ClingE coli Bacteria on target Harvard i GEM

Cling-E. coli : Bacteria on target Harvard i. GEM 2007 Ellenor Brown Stephanie Lo Alex Pickett Sammy Sambu Kevin Shee Perry Tsai Shaunak Vankudre George Xu

The motivation To develop a system for targeting bacteria to a specific substrate and effecting a cellular response Introduction Harvard i. GEM 2007
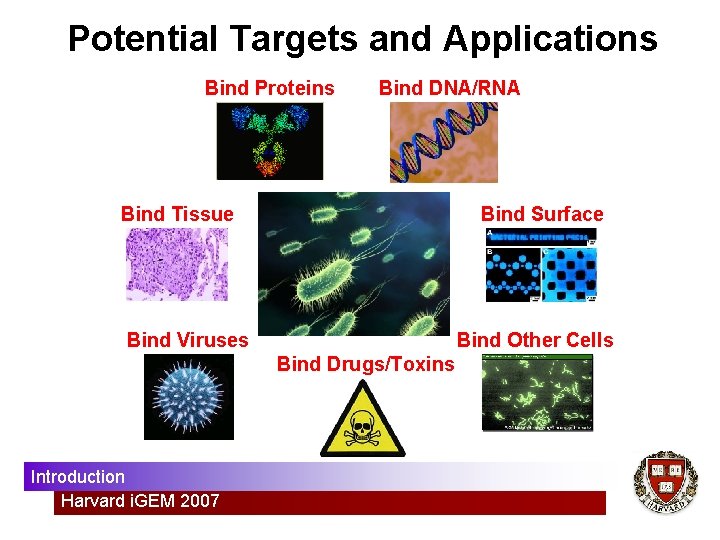
Potential Targets and Applications Bind Proteins Bind DNA/RNA Bind Tissue Bind Surface Bind Viruses Bind Other Cells Bind Drugs/Toxins Introduction Harvard i. GEM 2007
Bacterial targeting Quorum-sensing Introduction Harvard i. GEM 2007 Fec signal transduction
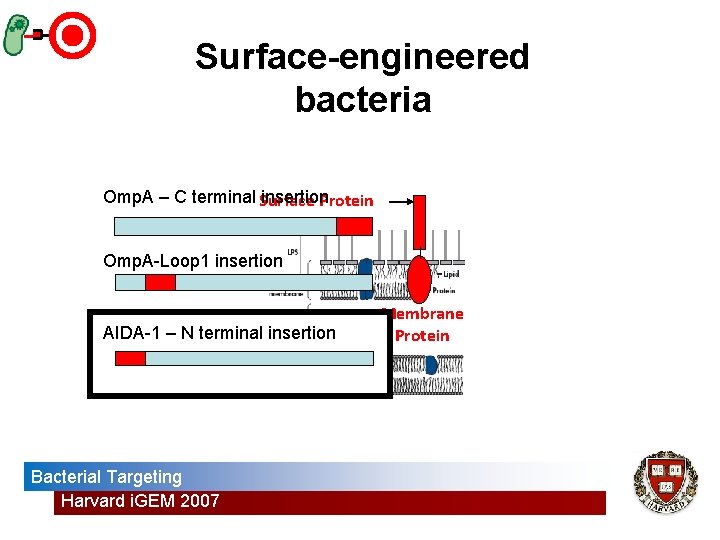
Surface-engineered bacteria Omp. A – C terminal Surface insertion Protein Omp. A-Loop 1 insertion AIDA-1 – N terminal insertion Bacterial Targeting Harvard i. GEM 2007 Membrane Protein

