Classification of Minerals by Anionic Species Anions are

Classification of Minerals by Anionic Species (Anions are negative ions)

Anions are Negative Ions • May be single species – O 2 -, F-, Cl-, S 2 - • May be anionic group (polyanion): – CO 32 -, SO 42 -, PO 43 - • Silicates are classified by polymerization. – Isolated tetrahedra – Chains – Sheets – Frameworks
Periodic Table

Native Elements (no anions) • Pure elements that occur as minerals – Graphite, diamond (C) – Sulfur (S) – Gold (Au), Silver (Ag), Copper (Cu) – Iron (kamacite) – Nickel (taenite) – As, Te, Se, Pt, Ir, Os, Pd, Ru, Rh
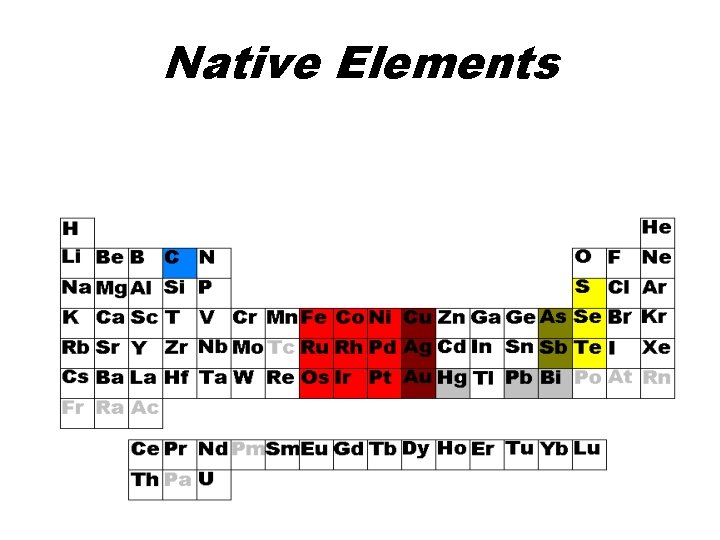
Native Elements

Halides: anion is F, Cl, Br or I • • • Halogens are the elements F, Cl, Br, I Halogens are monovalent anions Halite (Na. Cl), Sylvite (KCl) Fluorite (Ca. F 2) Cryolite Na 3 Al. F 6

Sulfides: anion is S • Sulfur is “anion”, but mostly it is covalent – Pyrite, marcasite (Fe. S 2), Chalcopyrite (Cu. Fe. S 2) – Sphalerite Zn. S, Galena (Pb. S) – Chalcocite Cu 2 S, Covellite (Cu. S), Bornite (Cu 5 Fe. S 4) – Realgar (As. S), Orpiment (As 2 S 3), Cinnabar (Hg. S) – Stibnite (Sb 2 S 3), Molybdenite (Mo. S 2) • Sulfide minerals typically have metallic luster, covalent bonding, and occur in hydrothermal veins. • Only rarely do they occur at the surface.
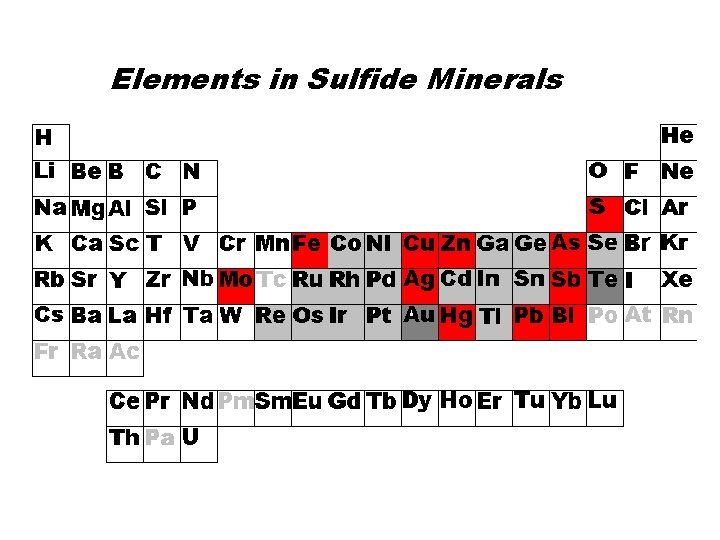
Elements in Sulfide Minerals

Oxides (Single): anion is O (no polyanion) • Hemi-oxide Cuprite (Cu 2 O), Ice (H 2 O) • Monoxides – Periclase group Mg. O, Fe. O, Ca. O, Mn. O – Zincite Zn. O, Bromellite Be. O • Sesquioxides – Corundum Al 2 O 3, Hematite Fe 2 O 3 • Dioxides – Rutile Ti. O 2, Cassiterite Sn. O 2, Pyrolusite Mn. O 2

