Clathrates Clathrates Cagelike frameworks of metals with other






















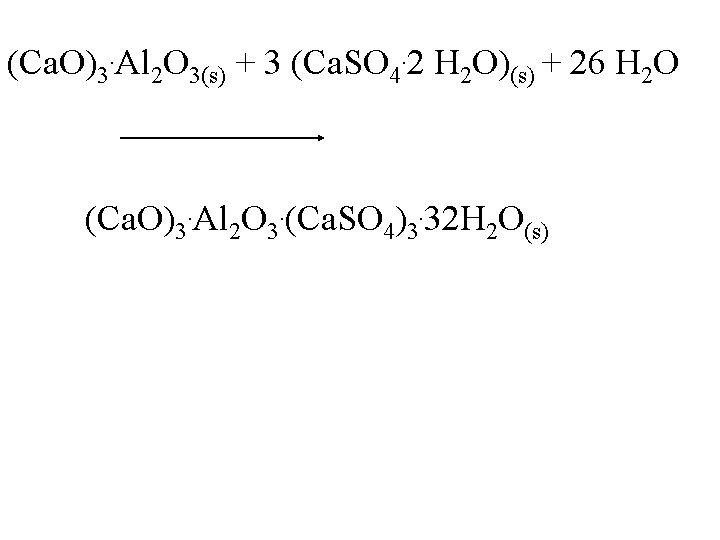
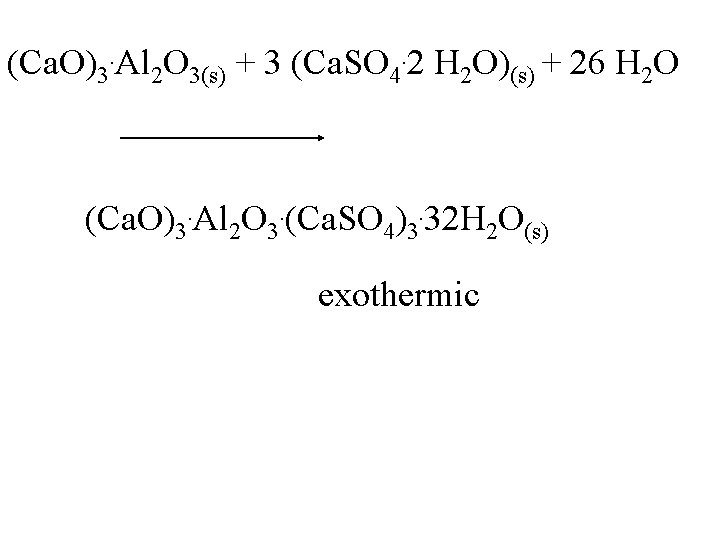

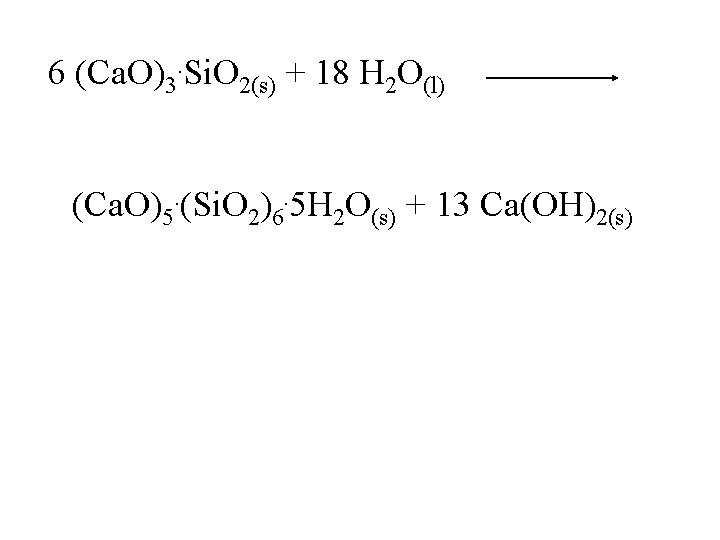
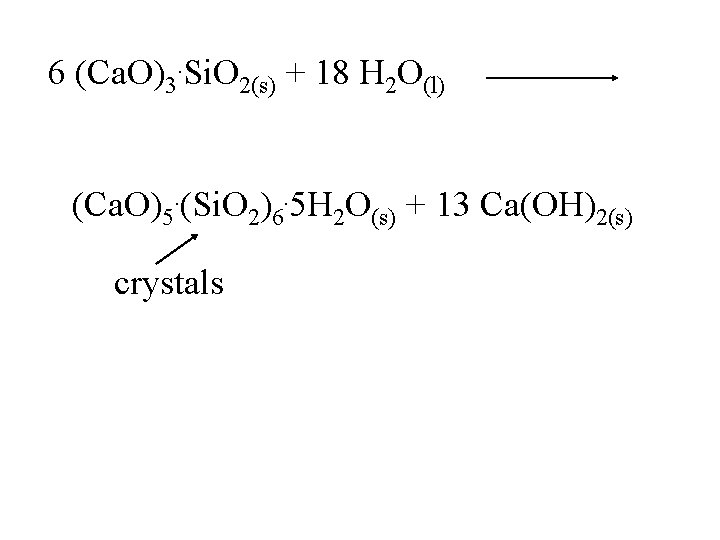

- Slides: 57

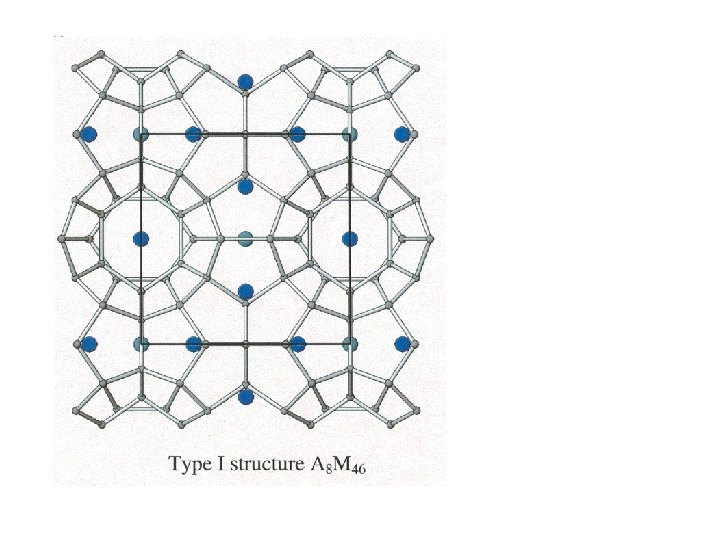
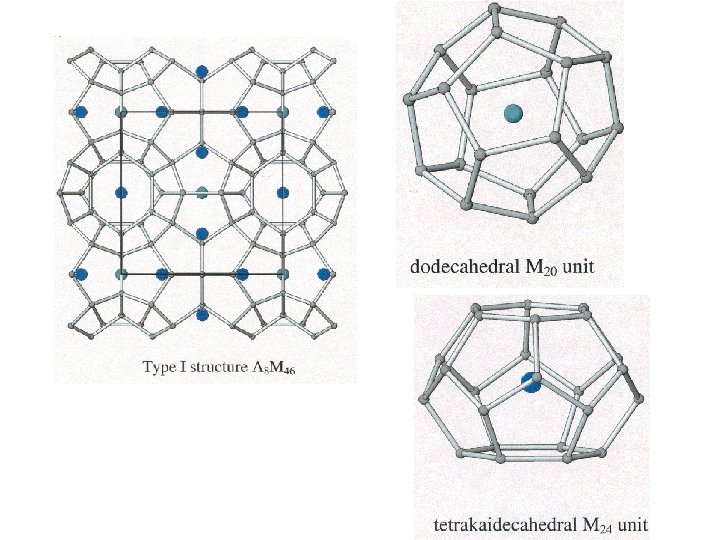
Clathrates

