CLASSIFYING MATTER PURE SUBSTANCES Every sample of a
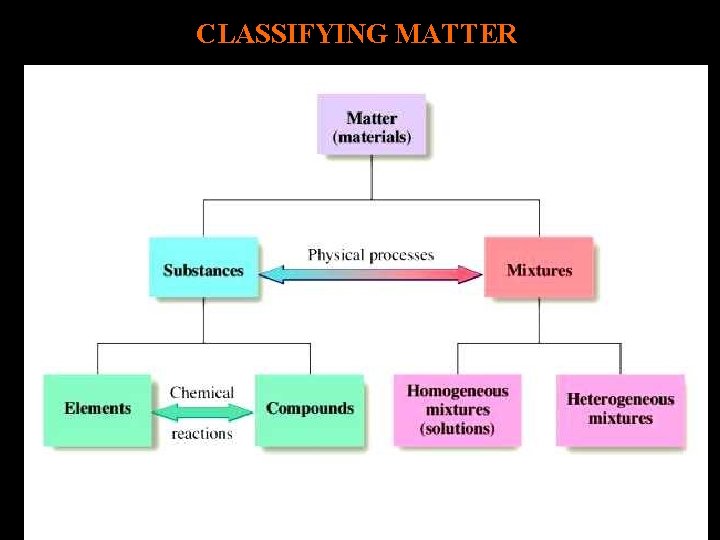
CLASSIFYING MATTER
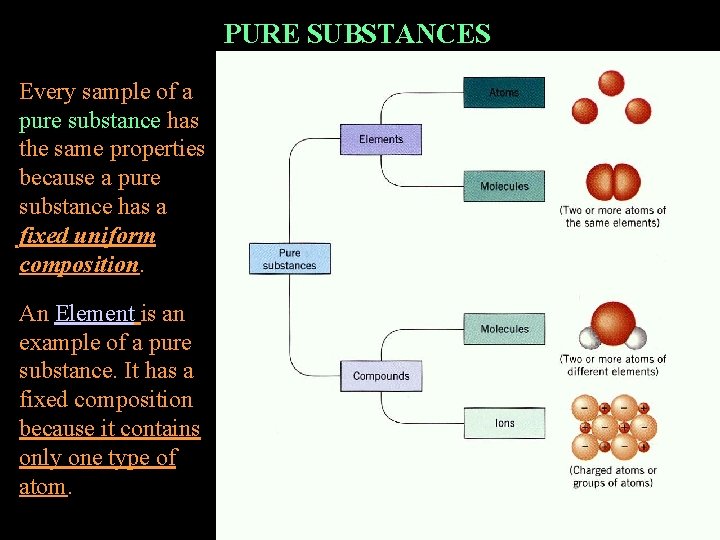
PURE SUBSTANCES Every sample of a pure substance has the same properties because a pure substance has a fixed uniform composition. An Element is an example of a pure substance. It has a fixed composition because it contains only one type of atom.
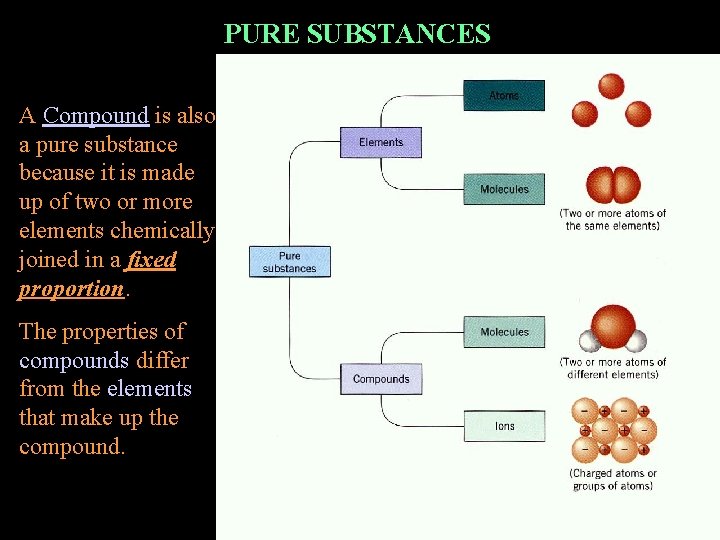
PURE SUBSTANCES A Compound is also a pure substance because it is made up of two or more elements chemically joined in a fixed proportion. The properties of compounds differ from the elements that make up the compound.

