ µg/m. L * h 37725, 5 22821, 6 37221, 0 21313, 7 101, 4 107, 1 92, 5 -111, 1 86, 7 -131, 9 Cmax, ss µg/m. L 152, 7 133, 9 147, 8 136, 6 103, 3 98, 0 95, 4 -111, 9 85, 1 -113, 0 *Cmin, ss µg/m. L 3, 7 0, 8 3, 2 0, 6 - - *CLss m. L/h 10, 1 16, 2 10, 3 20, 7 - - *T 1/2 h 310, 6 193, 2 304, 7 181, 8 - - *Vss m. L 3828 3136, 5 3809, 8 3223, 1 - - *Values measured after the 5 th dose (112 -110 pts) KERPEL-FRONIUS S. 35
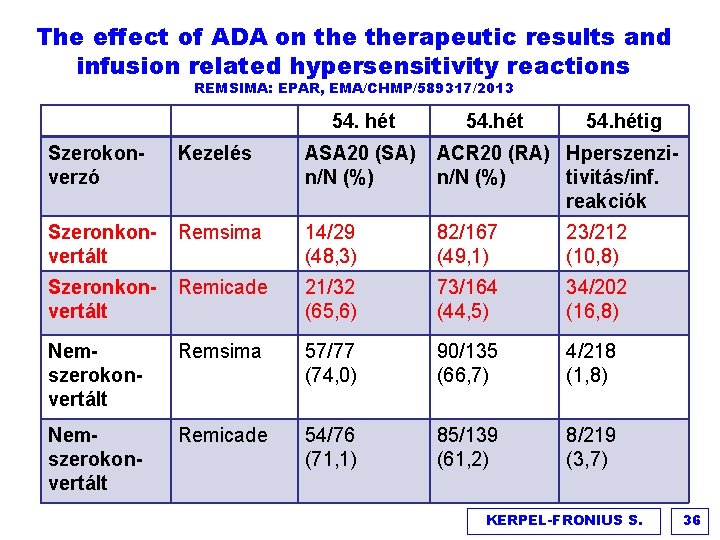
The effect of ADA on therapeutic results and infusion related hypersensitivity reactions REMSIMA: EPAR, EMA/CHMP/589317/2013 54. hétig Szerokonverzó Kezelés ASA 20 (SA) n/N (%) ACR 20 (RA) Hperszenzin/N (%) tivitás/inf. reakciók Szeronkonvertált Remsima 14/29 (48, 3) 82/167 (49, 1) 23/212 (10, 8) Szeronkonvertált Remicade 21/32 (65, 6) 73/164 (44, 5) 34/202 (16, 8) Nemszerokonvertált Remsima 57/77 (74, 0) 90/135 (66, 7) 4/218 (1, 8) Nemszerokonvertált Remicade 54/76 (71, 1) 85/139 (61, 2) 8/219 (3, 7) KERPEL-FRONIUS S. 36
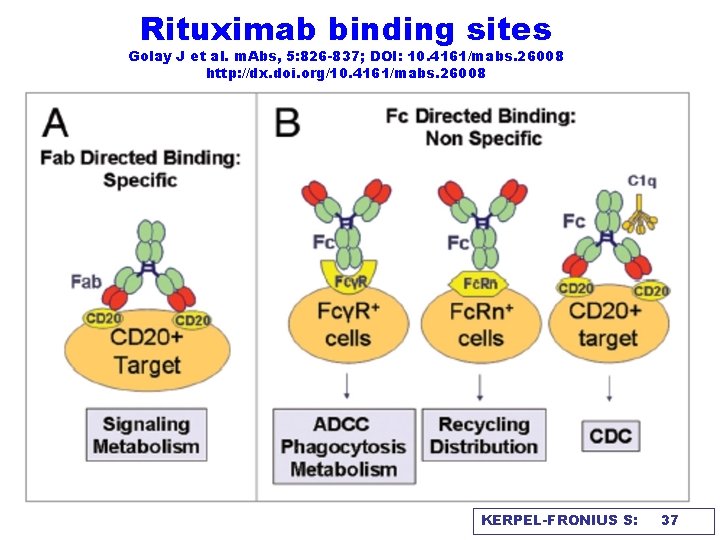
Rituximab binding sites Golay J et al. m. Abs, 5: 826 -837; DOI: 10. 4161/mabs. 26008 http: //dx. doi. org/10. 4161/mabs. 26008 KERPEL-FRONIUS S: 37
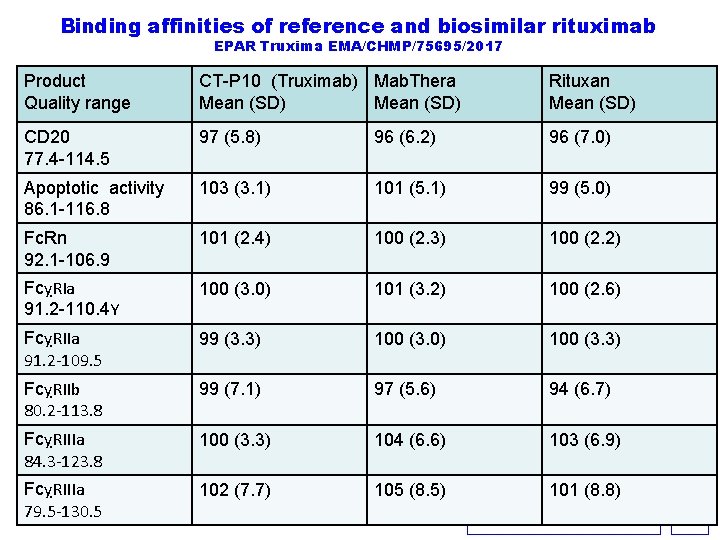
Binding affinities of reference and biosimilar rituximab EPAR Truxima EMA/CHMP/75695/2017 Product Quality range CT-P 10 (Truximab) Mab. Thera Mean (SD) Rituxan Mean (SD) CD 20 77. 4 -114. 5 97 (5. 8) 96 (6. 2) 96 (7. 0) Apoptotic activity 86. 1 -116. 8 103 (3. 1) 101 (5. 1) 99 (5. 