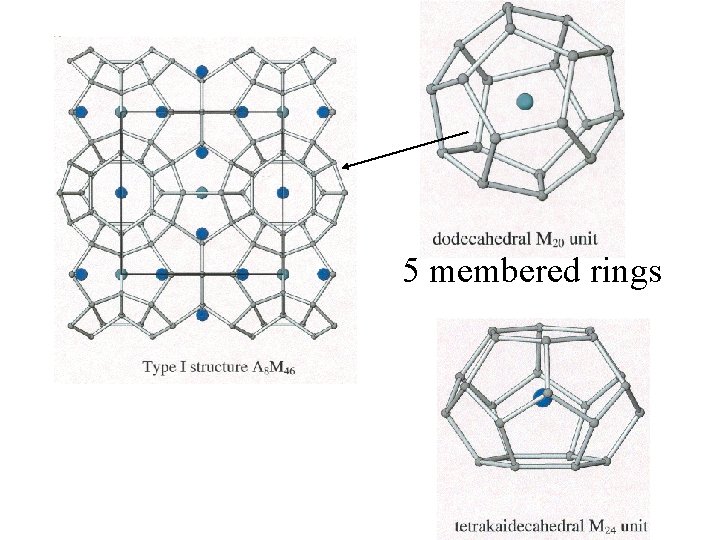
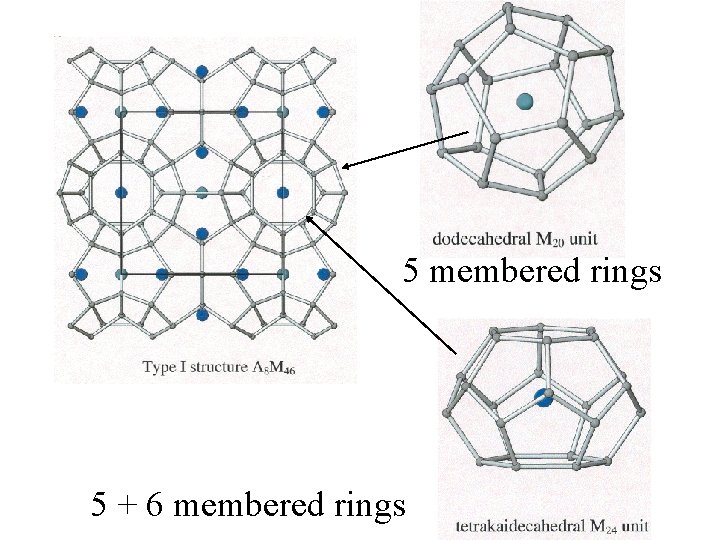
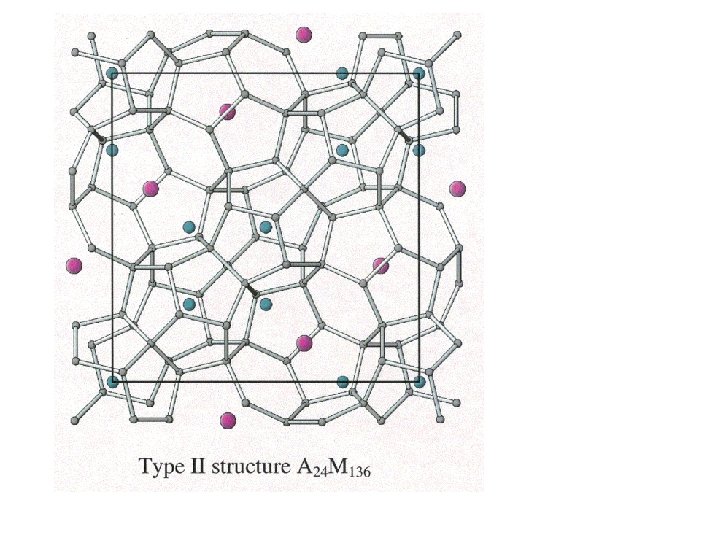
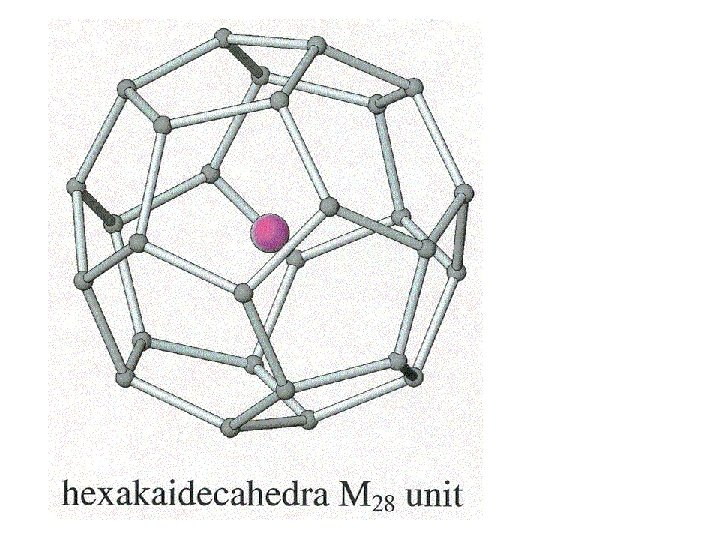
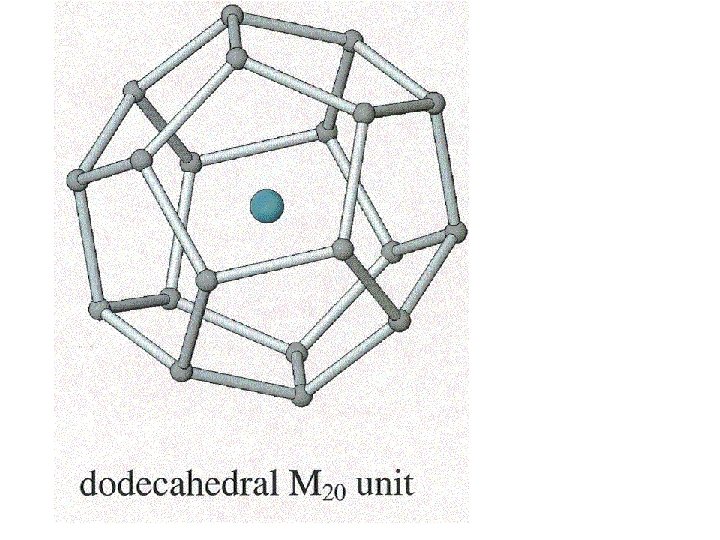
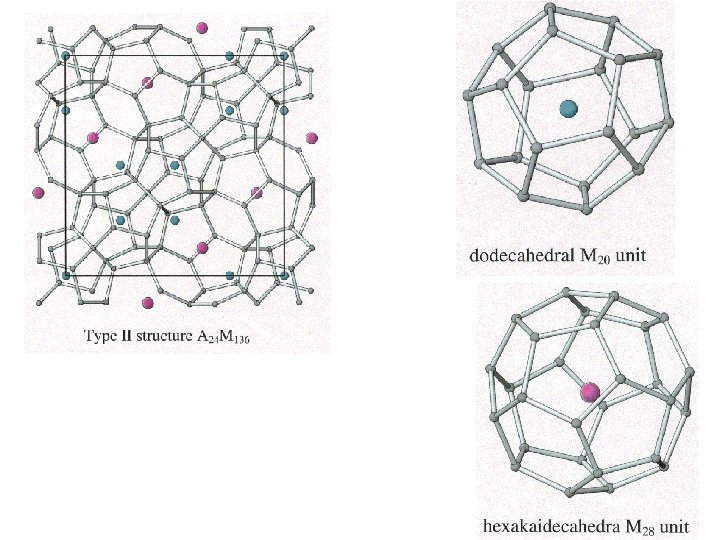
Clathrates Cage-like frameworks of metals with other metals occupying the cavities of the cages.