Selecting/enriching for surface-engineered bacteria • Tags – Histidine tag + nickel – Strep 2 tag + streptavidin • Assay • Magnetic Activated Cell Sorting (MACS) Bacterial Targeting Harvard i. GEM 2007 Magnetic Bead Assays
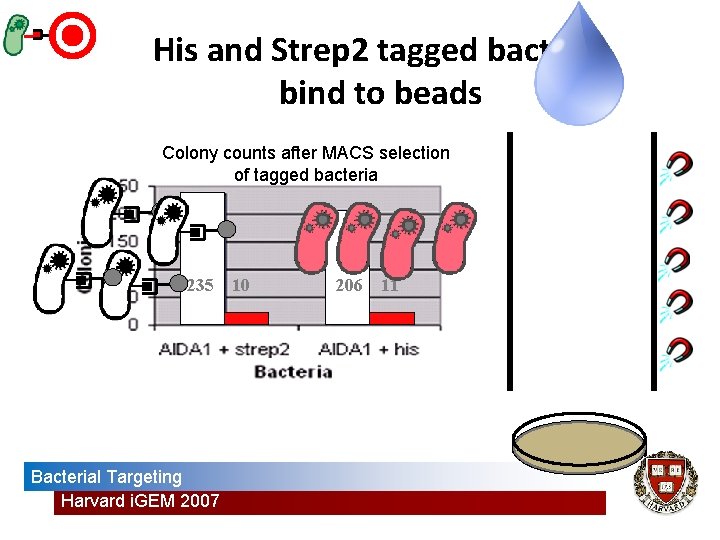
His and Strep 2 tagged bacteria bind to beads Colony counts after MACS selection of tagged bacteria 235 Bacterial Targeting Harvard i. GEM 2007 10 206 11

Results • Surface-expression vehicle – AIDA 1 • Engineered surface-displayed histidine tag and strep 2 tag • Demonstrated bacterial targeting to nickel and streptavidin beads through MACS Bacterial Targeting Harvard i. GEM 2007
Bacterial targeting Quorum-sensing Quorum Sensing Harvard i. GEM 2007 Fec signal transduction
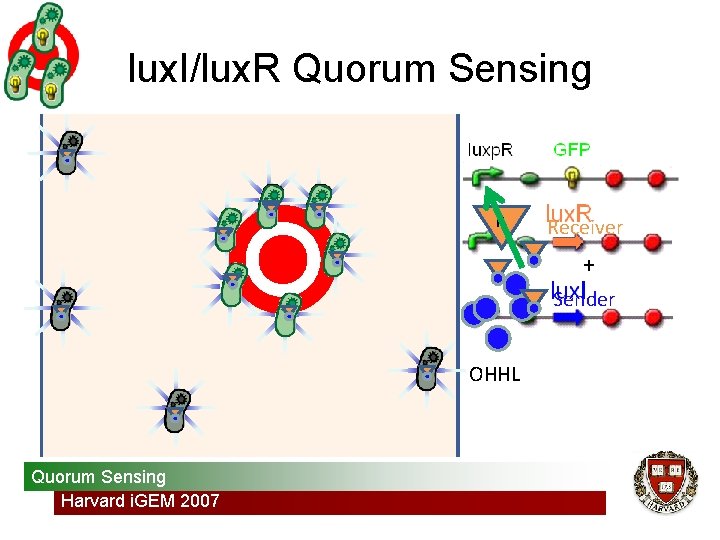