0) Fc. Rn 92. 1 -106. 9 101 (2. 4) 100 (2. 3) 100 (2. 2) FcỵRIa 91. 2 -110. 4 Y 100 (3. 0) 101 (3. 2) 100 (2. 6) FcỵRIIa 91. 2 -109. 5 99 (3. 3) 100 (3. 0) 100 (3. 3) FcỵRIIb 80. 2 -113. 8 99 (7. 1) 97 (5. 6) 94 (6. 7) FcỵRIIIa 84. 3 -123. 8 100 (3. 3) 104 (6. 6) 103 (6. 9) FcỵRIIIa 79. 5 -130. 5 102 (7. 7) 105 (8. 5) 101 (8. 8) KERPEL-FRONIUS S. 38
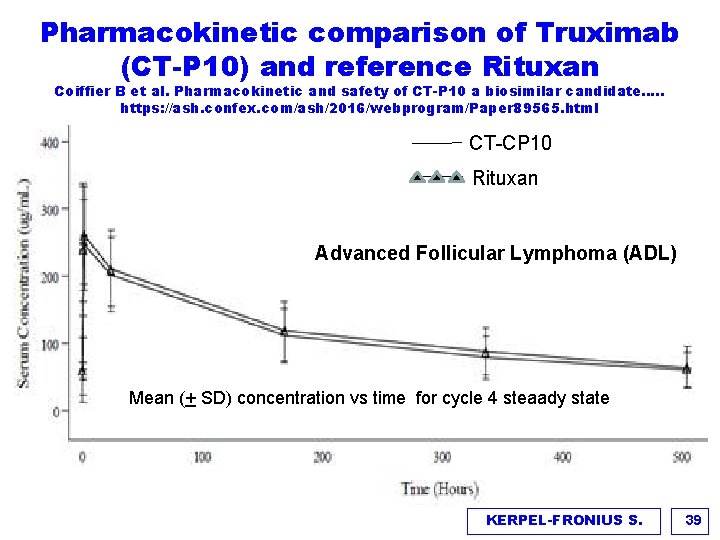
Pharmacokinetic comparison of Truximab (CT-P 10) and reference Rituxan Coiffier B et al. Pharmacokinetic and safety of CT-P 10 a biosimilar candidate…. . https: //ash. confex. com/ash/2016/webprogram/Paper 89565. html CT-CP 10 Rituxan Advanced Follicular Lymphoma (ADL) Mean (+ SD) concentration vs time for cycle 4 steaady state KERPEL-FRONIUS S. 39
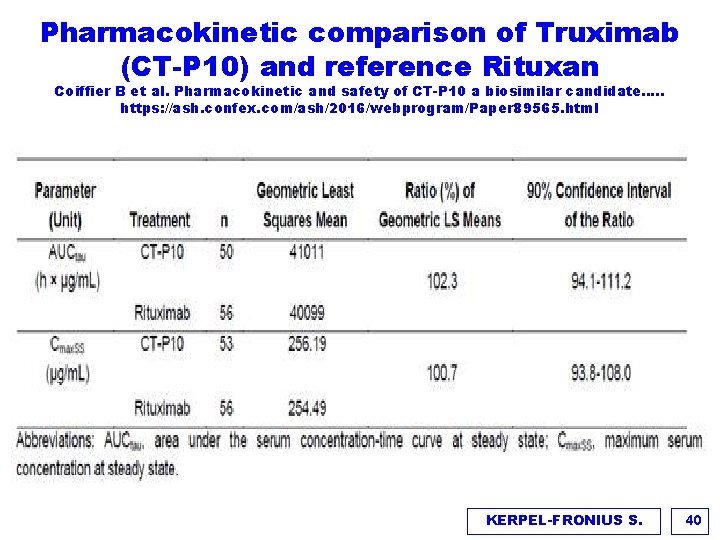
Pharmacokinetic comparison of Truximab (CT-P 10) and reference Rituxan Coiffier B et al. Pharmacokinetic and safety of CT-P 10 a biosimilar candidate…. . https: //ash. confex. com/ash/2016/webprogram/Paper 89565. html KERPEL-FRONIUS S. 40
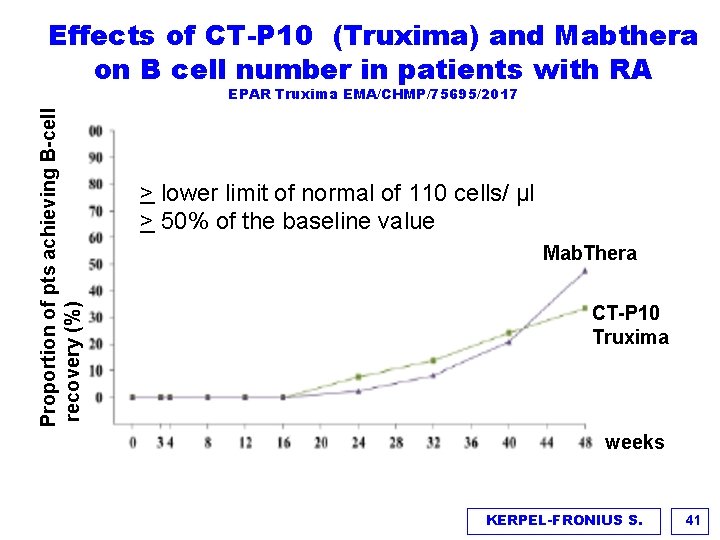
Effects of CT-P 10 (Truxima) and Mabthera on B cell number in patients with RA Proportion of pts achieving B-cell recovery (%) EPAR Truxima EMA/CHMP/75695/2017 > lower limit of normal of 110 cells/ µl > 50% of the baseline value Mab. Thera CT-P 10 Truxima weeks KERPEL-FRONIUS S. 41
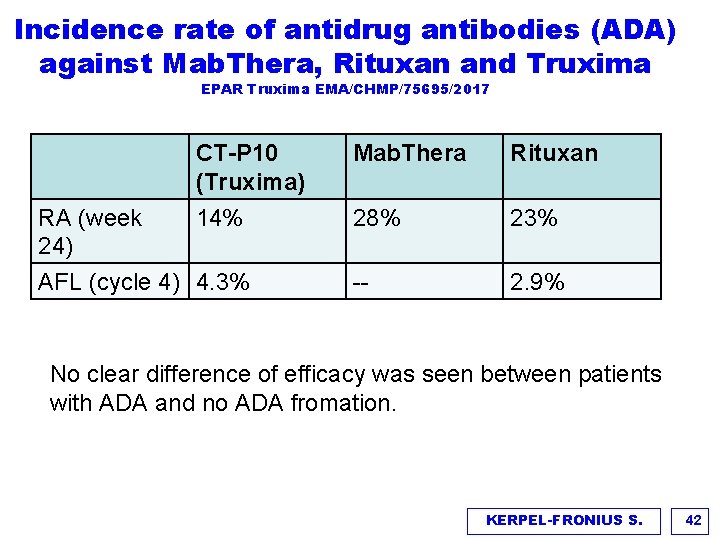