Clathrates These can be synthesized by mixing finely divided quantities of the metals in the correct proportions and careful heating and cooling.





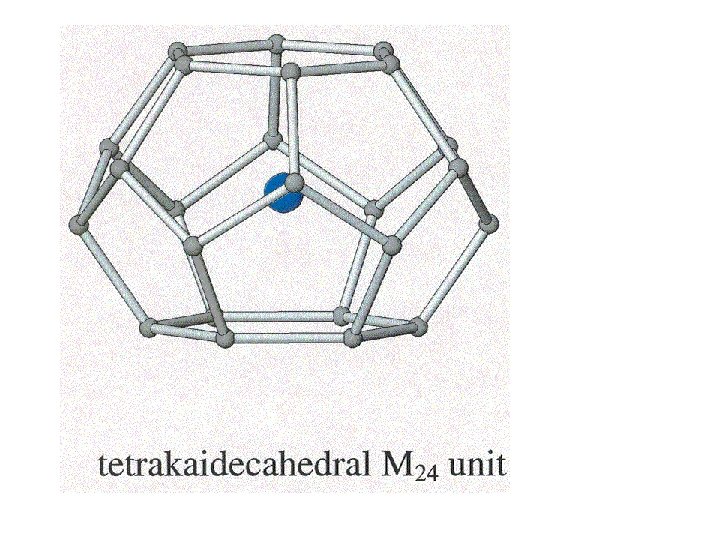
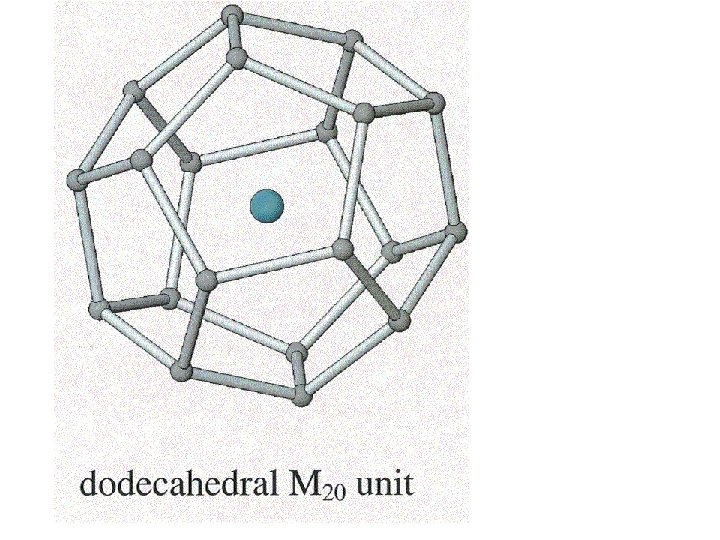
5 membered rings

5 membered rings 5 + 6 membered rings





Clathrates of this type have useful thermal and semiconductor properties.

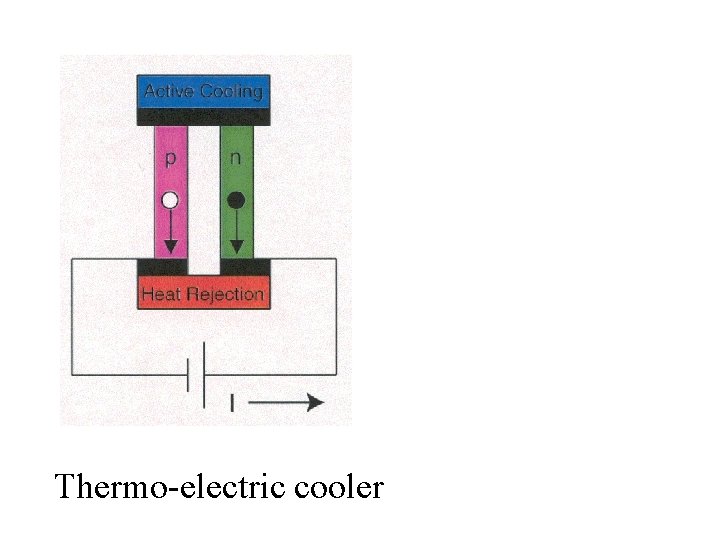
Clathrates of this type have useful thermal and semiconductor properties. A good semiconductor that has poor thermal conductivity is useful for making a thermo-electric device.