lux. I/lux. R Quorum Sensing R Receiver + Sender OHHL Quorum Sensing Harvard i. GEM 2007
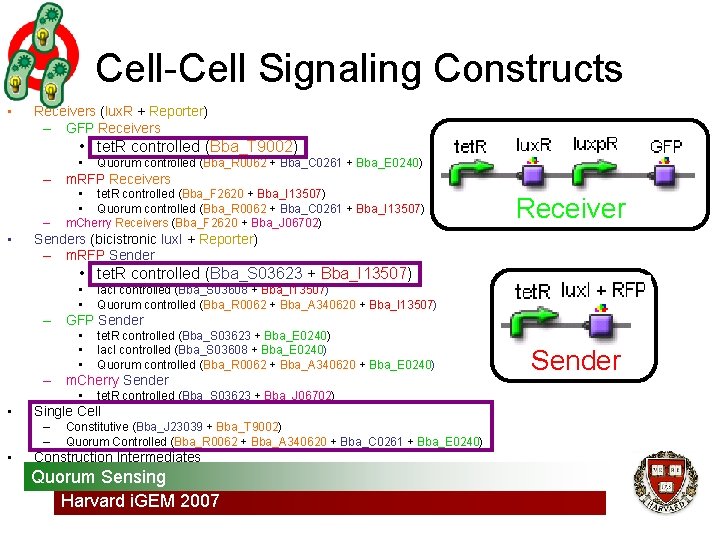
Cell-Cell Signaling Constructs • Receivers (lux. R + Reporter) – GFP Receivers • tet. R controlled (Bba_T 9002) • Quorum controlled (Bba_R 0062 + Bba_C 0261 + Bba_E 0240) – m. RFP Receivers – • • tet. R controlled (Bba_F 2620 + Bba_I 13507) • Quorum controlled (Bba_R 0062 + Bba_C 0261 + Bba_I 13507) m. Cherry Receivers (Bba_F 2620 + Bba_J 06702) Receiver Senders (bicistronic lux. I + Reporter) – m. RFP Sender • tet. R controlled (Bba_S 03623 + Bba_I 13507) • • lac. I controlled (Bba_S 03608 + Bba_I 13507) Quorum controlled (Bba_R 0062 + Bba_A 340620 + Bba_I 13507) – GFP Sender • • • tet. R controlled (Bba_S 03623 + Bba_E 0240) lac. I controlled (Bba_S 03608 + Bba_E 