Incidence rate of antidrug antibodies (ADA) against Mab. Thera, Rituxan and Truxima EPAR Truxima EMA/CHMP/75695/2017 RA (week 24) CT-P 10 (Truxima) Mab. Thera Rituxan 14% 28% 23% -- 2. 9% AFL (cycle 4) 4. 3% No clear difference of efficacy was seen between patients with ADA and no ADA fromation. KERPEL-FRONIUS S. 42

The influence of human antibody binding on the pharmacokinetics and function of biological medicinal products v Various amounts of antibodies are produced against the different foreign proteins v The effect on pharmacokinetics usually depends on the binding affinity, the number of antigen sites the endogenous antibodies are directed against v Antibody binding usually increases elimination rate by uptake of the biological drugs into the RES system v In case of smaller proteins filtered by the kidney the binding of Abs might decrease the elimination rate (growth hormone) v The extent of formation of human antibodies against mouse (M), chimeric (C), humanized and fully human (H) Abs: Ø HAMA > HACA > HAHA KERPEL-FRONIUS S: 43

Medicinal products containing ESAs within the EU http: //www. emea. europa. eu/htms/human/epar/a. htm http: //www. whocc. no/atcddd/ Biosimilar medicinal products B 03 XA 01 - erythropoietin 1. Binocrit (epoetin alpha), Sandoz Gm. BH 2. Epoetin alpha-Hexal Biotech Forschungs Gm. BH 3. Abseamed (epoetin alpha), Medizin Arzneimittel Pütter Gm. BH & Co 4. Silapo (epoetin zeta), Stada 5. Retacrit (epoetin zeta), Hospira [Maine] v The 1 -3 alpha, and the 4 -5 zeta products are produced by the same manufacturer, i. e. they are identical products within the group, but