Clinical and Translational Research 101 Informatics and Biostatistics

Clinical and Translational Research 101 Informatics and Biostatistics Support BRB 14 th Floor Faculty Lounge September 6, 2018, 10: 30 -10: 50 AM Justine Shults, Ph. D & Andrew J. Cucchiara, Ph. D ITMAT Center for Human Phenomic Science (CHPS) shultsj@email. chop. edu & andy@upenn. edu Research Electronic Data Capture (REDCap) redcap@mail. med. upenn. edu ITMAT Study Design and Biostatistics Core (SDAB) sdab@mail. med. upenn. edu UPenn’s Office of Software Licensing https: //secure. www. upenn. edu/business-services/softwarelicenses

Institute for Translational Medicine And Therapeutics Become an ITMAT member: www. ITMAT. upenn. edu

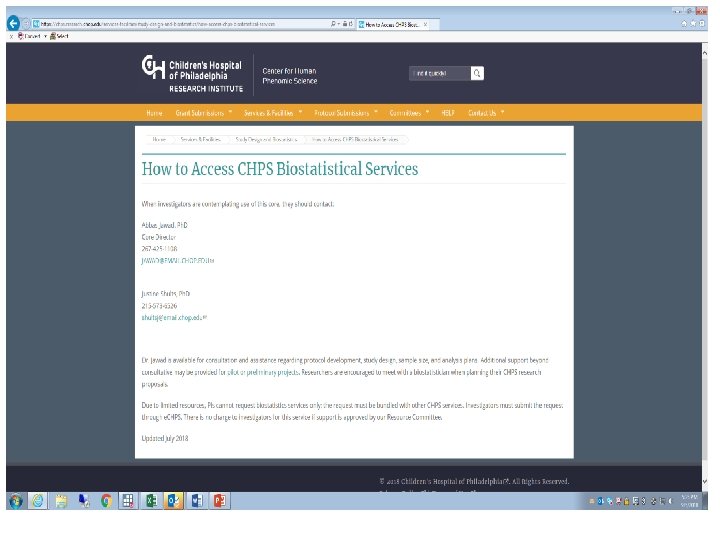
Center for Human Phenomic Science (CHPS)

ITMAT Funding Opportunities Available at www. itmat. upenn. edu/funding. shtml Sharing Partnership for Innovative Research in Translation (SPIRi. T) Program

CHPS Junior Investigator Pilot Grant (JIPG)

Research Electronic Data Capture (REDCap) https: //redcap. med. upenn. edu

Requirements for Informatics Systems • Provide Adequate Capacity • Maintain Secure Storage • Allow Appropriate Access to Data • Generate Case Report Forms • Develop Good Data Structures • Allow Export to Other Applications • Provide Access to Technical Support • Easy to Apply for REDCap Account • Reasonably priced (or free? ) access

Advantages of the REDCap System (Research Electronic Data Capture): • • • Free of Cost Availability - REDCap software has no license fees Web-based - build a project and input data from anywhere Secure – authenticated web access to data with audit trails of access Fast and flexible - project conception to production within a day Multi-site access - concurrent access to project from many sites Autonomy - researcher controls user access, completely Exportability - for analysis using SAS, Stata, SPSS, S-Plus, R et al. Customizable - investigators have control of project development Collaborative –libraries of modifiable data dictionaries Local and Developer Support – REDCap Technical Support Team and Monthly User Group Meetings – List of Frequently Asked Questions – Local Support access to REDCap developers via weekly meetings • Advanced features include … – – – Mid-study database modification Branching logic {if ever smoked then for when and amount per day} Calculated fields {e. g. body mass index from height & weight} Automatic validation (range check/data integrity) in real time Double data entry for verification Mobile interface for smart phones and tablet computers
![REDCap Documentation • Training Resources for REDCap: § Frequently Asked Questions [Help/FAQ] § REDCap REDCap Documentation • Training Resources for REDCap: § Frequently Asked Questions [Help/FAQ] § REDCap](http://slidetodoc.com/presentation_image_h2/60c5b3501e5c240b858c8a23b5816008/image-9.jpg)
REDCap Documentation • Training Resources for REDCap: § Frequently Asked Questions [Help/FAQ] § REDCap Video [Training Resources] § Coursera Data Mngmt for Clinical Research § Joint CHOP/UPenn REDCap Users’ Group: Ø Ø Second Tuesday of even numbered months or Second Wednesday of odd numbered months Meetings at Roberts Ctr or Colket TRB Meeting alerts e-mailed a week before

ITMAT’s CHPS Informatics Support • Assistance with Research Electronic Data Capture to – Design new REDCap data dictionary structures – Access existing REDCap data structure libraries – Validate and Verify REDCap data entries – Assign REDCap access privileges to collaborators – Import data into REDCap created by other systems – Export REDCap data to various software packages – Provide guidance in the use of Statistical Software – Facilitate use of graphical display software • Assistance with Access to the Penn Data Store – By CHPS Facilitator Services: • Michael R Rickels, MD, MS, Director • rickels@mail. med. upenn. edu • Carl Shaw, Med, MBA, CIP • shawcarl@upenn. edu