0240) Quorum controlled (Bba_R 0062 + Bba_A 340620 + Bba_E 0240) – m. Cherry Sender • • Single Cell – – • tet. R controlled (Bba_S 03623 + Bba_J 06702) Constitutive (Bba_J 23039 + Bba_T 9002) Quorum Controlled (Bba_R 0062 + Bba_A 340620 + Bba_C 0261 + Bba_E 0240) Construction Intermediates Quorum Sensing Harvard i. GEM 2007 Sender
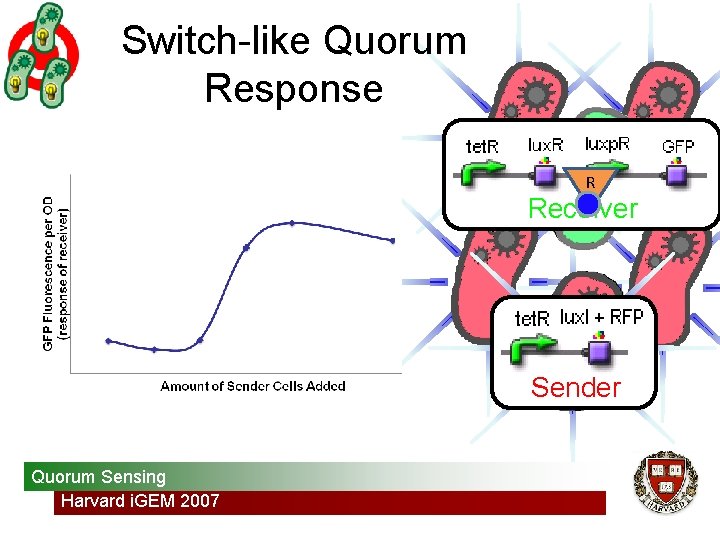
Switch-like Quorum Response R Receiver Sender Quorum Sensing Harvard i. GEM 2007
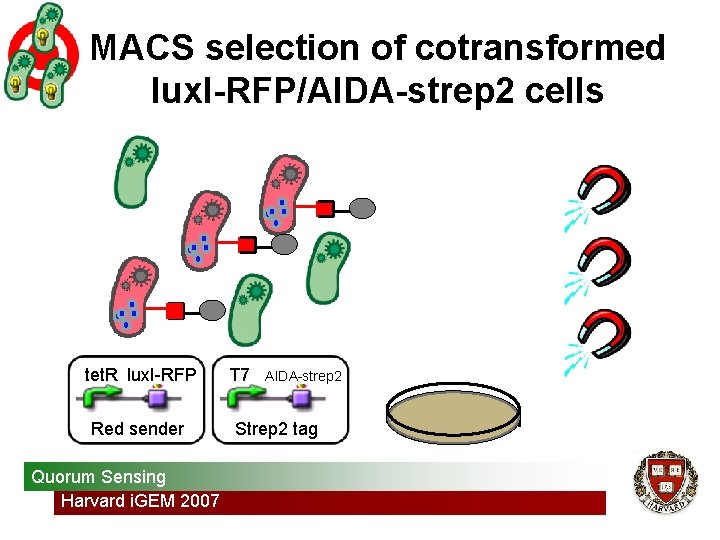
MACS selection of cotransformed lux. I-RFP/AIDA-strep 2 cells tet. R lux. I-RFP Red sender Quorum Sensing Harvard i. GEM 2007 T 7 AIDA-strep 2 Strep 2 tag
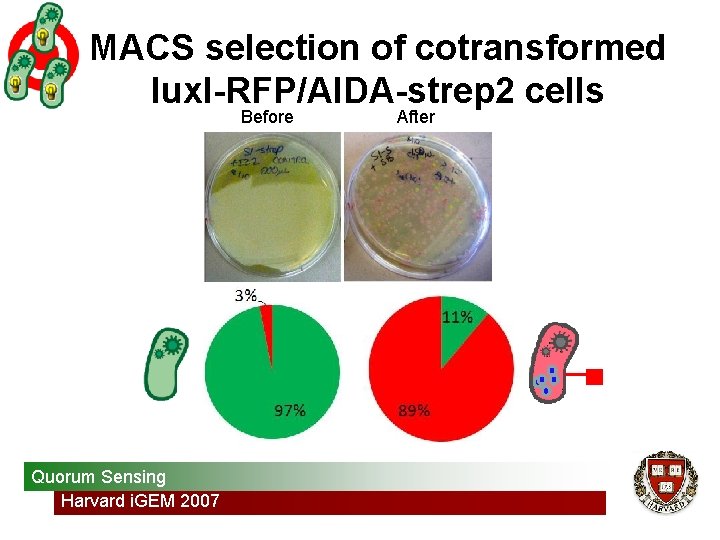
MACS selection of cotransformed lux. I-RFP/AIDA-strep 2 cells Before Quorum Sensing Harvard i. GEM 2007 After
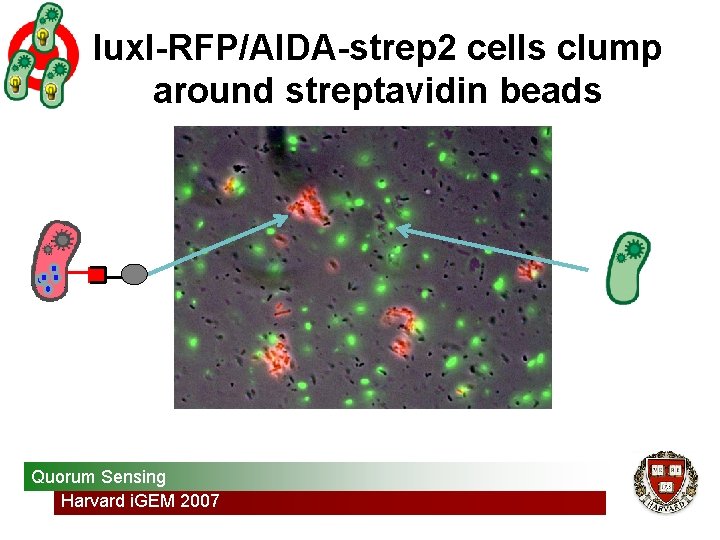
