Classifying Chemical Reactions Synthesis Reaction Two or more











- Slides: 12

Classifying Chemical Reactions

Synthesis Reaction Two or more reactants form a single product Example: A + B AB 2 Fe + 3 Cl 2 2 Fe. Cl 3

Decomposition Reaction Single compound breaks down into two or more elements or compounds Example: AB A + B NH 4 NO 3 N 2 O + 2 H 2 O

Single Replacement Reaction A reaction in which the atoms of one element replace the atoms of another element in a compound Example: A + BX AX + B 2 Li + 2 H 2 O 2 Li. OH + H 2

Double Replacement Reaction Exchange of ions between two compounds Example: AX + BY AY + BX Ca(OH)2 + 2 HCl Ca. Cl 2 + 2 H 2 O

Combustion Reaction Oxygen combines with a substance and releases energy in the form of heat and light (water is a product) Examples: 2 H 2 + O 2 2 H 2 O CH 4 + 2 O 2 CO 2 + 2 H 2 O All reactions where a hydrocarbon reacts with oxygen will always form carbon dioxide and water as

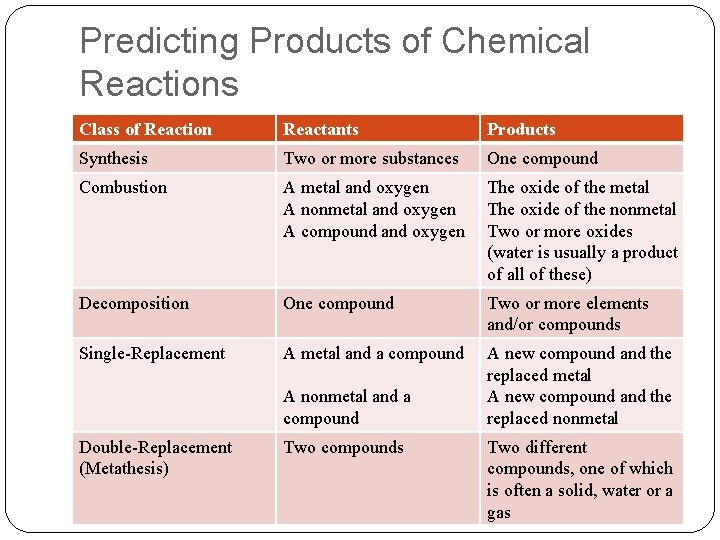
Predicting Products of Chemical Reactions Class of Reaction Reactants Products Synthesis Two or more substances One compound Combustion A metal and oxygen A nonmetal and oxygen A compound and oxygen The oxide of the metal The oxide of the nonmetal Two or more oxides (water is usually a product of all of these) Decomposition One compound Two or more elements and/or compounds Single-Replacement A metal and a compound A new compound and the replaced metal A new compound and the replaced nonmetal A nonmetal and a compound Double-Replacement (Metathesis) Two compounds Two different compounds, one of which is often a solid, water or a gas

Practice Problem How would you classify the following chemical reaction? 2 C 4 H 10 + 13 O 2 8 CO 2 + 10 H 2 O Answer: Combustion

Practice Problem How would you classify the following reaction? Al + I 2 Al. I 3 Answer: Synthesis

Practice Problem How would you classify the following reaction Ag. NO 3 + Na 2 S Ag 2 S + Na. NO 3 Answer: Metathesis (Double Replacement)

Practice Problem Predict the products to the following reaction. 2 K + Zn. Cl 2 Answer: Single Replacement 2 KCl + Zn

Predict the products to the following chemical reaction. 2 Na. N 3 Answer: Decomposition 3 N 2 + 2 Na