marketed by different companies under different trade names Original analogue medicinal products v B 03 XA 01 - erythropoietin Ø Eprex (epoetin alfa), Janssen. Cilag (CHO) Ø Neorecormon (epoetin beta), Roche (CHO) Ø Dynepo (epoetin delta), Shire, (Human cell line) v B 03 XA 02 – darbepoetin alfa Ø Aranesp (Darbepoetin alfa), Amgen Europe, v B 03 XA 03 –peg-epoetin beta Ø Mircera (Methoxy polyethylene glycol-epoetin beta), Roche, Kerpel-Fronius S. : 44

How to differentiate between biosimilar products for pharmacovigilance FDA INN + Extension with 4 characters Alternatively: US code of 10 digits consisting of 3 parts: manufacturer, product, Box size EMEA WHO INN nomenclature The chemical-functional nomenclature cannot handle subtle structural variations resulting in immunogenicity differences In the SPC possible, but not yet demonstrated immunogenicity differences cannot be included Kerpel-Fronius S. : 45
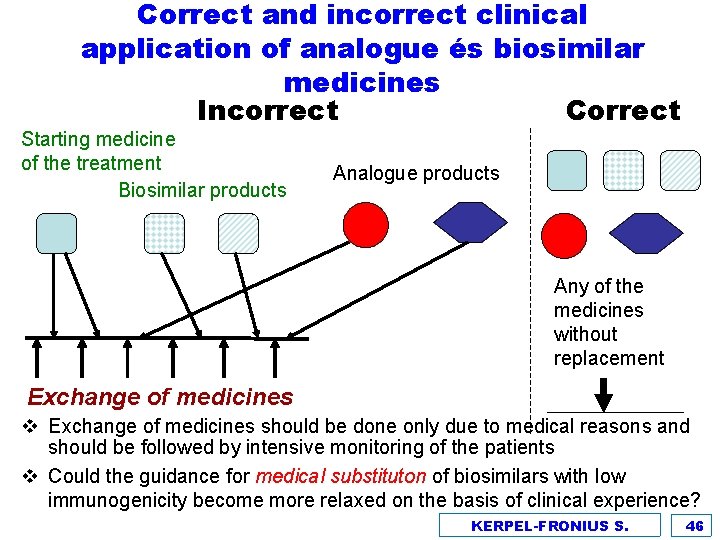
Correct and incorrect clinical application of analogue és biosimilar medicines Incorrect Correct Starting medicine of the treatment Biosimilar products Analogue products Any of the medicines without replacement Exchange of medicines v Exchange of medicines should be done only due to medical reasons and should be followed by intensive monitoring of the patients v Could the guidance for medical substituton of biosimilars with low immunogenicity become more relaxed on the basis of clinical experience? KERPEL-FRONIUS S. 46
- Slides: 46