lux. I-RFP/AIDA-strep 2 cells clump around streptavidin beads Quorum Sensing Harvard i. GEM 2007
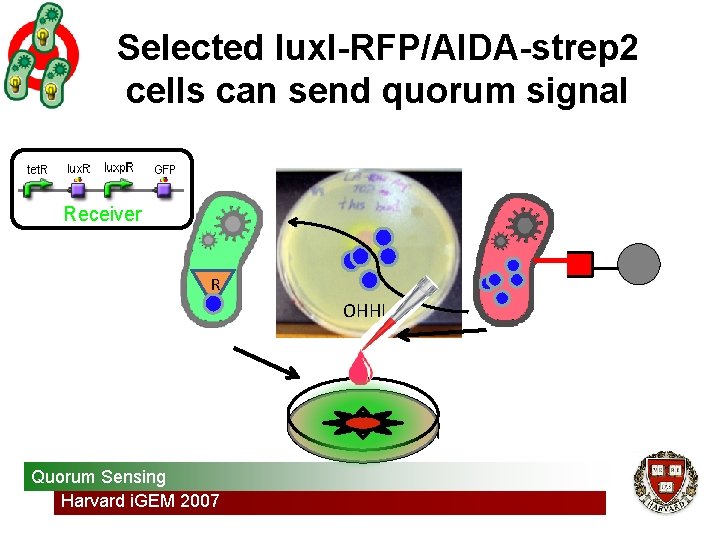
Selected lux. I-RFP/AIDA-strep 2 cells can send quorum signal Receiver R OHHL Quorum Sensing Harvard i. GEM 2007
Bacterial targeting Quorum-sensing Fec signal transduction Harvard i. GEM 2007 Fec signal transduction

Motivation: Fec System • Goal: Direct cell signaling • Method: Re-engineer an existing signal transduction pathway • Fec system: – well-characterized – only outer membrane signal transducer Fec signal transduction Harvard i. GEM 2007
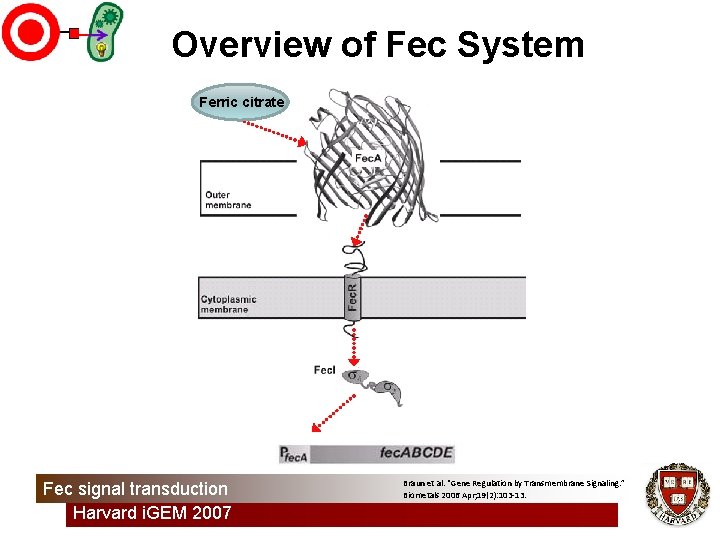
Overview of Fec System Ferric citrate Fec signal transduction Harvard i. GEM 2007 Braun et al. “Gene Regulation by Transmembrane Signaling. ” Biometals 2006 Apr; 19(2): 103 -13.