REDCap Security Answers • Was designed to be HIPAA compliant • Is Title 21 CFR, Part 11 capable, but … • Is NOT Title 21 CFR, Part 11 compliant • Consortium Title 21 CFR, Part 11 WIKI • Title 21 CFR, Part 11 Working Group 11

REDCap Common Misconceptions: • • • Have Penn. Key Have REDCap? REDCap server to all at Vanderbilt? REDCap servers are interconnected? One account for team is good idea? Support 24/7/365? (No, it’s 9 -5 M-F) Support will create projects for you? Faster response to personal e-mail? Access to one project access to all? Multicenters have access to all data?

Roles for Biostatistics in Study Design • • Estimate Adequate Sample Size Implement Appropriate Control Group Develop an Optimal Analysis Plan Produce Complementary Graphs Help Communicate Results, Clearly Reduce Time to Approval/Publication Generate a Realistic Budget Review Drafts of Proposal/Manuscript . . . so, how does one obtain biostatistical support?

Study Design and Biostatistics (SDAB) sdab@mail. med. upenn. edu

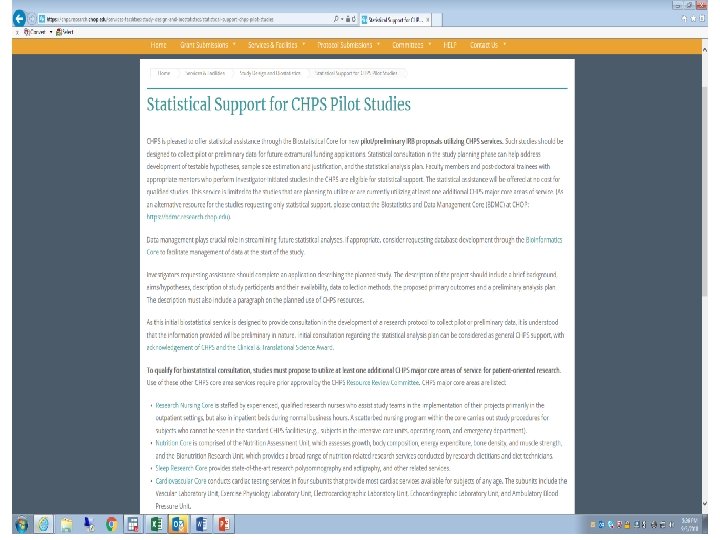
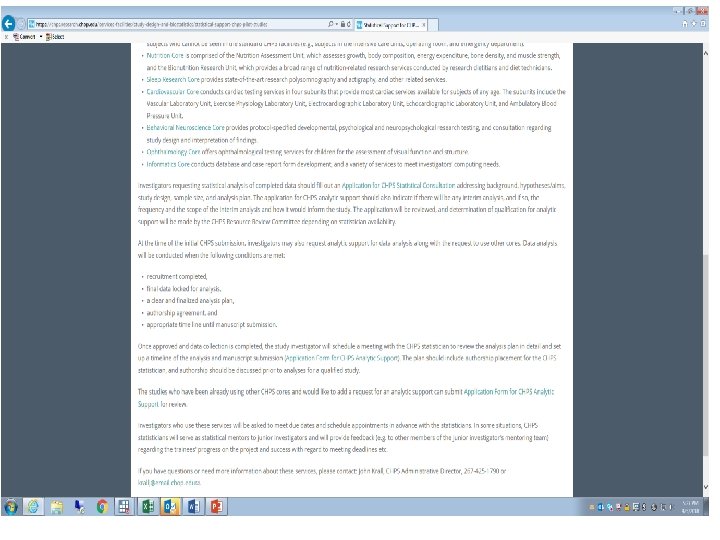
CHPS Biostatistical Support • Obtain Guidance or Assistance from – Abbas Jawad (CHOP) – Andy Cucchiara (HUP & PPMC) • • with designing your experiment in estimating Sample Size or Power to develop Statistical Analysis Plan for production of Statistical Results with Interpretation of Results in Generating Presentation Graphics for Reviewing Drafts of Manuscript
UPenn Supported Software
UPenn Site Licensing
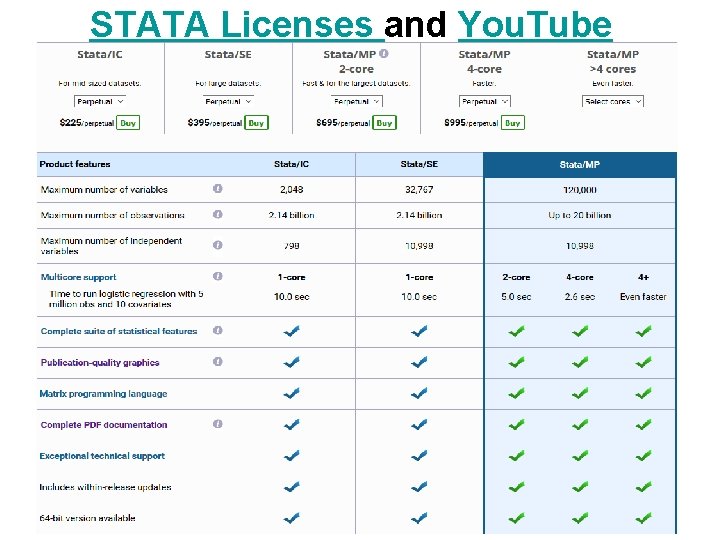
STATA Licenses and You. Tube
Free Last. Pass Premium License