Oxides (Complex): anion is O • Ilmenite Fe. Ti. O 3 • Spinel Group – Spinel Mg. Al 2 O 4, Magnetite Fe 3 O 4 • Perovskite Ca. Ti. O 3

Hydroxides: anion is OH

Hydroxides: anion is OH • • Brucite Mg(OH)2 Gibbsite Al(OH)3 Diaspore Al. OOH(Bauxite) Goethite Fe. OOH

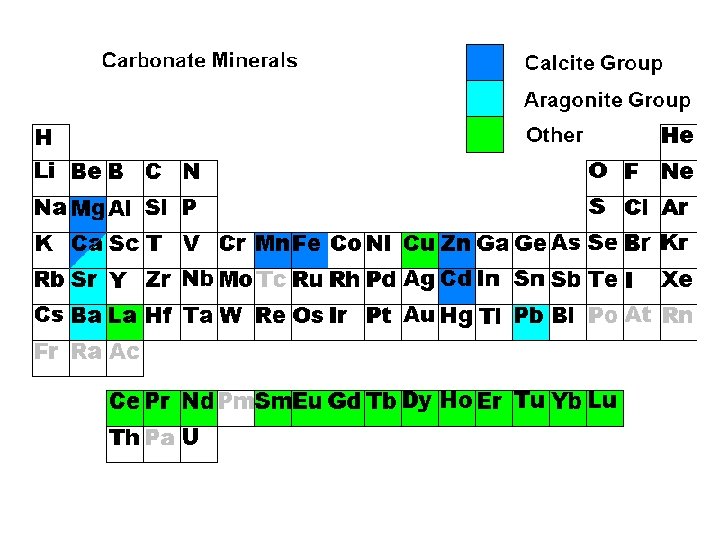
Carbonates 2 anion is (CO 3)

Carbonates 2 anion is (CO 3) • • Calcite Ca. CO 3, Rhodochrosite Mn. CO 3 Aragonite Ca. CO 3, Witherite Ba. CO 3 Dolomite Ca. Mg(CO 3)2 Malachite and Azurite
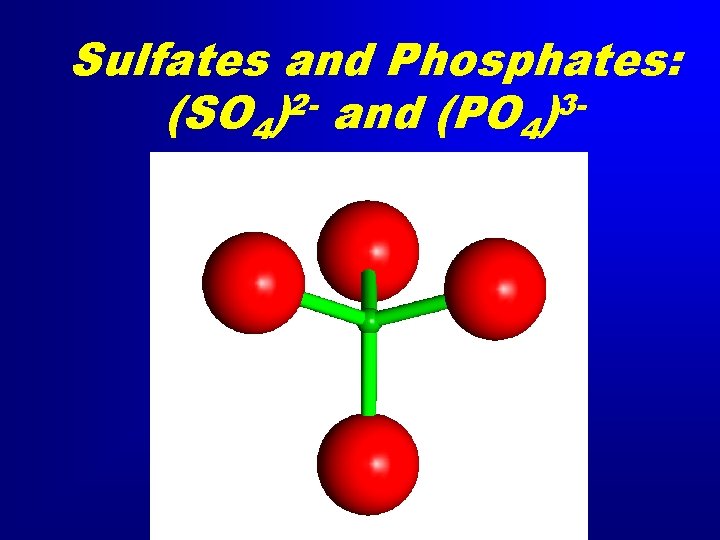
Sulfates and Phosphates: 23(SO 4) and (PO 4)

Sulfates and Phosphates: 23(SO 4) and (PO 4) • Barite (Ba. SO 4), Celestine (Sr. SO 4) • Gypsum Ca. SO 4. 2 H 2 O • Anhydrite Ca. SO 4 • Apatite Ca 5(PO 4)3 OH • Turquoise Cu. Al 6(PO 4)4(OH)8· 4 H 2 O

Silicates are classified by polymerization • Orthosilicates (Si. O 4)4 -: Isolated tetrahedra • Sorosilicates (Si 2 O 7)6 - ‘Bow-Ties’ • Chain silicates – (Si. O 3)2 - : Infinite single chains – (Si 4 O 11)6 - : Double Chains • Sheet Silicates (Si 4 O 10)4 - sheets • Framework Silicates (Si. O 2) framework

