Classification of Matter Breakdown of Matter Matter can

Classification of Matter

Breakdown of Matter

Matter can be classified into… n Pure Substances n A sample of matter, either an element or a compound, that consists of only one component with definite physical and chemical properties and a definite composition.

Pure Substances Elements: n simplest form of pure substance. n They cannot be broken into anything else by physical or chemical means (one type of atom) n Ex: iron (Fe), gold (Au) Compounds: n pure substances that are the unions of two or more elements. n They can be broken into simpler substances ONLY by chemical means n Ex: salt (Na. Cl), water (H 2 O)

Matter can be classified into…. . n Mixtures n two or more substances that are not chemically combined with each other and can be separated by physical means. The substances in a mixture retain their individual properties. Heterogeneous Mixture: not uniform in composition; You can see the different parts; Ex: oil and water, concrete, blood, pizza, sand in water Homogeneous Mixture: also called solution; uniform in composition; no visible parts; Ex: vinegar, clear air, salt water, brass

Physical Separation Techniques n n Filtration- solid part is trapped by filter paper and the liquid part runs through the paper Vaporization- where the liquid portion is evaporated off to leave solid

Physical Separation Techniques n n Decanting- when liquid is poured off after solid has settled to bottom Centrifuge- machine that spins a sample very quickly so that components with different densities will separate

Physical Separation Techniques n Paper Chromatography- used to separate mixtures because different parts move quicker on paper than other

Solid States of Matter Liquid Ø definite volume Ø definite shape Ø atoms are packed together Ø definite volume Ø indefinite shape Ø atoms are close together Ø atoms can overcome attractive forces to flow in fixed positions Ø strong attractive forces between atoms Ø indefinite volume and shape Ø only vibrate in place Ø atoms move quickly Ø atoms are far apart Ø weak attractive forces Ø vapor refers to the gaseous state of a substance that is a solid or liquid at room temperature. Gas

Physical Changes in Matter n n change in a substance that doesn’t change the identity of the substance n Ex. grinding, cutting, melting, boiling Includes all changes of state (physical changes of a substance from one state to another)
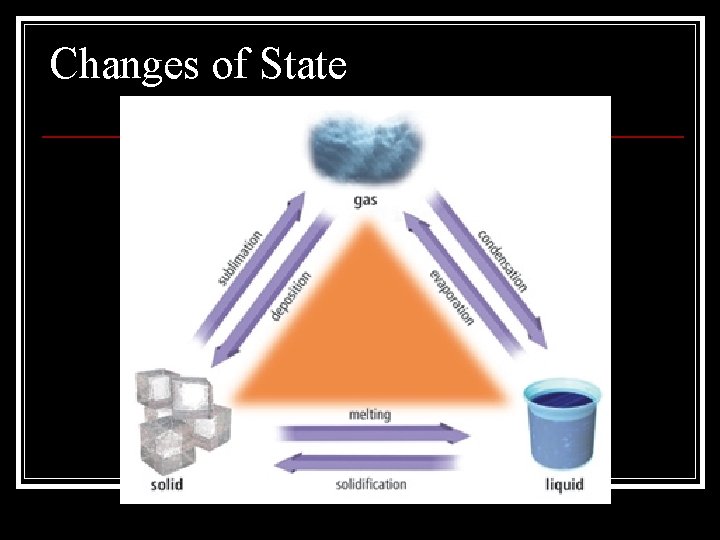
Changes of State

Chemical Changes in Matter n a change in which a substance is converted into a different substance n n Reactants = Products doesn’t change the amount of matter present (According to the law of conservation of mass: Mass is neither created nor destroyed; it is conserved. )
- Slides: 12