MIXTURES The properties of a mixture can vary because their composition is not fixed. Mixtures are not combined chemically like compounds. Heterogeneous Mixture – The parts of the mixture are noticeably different and are distributed unevenly Homogeneous Mixture – The parts of a the mixture are distributed evenly (not chemically), which makes it difficult to distinguish one substance from the other. The particles that make up a homogeneous mixture are very small


HETEROGENEOUS MIXTURE The parts of a burger are noticeably different. They are not chemically combined, and can be separated. A suspension is an example of a heterogeneous mixture that contains noticeably different particles. However, They are small enough to appear evenly distributed, but after a period of time the particles separate into layers.


HOMOGENEOUS MIXTURE A solution is an example of a homogeneous mixture. It appears to contain only one substance. The substances in a solution that are in the least abundance are termed the solutes. They are dissolved in the majority substance called the solvent. Too much solute saturates the solution, which results in undissolved solute left over. 1. Solutions can not be filtered, but they can be separated by other means. Distillation is one of those processes.

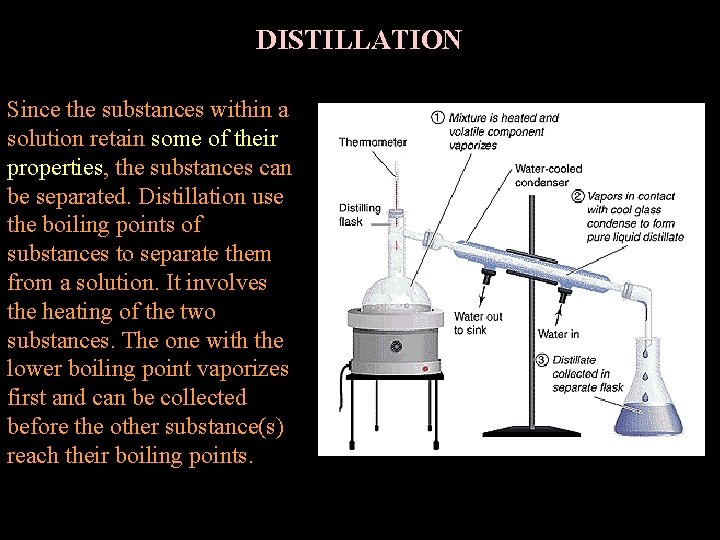
DISTILLATION Since the substances within a solution retain some of their properties, the substances can be separated. Distillation use the boiling points of substances to separate them from a solution. It involves the heating of the two substances. The one with the lower boiling point vaporizes first and can be collected before the other substance(s) reach their boiling points.

COLLOIDS 1. Colloids are more homogenous than heterogeneous in appearance with intermediate particle size 2. The particles are too large to form true solutions, but small enough so they are not easily distinguished and do not settle into layers like suspensions. 3. Like solutions colloids can not be filtered. Glasses filled with gelatin 4. Colloids scatter light when light attempts to pass through it. This is an identifying characteristic that separates a colloid from a solution.

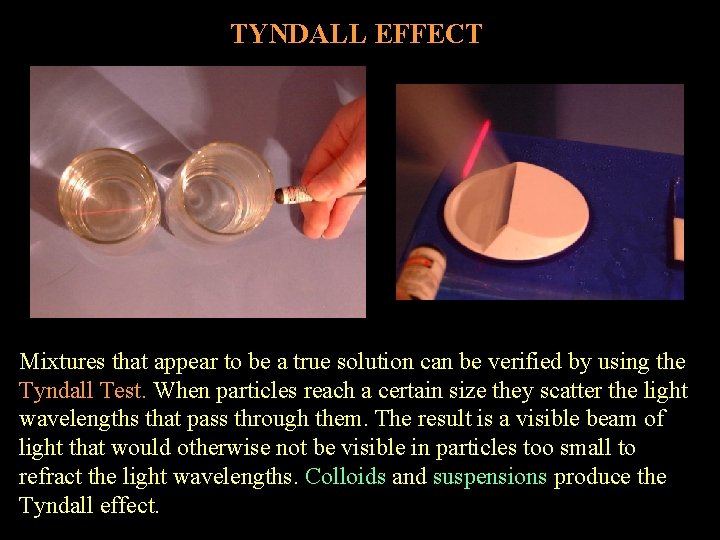
TYNDALL EFFECT Mixtures that appear to be a true solution can be verified by using the Tyndall Test. When particles reach a certain size they scatter the light wavelengths that pass through them. The result is a visible beam of light that would otherwise not be visible in particles too small to refract the light wavelengths. Colloids and suspensions produce the Tyndall effect.