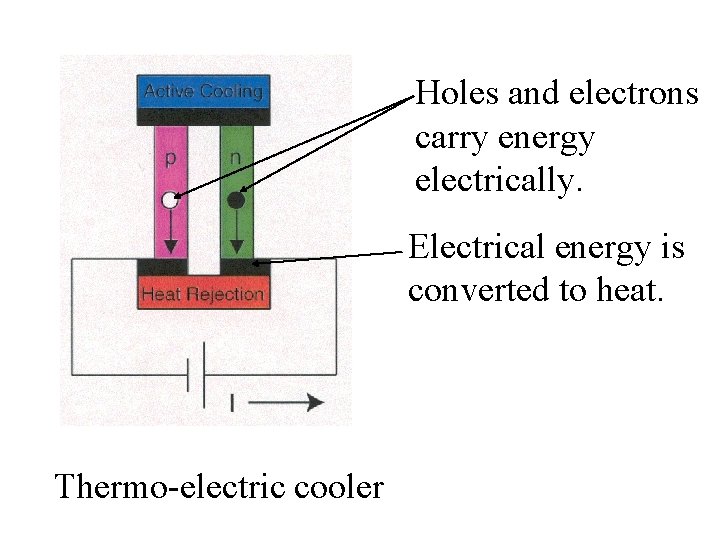
Thermo-electric cooler

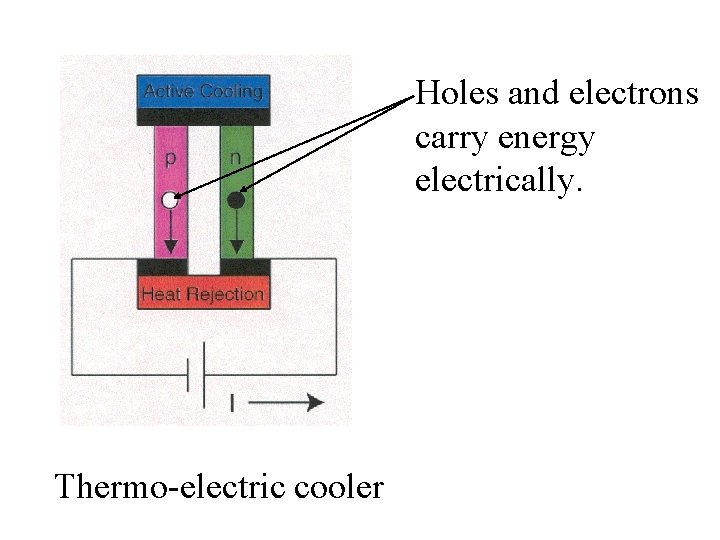
Holes and electrons carry energy electrically. Thermo-electric cooler

Holes and electrons carry energy electrically. Electrical energy is converted to heat. Thermo-electric cooler

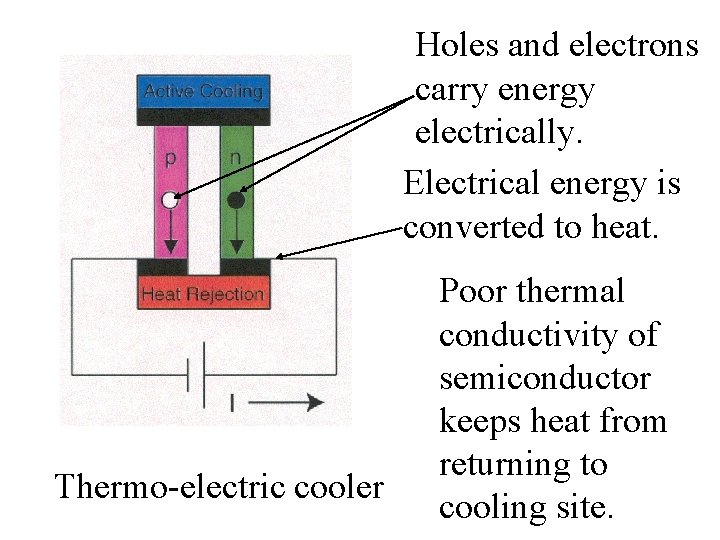
Holes and electrons carry energy electrically. Electrical energy is converted to heat. Thermo-electric cooler Poor thermal conductivity of semiconductor keeps heat from returning to cooling site.

Ceramics and glass

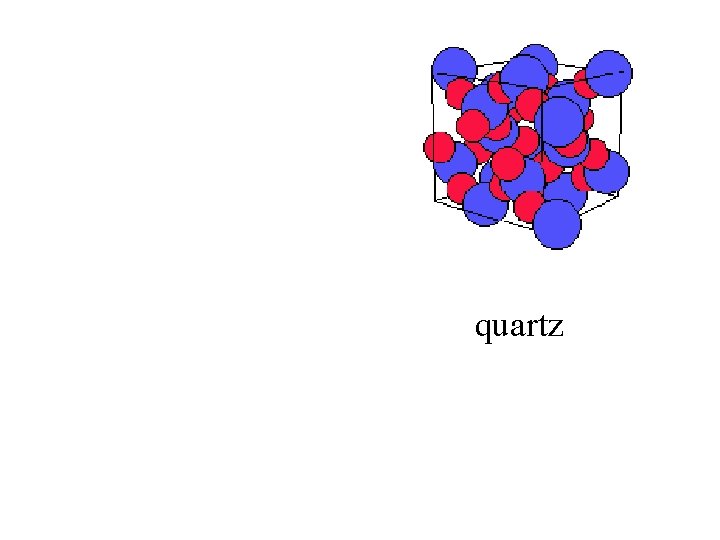
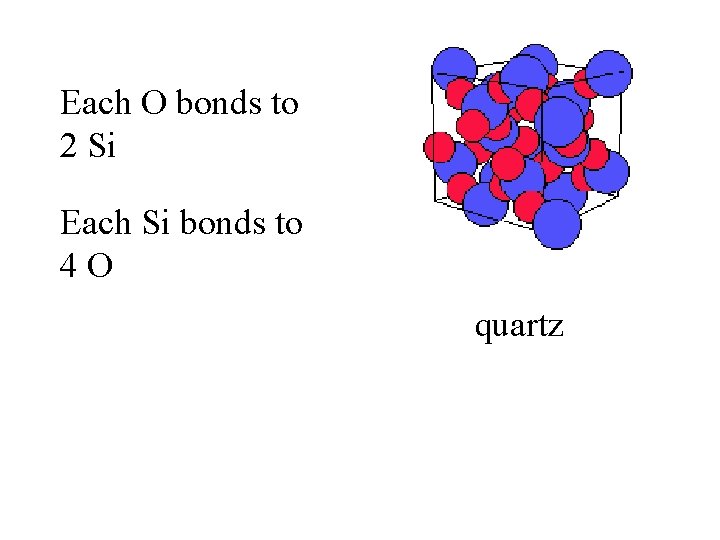
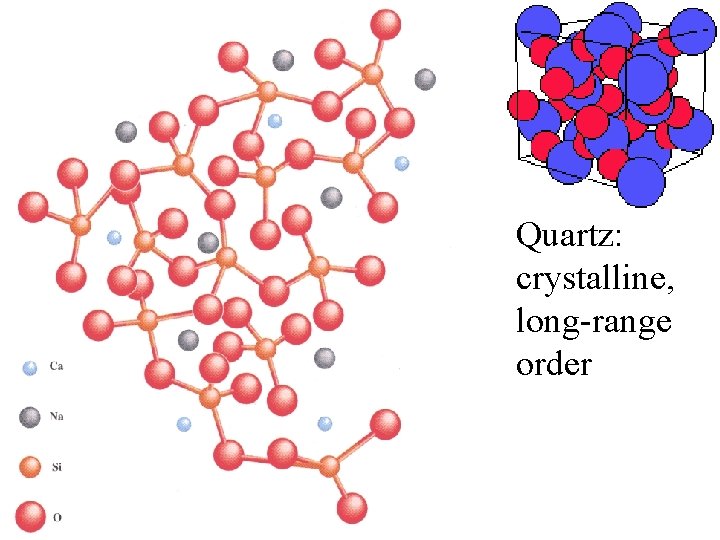
quartz