Orthosilicates: (Si. O 4)4 • • • Isolated Si. O 4 tetrahedra Olivine Group (Mg 2 Si. O 4) Garnet Group (Mg 3 Al 2 Si 3 O 12) Aluminosilicate Group (Al 2 Si. O 5) Staurolite, Zircon, Titanite

Orthosilicates: Isolated Tetrahedra

Sorosilicates and Cyclosilicates • • Epidote Group Ca 2 Al 2 Fe. Si 3 O 12(OH) Tourmaline Na. Mg 3 Al 5 B 3 Si 6 O 27(OH)4 Beryl Be 3 Al 2 Si 6 O 18 Cordierite (Mg, Fe)2 Al(Al. Si 5)O 18 • n. H 2 O

Chain Silicates • Single Chains – Orthopyroxenes Mg 2 Si 2 O 6 – Clinopyroxenes Ca. Mg. Si 2 O 6 – Pyroxenoids Ca 3 Si 3 O 9 • Double Chains – Amphiboles (Mg, Fe)7 Si 8 O 22(OH)2
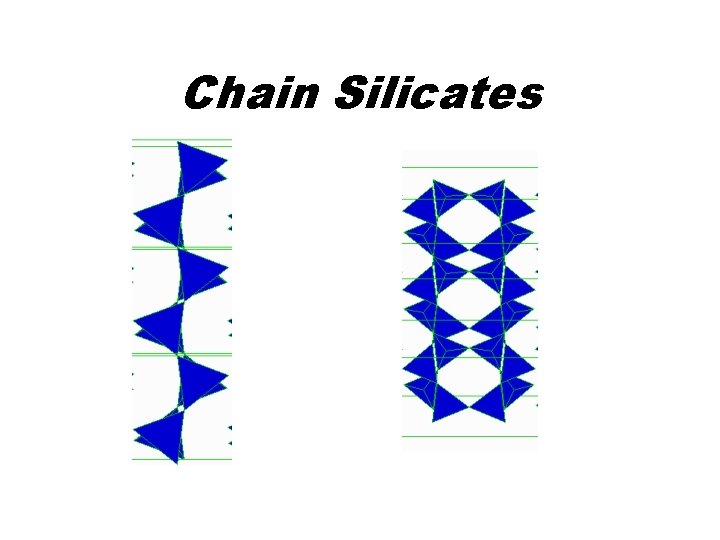
Chain Silicates

Layer Silicates • • Antigorite Talc Kaolinite Pyrophyllite Biotite Muscovite Chlorite Mg 3 Si 2 O 5(OH)4 Mg 3 Si 4 O 10(OH)2 Al 2 Si 2 O 5(OH)4 Al 2 Si 4 O 10(OH)2 K(Mg, Fe)3 Al. Si 3 O 10(OH)2 KAl 2 Al. Si 3 O 10(OH)2 (Mg, Fe)6 Al. Si 3 O 10(OH)2
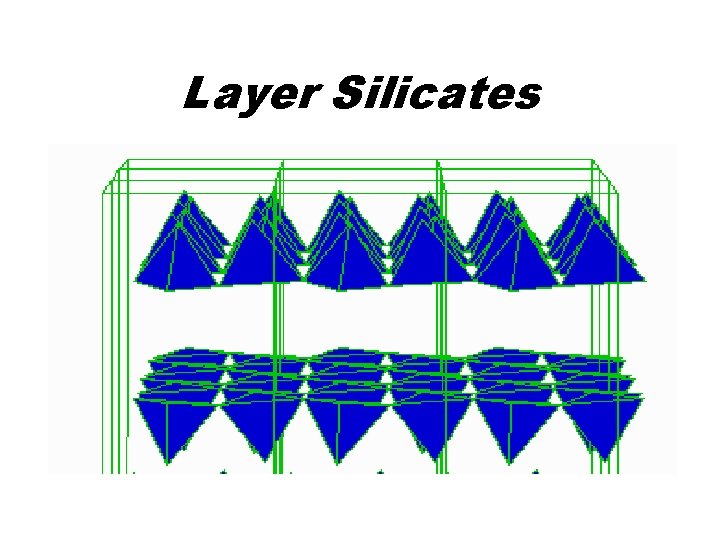
Layer Silicates

Framework Silicates (Tektosilicates) • • • Silica Group Si. O 2 Alkali Feldspar (Na, K)Al. Si 3 O 8 Plagioclase Na. Al. Si 3 O 8 -Ca. Al 2 Si 2 O 8 Feldspathoids (Leucite, Kalsilite, etc) Zeolites (open hydrous frameworks)

Tektosilicates (Framework Silicates)
- Slides: 28