PHYSICAL PROPERTIES OF MATTER • Density: The ratio of mass to volume in any given sample of a substance • Viscosity: The resistance of a liquid to flow • Conductivity The ability to allow heat or electricity to flow through it • Malleability: The ability of a solid to be shaped without breaking. This property is sometimes referred to as Ductility • Hardness: A comparison between two material’s abilities to scratch the other. • Melting Point: The point at which a substance changes from a solid to a liquid • Boiling Point: The point at which a substance changes from a liquid to a gas
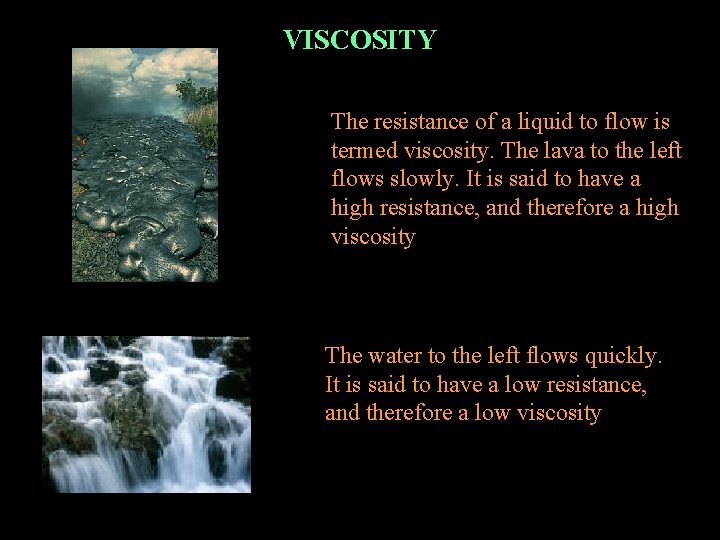
VISCOSITY The resistance of a liquid to flow is termed viscosity. The lava to the left flows slowly. It is said to have a high resistance, and therefore a high viscosity The water to the left flows quickly. It is said to have a low resistance, and therefore a low viscosity
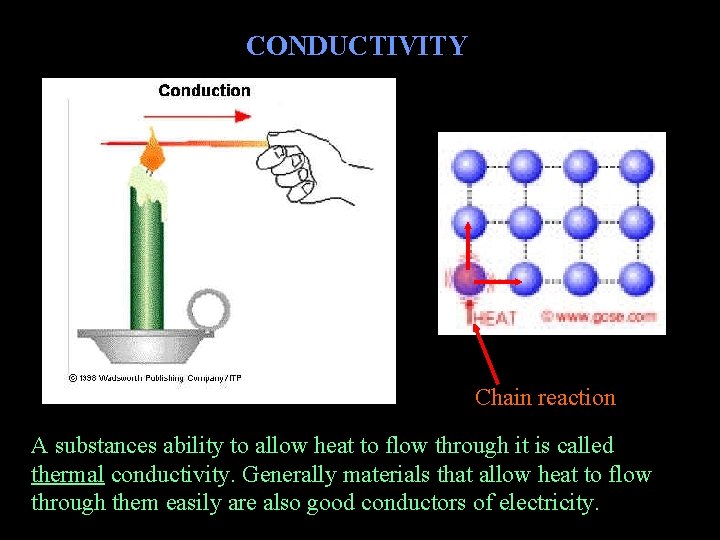
CONDUCTIVITY Chain reaction A substances ability to allow heat to flow through it is called thermal conductivity. Generally materials that allow heat to flow through them easily are also good conductors of electricity.