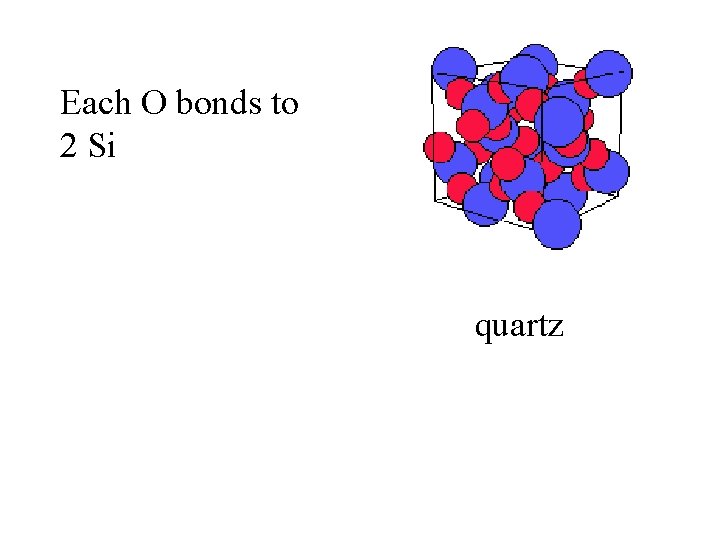
Each O bonds to 2 Si quartz

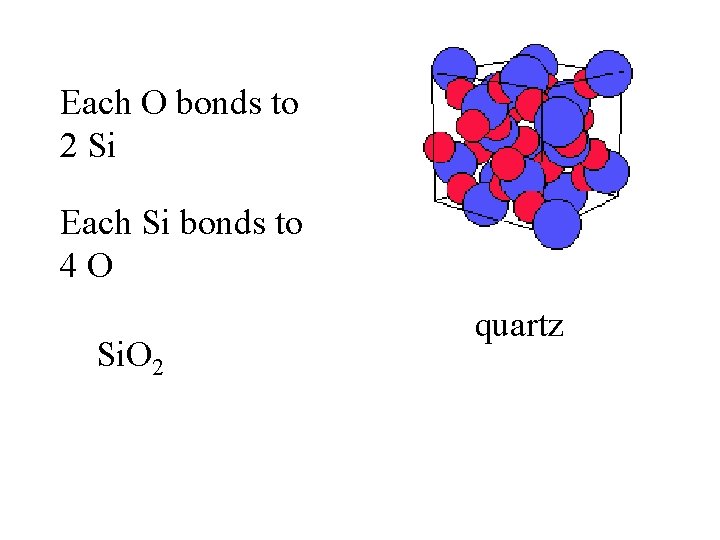
Each O bonds to 2 Si Each Si bonds to 4 O quartz

Each O bonds to 2 Si Each Si bonds to 4 O Si. O 2 quartz

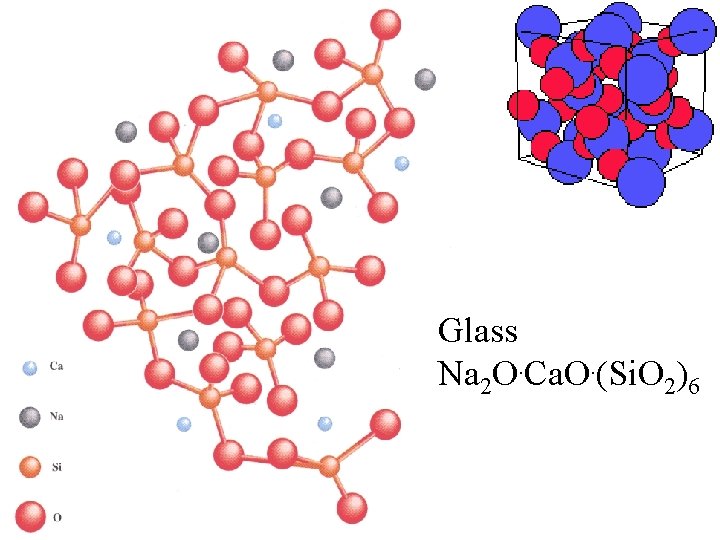
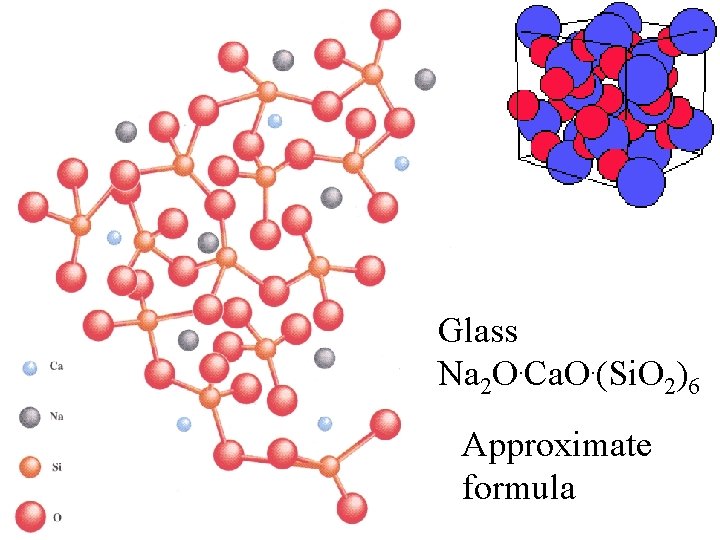
Glass Na 2 O. Ca. O. (Si. O 2)6