Do spreadsheets help you Excel? • • Yes for Financial Accountants No for Analysis (IBM “White Paper”) No for Presentation Graphics No for Data Management Yes to develop Time & Events Chart Powerpoint for presenting Schema Which Statistical Package to use? – SAS or JMP – IBM’s SPSS – STATA - R (Microsoft) - Minitab - Mat. Lab

Take Away Messages: • • • Slides and Video will be Available Be Sure to Sign Attendance Sheet Do Apply for ITMAT Membership Request a REDCap Account, ASAP Ask for a Tour of CHPS Sites – at Dulles, Perelman, Presby or CHOP • • Tell us about your research idea Describe your database issues Statistical questions are welcomed Request a Last. Pass Premium license

Tools for Planning Research Assessments / Interventions Longitudinal Progression of Trial Time and Events Chart
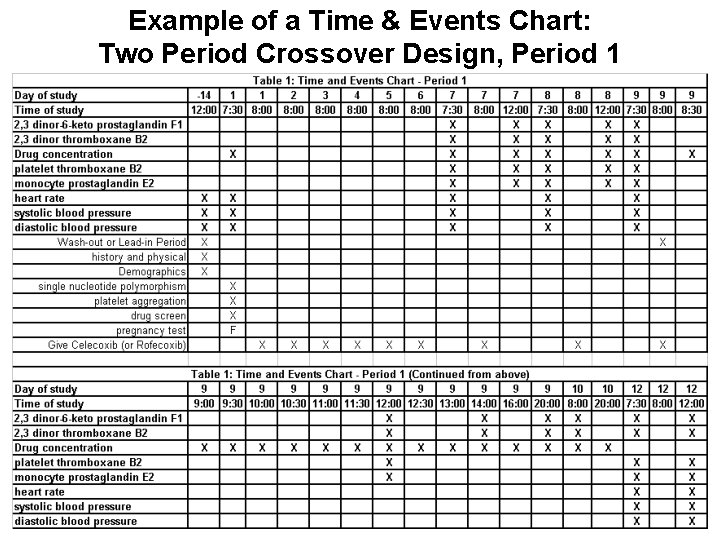
Example of a Time & Events Chart: Two Period Crossover Design, Period 1

Why develop Time & Events Chart? • Time and Events Charts can help you… – – – – – Prepare for In-Service Presentation to Facilitators Develop Case Report and Informed Consent Forms Screen for Inclusion/Exclusion Criteria Guidance for (CHPS? ) Support Staff and Facilitators Facilitator Checklist for Processing Study Subjects Design your REDCap Project Database Dictionary Demonstrate Study Feasibility to Reviewers/Funders Provide Guidance for Statistical Analyst and Analysis Select Literature Review and Background Material Generate and Complement Study Design Narrative
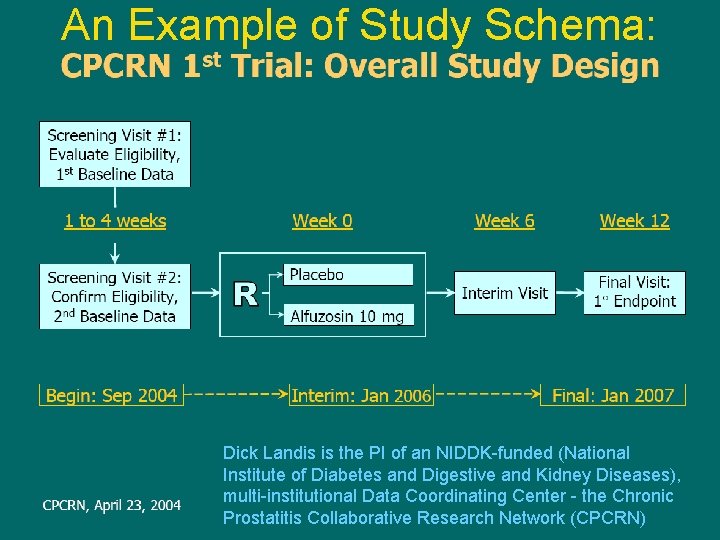
An Example of Study Schema: Dick Landis is the PI of an NIDDK-funded (National Institute of Diabetes and Digestive and Kidney Diseases), multi-institutional Data Coordinating Center - the Chronic Prostatitis Collaborative Research Network (CPCRN)
- Slides: 28