MALLEABILITY / DUCTILITY The ability to bend or hammer a substance without breaking it is called malleability or ductility. This property is important when working with metals, which are often reshaped in manufacturing processes.

MELTING AND BOILING POINTS All matter can change from one state to another. The change in state does change some of the properties of matter, but the composition remains unchanged. Water freezes / melts at 0˚ Celsius and Boils/Condenses at 100˚ Celsius. These temperatures are unique to each element / compound.

MATTER HAS MASS All matter is made up of tiny particles called atoms. Even the subatomic particles of an atom have mass. Mass is a measure of how much matter is in an object.


VOLUME BEFORE AFTER Since all matter is made up of tiny atoms, it must occupy space. The space it occupies, called volume, is three dimensional and can be measured with a ruler (length x height x width), or by displacement of water. When an object is placed in a liquid, the liquid rises to accommodate the object’s space. The rise in the water level, if performed in a graduated cylinder, can be measured which equals the volume of the part of the object that is in the water.


A DENSITY B C Density is the ratio of mass to volume in any given sample of matter. Example A has a low density because there is a large amount of space between the particles. Example B has a larger number of particles in a smaller space than A. Example C has the greatest number of particles in an occupied space. Example C is said to be the densest of the three.

DENSITY • Two objects may have the same volume, but have different densities because they contain different amounts of matter “mass”. • Both containers have the same volume, therefore, they both take up the same amount of space. • The container on the right has the lower density because it contains fewer particles than the container on the left. The ratio of mass to volume is less in the container on the right.

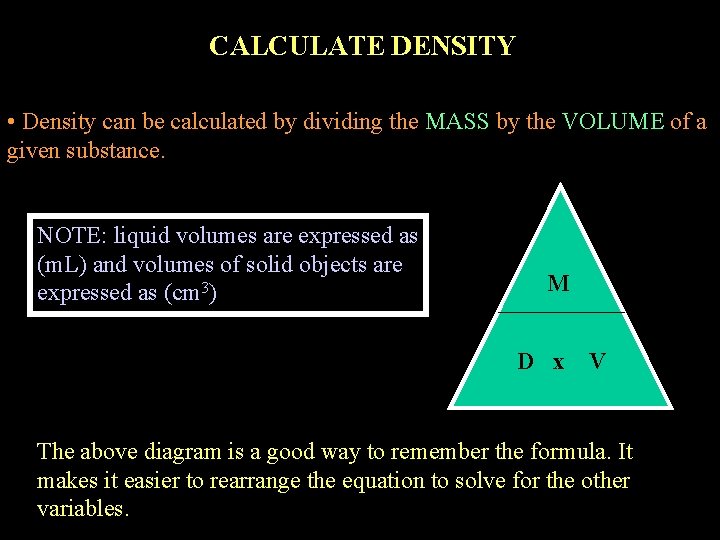
CALCULATE DENSITY • Density can be calculated by dividing the MASS by the VOLUME of a given substance. NOTE: liquid volumes are expressed as (m. L) and volumes of solid objects are expressed as (cm 3) M D x V The above diagram is a good way to remember the formula. It makes it easier to rearrange the equation to solve for the other variables.

TEMPERATURE AND DENSITY An increase in temperature causes atoms within a substance to move faster, therefore occupying more space. This movement increases their volume and decreases the density. When substances are cooled, the molecules move slowly and occupy less space. The result is a decrease in volume and an increase in density. The mass of the substance remains the same. Thermal expansion or contraction is a physical change.

BUOYANCY WEIGHT Archimedes’ Principle: The buoyant force on an object is equal to the weight of the fluid displaced by the object. The buoyant force of the fluid acting on the ball is equal to the weight (gravitational pull) of the water displaced by the ball Buoyancy is the ability of a liquid to exert an upward force It is the apparent loss of weight an object experiences when placed in a fluid. In the above example the buoyant force is not strong enough to support the weight of the golf ball, but it still exerts an upward force, which reduces the weight of the golf ball (not shown).