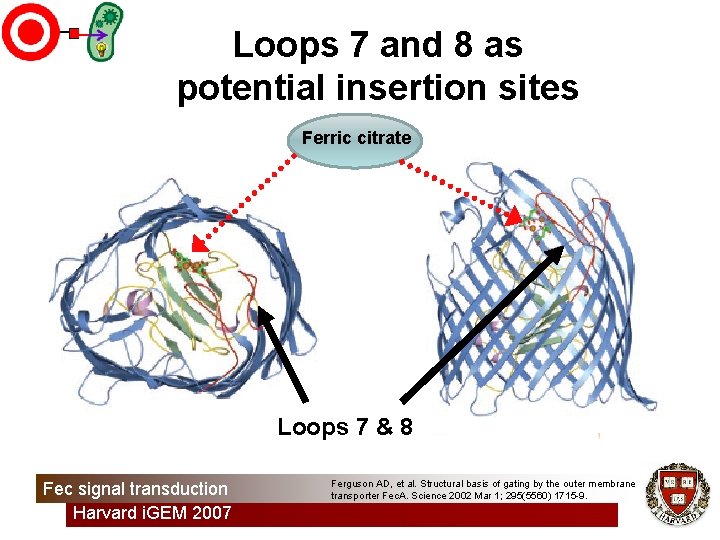
Loops 7 and 8 as potential insertion sites Ferric citrate Loops 7 & 8 Fec signal transduction Harvard i. GEM 2007 Ferguson AD, et al. Structural basis of gating by the outer membrane transporter Fec. A. Science 2002 Mar 1; 295(5560) 1715 -9.

Constructs • From Braun lab (U. Tuebingen, Germany) – Fec knock-out strain, AA 93 – Fec. IRA plasmid – PFec-GFP plasmid • p. Col. A Duet Vector – Allows regulated expression of Fec genes under T 7 promoter Fec signal transduction Harvard i. GEM 2007
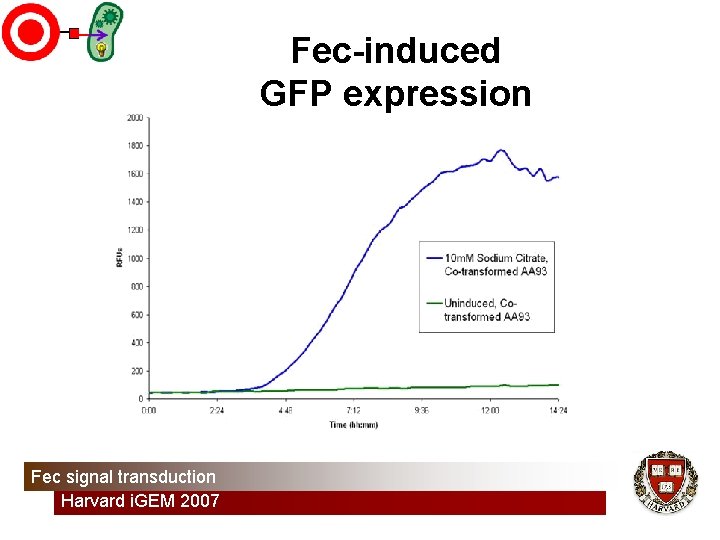
Fec-induced GFP expression Fec signal transduction Harvard i. GEM 2007

Troubleshooting and Next Steps • Problems: – Growth media: WL vs. LB? – Toxicity: membrane disruption? • Goals: – Nickel and Streptavidin Binding – Finding new targets with signaling • Random library • Computational Approach (Maranas lab) Fec signal transduction Harvard i. GEM 2007
Conclusions and Future Directions • Surface engineering – AIDA 1 • Histidine/Strep 2 tags • MACS Bacterial targeting Random peptide libraries • One-cell/two-cell quorum systems • Characterized • Targeted quorum senders Fec signal Quorum-sensing Optimize localized transduction quorum response Conclusion Harvard i. GEM 2007 • Characterized wildtype Fec signaling Computational design

Acknowledgements Advisors Teaching Fellows George Church Debra Auguste Jagesh V. Shah William Shih Pamela Silver Alain Viel Tamara Brenner Nicholas Guido Bill Senapedis Mike Strong Harris Wang Special thanks to… Volkmar Braun (U of Tuebingen) Costas Maranas (Penn State U) Conclusion Harvard i. GEM 2007 Funding HHMI Harvard Provost Harvard Life Sciences Division Harvard School of Engineering and Applied Sciences
- Slides: 25