Glass Na 2 O. Ca. O. (Si. O 2)6 Approximate formula

Quartz: crystalline, long-range order

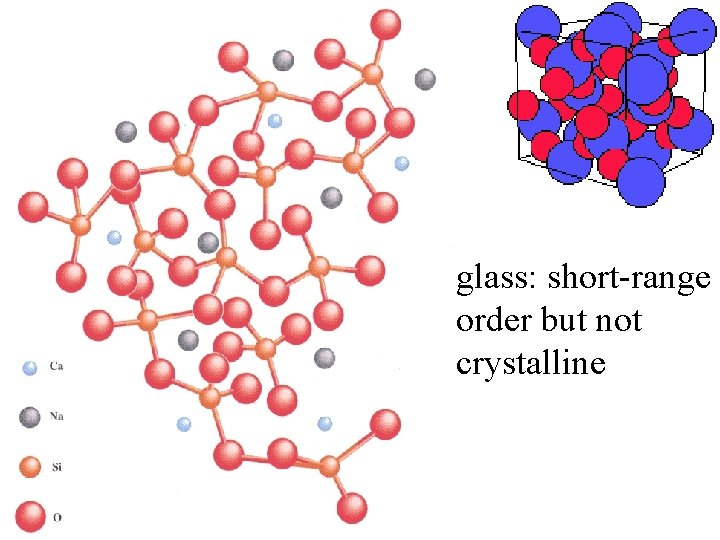
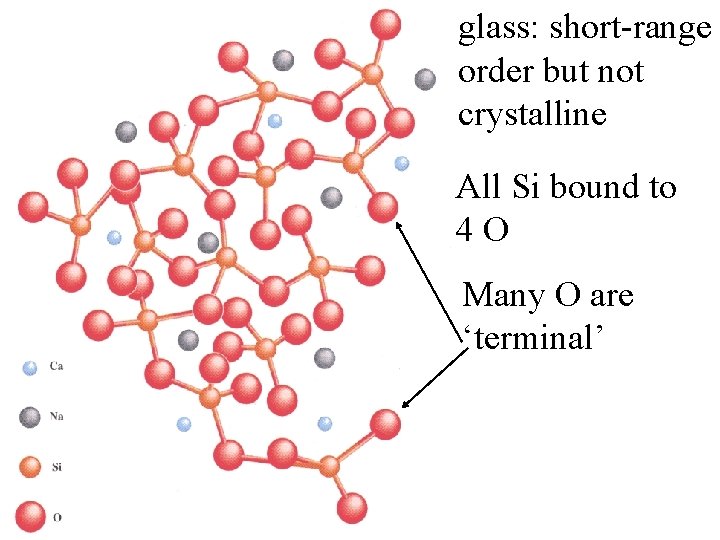
glass: short-range order but not crystalline

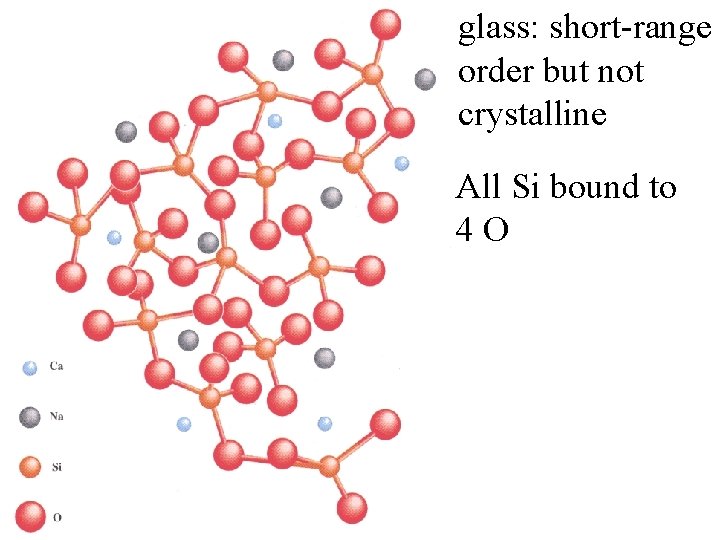
glass: short-range order but not crystalline All Si bound to 4 O

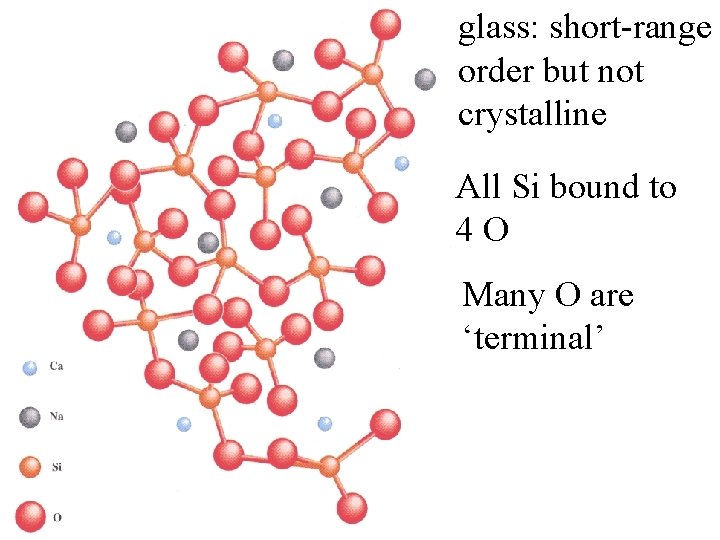
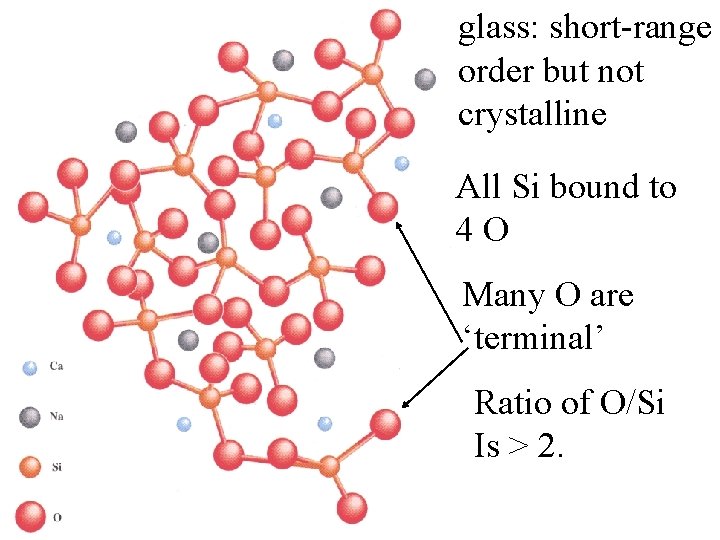
glass: short-range order but not crystalline All Si bound to 4 O Many O are ‘terminal’

glass: short-range order but not crystalline All Si bound to 4 O Many O are ‘terminal’