BUOYANCY CONTINUED The weight of the displaced liquid equals the buoyant force Buoyancy is the upward force of a fluid to support an object. When the weight of the displaced fluid (the buoyant force) equals the weight of the object, the object stops sinking. If the volume of the object can not displace enough fluid (buoyant force) to equal the weight of the object, then the object continues to sink because the weight of the object is greater than the weight of the displaced fluid (the buoyant force)

HOW ARE BUOYANCY AND DENSITY SIMILAR? HOW ARE THEY DIFFERENT? Density is the ratio of mass to volume for any given substance (solid, liquid, or gas). Buoyancy is the upward force a liquid exerts on an object. Density determines the buoyancy of a liquid. Every milliliter of liquid that is displaced has weight. The larger the density the more weight per each displaced milliliter of liquid. That is why objects must have a density less than the density of the liquid they are placed in.


The amount of sugar in regular Coke has more mass than the Nutrasweet used in Diet coke. The volumes of the containers are the same, but the weight is different. This results is a change in the density of the two cans. The density of Regular Coke is greater than the density of the water, therefore the can sinks

HYPERLINK http: //atlas. geo. cornell. edu/education/student/isostasy. html
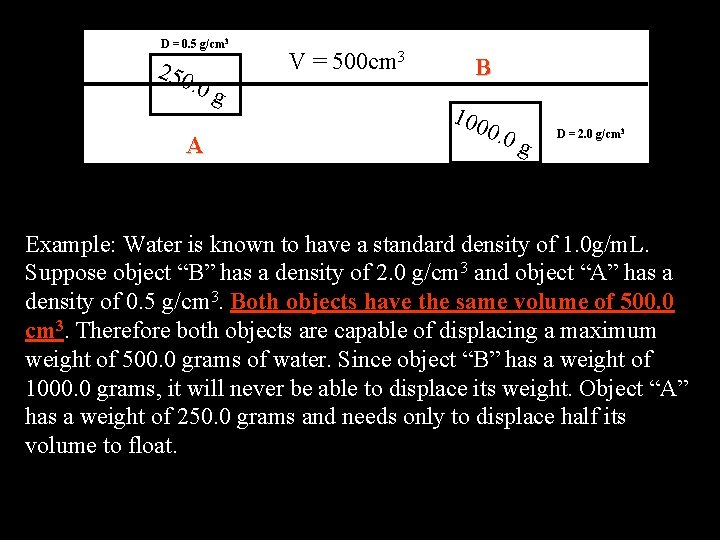
D = 0. 5 g/cm 3 250 . 0 g A V = 500 cm 3 B 100 0. 0 g D = 2. 0 g/cm 3 Example: Water is known to have a standard density of 1. 0 g/m. L. Suppose object “B” has a density of 2. 0 g/cm 3 and object “A” has a density of 0. 5 g/cm 3. Both objects have the same volume of 500. 0 cm 3. Therefore both objects are capable of displacing a maximum weight of 500. 0 grams of water. Since object “B” has a weight of 1000. 0 grams, it will never be able to displace its weight. Object “A” has a weight of 250. 0 grams and needs only to displace half its volume to float.

PHYSICAL CHANGES A physical change occurs when some of the properties of a substance change, but the composition of the matter does not change. For example, when water freezes its volume increases. The increased volume reduces the density (D = mass / volume). However, ice is still water. The composition does not change

CHEMICAL PROPERTIES A chemical property is any ability to produce a change in the composition of matter. The change alters the physical properties of matter by altering the composition of the substances. 1. Flammability – A material’s ability to burn in the presence of oxygen 2. Reactivity – How readily a substance combines chemically with other substances

CHEMICAL CHANGES AND REACTIVITY A chemical change occurs when a substance reacts with another substance to form one or more new substances 1. A change in color 2. Production of a gas 3. Formation of a precipitate 4. Change in thermal energy (chemical or physical)


CONSERVATION OF MATTER During a chemical change, within a closed system, the amount of matter remains constant--matter is neither created nor destroyed. To prove the conservation of matter, you simply mass the contents (reactants) of the chemical reaction before and after (products) the reaction takes place. The mass of the reactants and the products will be the same so long as the reaction takes place in a closed system. (Earth is a closed system!)
- Slides: 39