Classifying Matter Substances Mixtures Lesson 1 Substances and

Classifying Matter: Substances & Mixtures

Lesson 1: Substances and Mixtures Matter What is matter What is NOT Matter Types of energy: heat, Anything that has mass AND takes up space light, sound, electrical and electromagnetic Solids, Liquids, Gases, and Plasma Types of forces: gravitational and magnetic

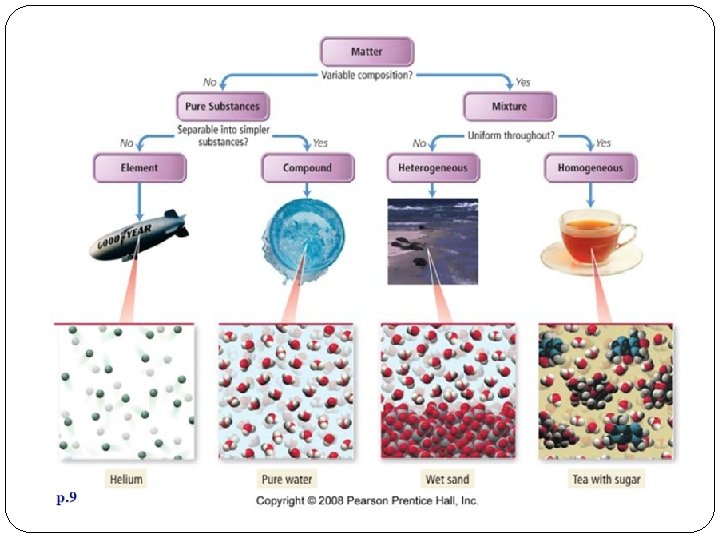
What is Matter Made of? All matter is made up of particles called Atoms. The different ways atoms combine with each other help us identify different types of matter. Matter is organized into 2 main groups: 1. Pure Substances 2. Mixtures

Pure Substances Matter with a composition that is always the same is a pure substance. Pure substances can be: q. Elements q. Compounds
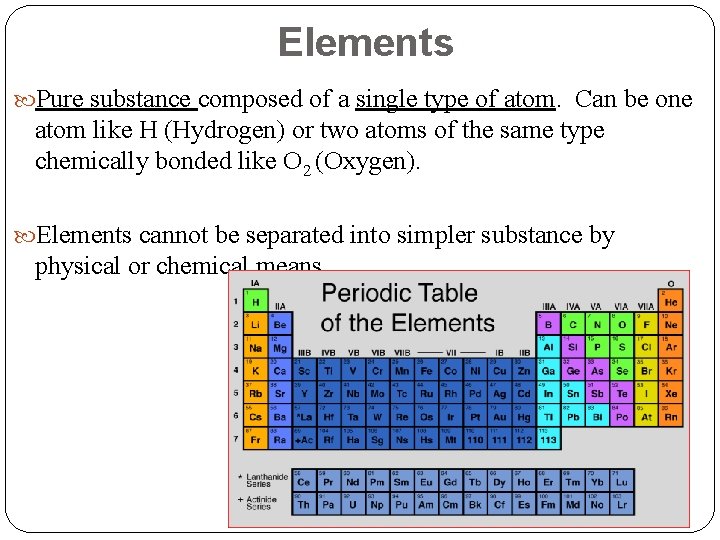
Elements Pure substance composed of a single type of atom. Can be one atom like H (Hydrogen) or two atoms of the same type chemically bonded like O 2 (Oxygen). Elements cannot be separated into simpler substance by physical or chemical means.


Compounds Pure substances composed of two or more different elements joined by chemical bonds. Made of elements in a specific ratio that is always the same Has a chemical formula Can only be separated by chemical means, not physically

Practice with types of substances: elements and compounds Explain which of these are elements and which are compounds: 1. 2. 3. 4. 5. Hydrogen Water (H 2 O) Carbon Dioxide (CO 2) Oxygen (O 2)

Practice with elements and compounds Explain which of these are elements and which are compounds: 1. 2. 3. 4. 5. Hydrogen (H) ELEMENT Water (H 2 O) COMPOUND Carbon ( C) ELEMENT Carbon Dioxide (CO 2) COMPOUND Oxygen (O 2) ELEMENT

Molecules A MOLECULE is two or more atoms that are bonded and act as a unit. . Examples are most compounds like H 2 O (water) , CO 2 (carbon dioxide gas), CH 4 (methane gas)

Mixtures A combination of two or more pure substances that are NOT chemically combined. substances held together by physical forces, not chemical No chemical change takes place Each item retains its properties in the mixture They can be separated physically

HOMOGENEOUS MIXTURES A mixture in which 2 or more substances are evenly mixed but not bonded together. (Solution) You cannot see the different parts and therefore seem to be a single substance. Ex: Salt water (liquid) Dry air: Oxygen & Nitrogen (gas) Brass- Zinc & Copper (solid)

HETEROGENEOUS MIXTURES A mixture in which 2 or more substances are not evenly mixed. Suspensions: the particles are much larger than solutions and eventually settle out. § Can be stirred or shaken to mix q Ex: qsalad dressing qblood qsnow globe

HETEROGENEOUS MIXTURES Colloids: the particles are smaller than suspension but larger than particles in a solution Particles do not settle out Ex: Clouds , Whip cream , Gelatin , Fog & Smoke, Paint , Ink , Pearls, Opals Colloids can be distinguished from solutions using the Tyndall effect- a beam of light passing through a solution is visible because light is reflected or scattered by the particles in the colloid.

Can you identify the following? You will be shown a series of photos. Tell if each photo represents an item composed of an element, compound, or mixture. Review: An element contains just one type of atom. A compound contains two or more different atoms joined together. A mixture contains two or more different substances that are only physically joined together, not chemically. A mixture can contain both elements and compounds.

Element, Compound, or Mixture? Rocks

Element, Compound, or Mixture? Rocks

Element, Compound, or Mixture? Copper

Element, Compound, or Mixture? Copper

Element, Compound, or Mixture? Jelly Beans

Element, Compound, or Mixture? Jelly Beans

Element, Compound, or Mixture? Table Sugar

Element, Compound, or Mixture? Table Sugar

Element, Compound, or Mixture? Diamond

Element, Compound, or Mixture? Diamond

Element, Compound, or Mixture? Tea

Element, Compound, or Mixture? Tea

Element, Compound, or Mixture? Salt

Element, Compound, or Mixture? Salt

Element, Compound, or Mixture? Neon Gas

Element, Compound, or Mixture? Neon Gas

Element, Compound, or Mixture? Salad

Element, Compound, or Mixture? Salad

Element, Compound, or Mixture? Pure Water

Element, Compound, or Mixture? Pure Water

Element, Compound, or Mixture? Aluminum

Element, Compound, or Mixture? Aluminum

Element, Compound, or Mixture? Lemonade

Element, Compound, or Mixture? Lemonade

Element, Compound, or Mixture? Silver

Element, Compound, or Mixture? Silver

Element, Compound, or Mixture? Sand

Element, Compound, or Mixture? Sand

Notes Detailed notes are located at: http: //www. middleschoolscience. com/elementscompounds-mixtures-notes-isn. pdf Flow Chart: http: //www. middleschoolscience. com/matter-flow-chartisn. pdf
- Slides: 45