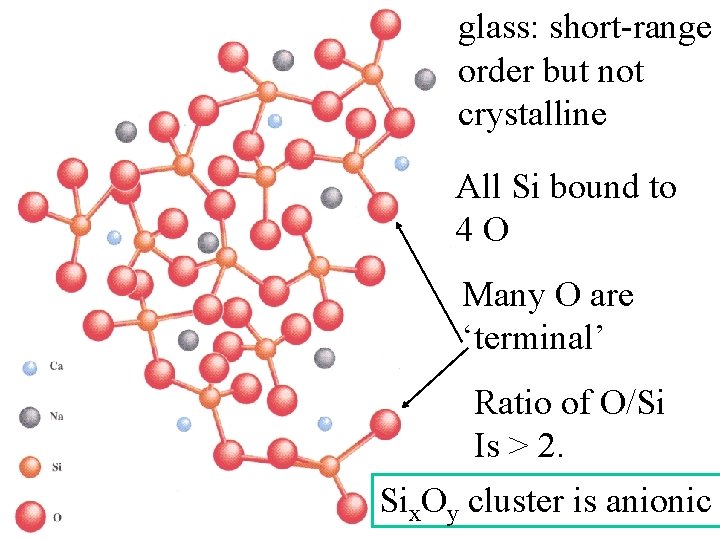
glass: short-range order but not crystalline All Si bound to 4 O Many O are ‘terminal’ Ratio of O/Si Is > 2.

glass: short-range order but not crystalline All Si bound to 4 O Many O are ‘terminal’ Ratio of O/Si Is > 2. Six. Oy cluster is anionic

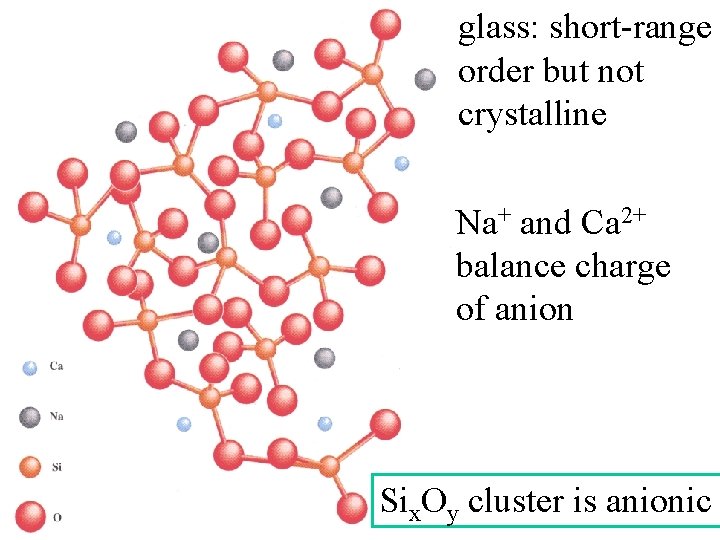
glass: short-range order but not crystalline Na+ and Ca 2+ balance charge of anion Six. Oy cluster is anionic

Properties of glass vs. quartz.

Properties of glass vs. quartz. Glass has a lower melting point

Properties of glass vs. quartz. Glass has a lower melting point Glass is softer Glass does not crystallize – this makes it easier to shape it as it cools to a solid form.

Special glasses:

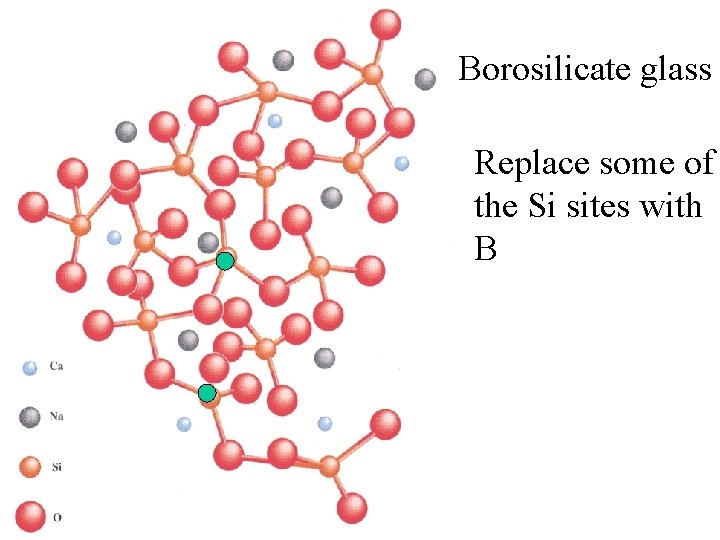
Special glasses: Borosilicate glass

Borosilicate glass Replace some of the Si sites with B

Borosilicate glasses have lower coefficients of expansion than soda-lime glasses.

Borosilicate glasses have lower coefficients of expansion than soda-lime glasses. Most materials expand when heated.

Most materials expand when heated. The coefficient of expansion is a factor, which when multiplied by the temperature change, gives the amount a material will expand or contract.

Since glasses are quite brittle, they are less likely to break when the temperature changes if they have a relatively low coefficient of expansion.

Borosilicate glasses have higher melting points than soda-lime glasses.

Borosilicate glasses have higher melting points than soda-lime glasses. Soda-lime glasses can be melted using a flame generated from methane and air.

Borosilicate glasses have higher melting points than soda-lime glasses. Soda-lime glasses can be melted using a flame generated from methane and air. It is necessary to use a methane/oxygen flame to work borosilicate glass.

Cements:

Cements: Portland cement is a specifically formulated powder.

Cements: Portland cement is a specifically formulated powder. When mixed with the proper amount of water it first forms a slurry which flows and can be formed.

When mixed with the proper amount of water it first forms a slurry which flows and can be formed. The slurry hardens and gains strength by the growth of a network of silicate crystals.
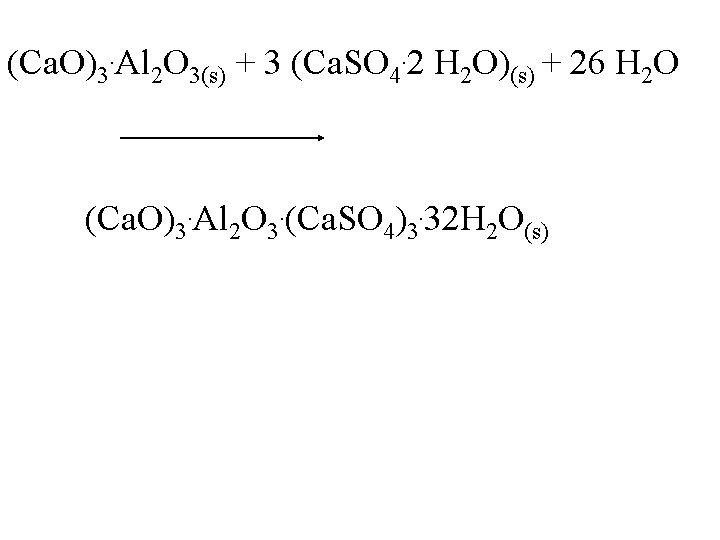
(Ca. O)3. Al 2 O 3(s) + 3 (Ca. SO 4. 2 H 2 O)(s) + 26 H 2 O (Ca. O)3. Al 2 O 3. (Ca. SO 4)3. 32 H 2 O(s)
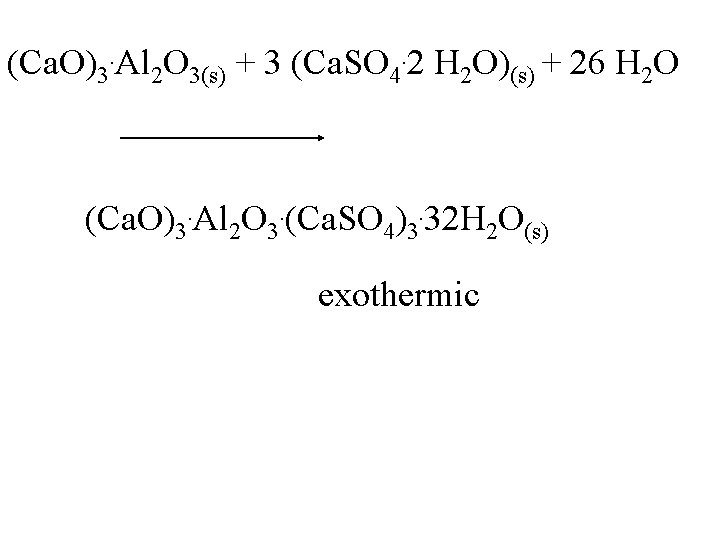
(Ca. O)3. Al 2 O 3(s) + 3 (Ca. SO 4. 2 H 2 O)(s) + 26 H 2 O (Ca. O)3. Al 2 O 3. (Ca. SO 4)3. 32 H 2 O(s) exothermic

(Ca. O)3. Al 2 O 3(s) + 3 (Ca. SO 4. 2 H 2 O)(s) + 26 H 2 O (Ca. O)3. Al 2 O 3. (Ca. SO 4)3. 32 H 2 O(s) exothermic Cooling should favor the formation of products.
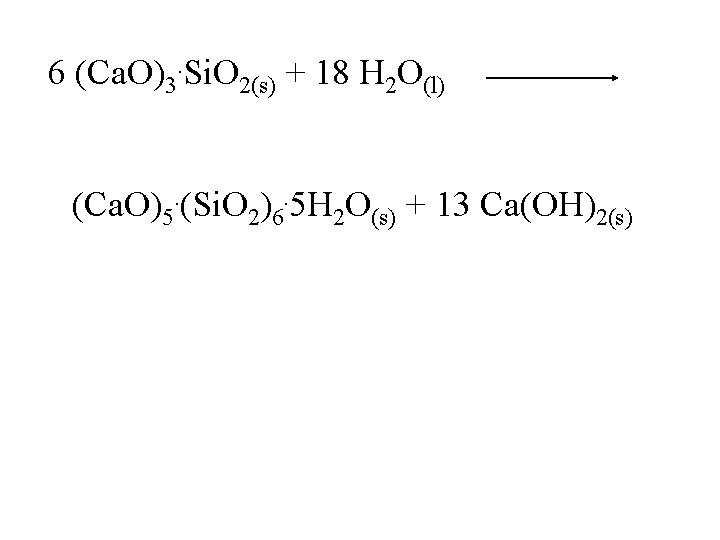
6 (Ca. O)3. Si. O 2(s) + 18 H 2 O(l) (Ca. O)5. (Si. O 2)6. 5 H 2 O(s) + 13 Ca(OH)2(s)
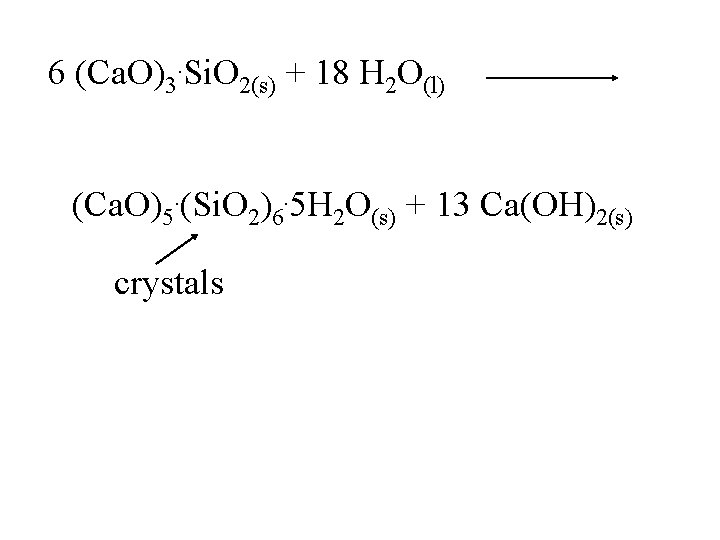
6 (Ca. O)3. Si. O 2(s) + 18 H 2 O(l) (Ca. O)5. (Si. O 2)6. 5 H 2 O(s) + 13 Ca(OH)2(s) crystals

6 (Ca. O)3. Si. O 2(s) + 18 H 2 O(l) (Ca. O)5. (Si. O 2)6. 5 H 2 O(s) + 13 Ca(OH)2(s) crystals If the mixture is allowed to dry too rapidly, sufficient water and time will not be available for crystal growth.