Classification of Inorganic Compounds Inorganic Chemistry Non carbon

Classification of Inorganic Compounds Inorganic Chemistry: Non carbon compounds Exceptions: Carbonate and bicarbonate ions Binary ionic Compounds: Contain two elements, one metal and one nonmetal. EX: KCl, Mg. Cl 2, Al. Cl 3, Ternary Ionic Compounds: Contain three elements, at least one metal and one nonmetal Ex: KNO 3, Ca(NO 3)2, Al(NO 3)3, Na 2 CO 3 Binary Molecular Compound: Contain two nonmetals Ex: NH 3, H 2 O, H 2 S

Classification of Inorganic Compounds Aqueous Solution: Is produced when a compound dissolves in water Binary Acid: An aqueous solution of a compound containing hydrogen and one other nonmetal Ex: HCl(aq), HBr(aq), H 2 S(aq) Ternary oxyacid: An aqueous solution of a compound containing hydrogen, a nonmetal, and oxygen Ex: HNO 3, H 2 SO 4, H 3 PO 3
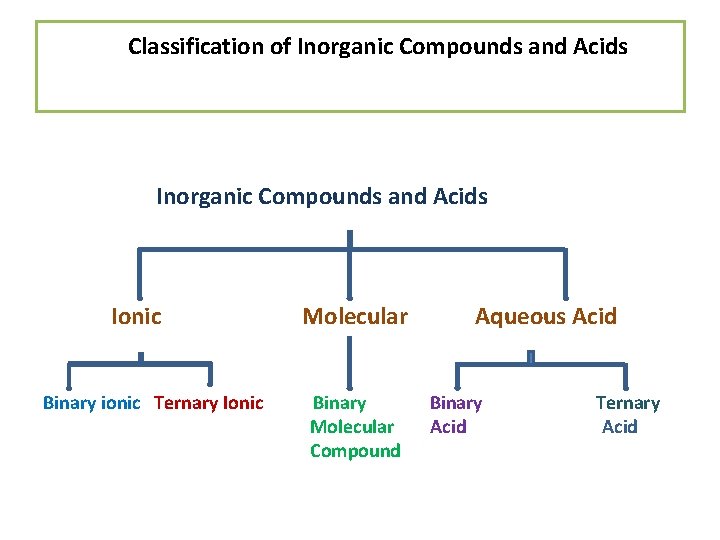
Classification of Inorganic Compounds and Acids Ionic Binary ionic Ternary Ionic Molecular Binary Molecular Compound Aqueous Acid Binary Acid Ternary Acid

Classification of Ionic Compounds and Acids Practice a) carbon disulfide, CS 2 b)Lithium dichromate, Li 2 Cr 2 O 7 c) Magnesium iodide, Mg. Cl 2 d) Nitric acid, HNO 3(aq) e) Hydrochloric acid, HCl(aq) How do you distinguish binary molecule and binary acid ?
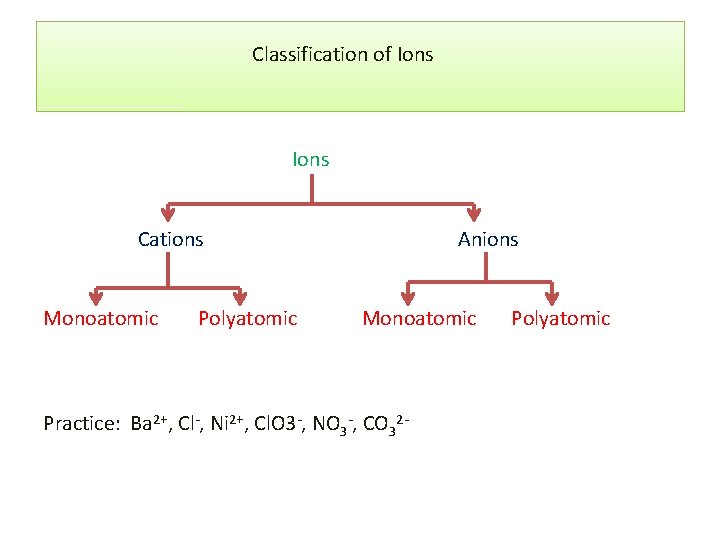
Classification of Ions Cations Monoatomic Polyatomic Anions Monoatomic Practice: Ba 2+, Cl-, Ni 2+, Cl. O 3 -, NO 3 -, CO 32 - Polyatomic

Monoatomic Ions Cations: Metals lose valence electrons to become positively charged cations. Ex: Li+, Na+, Mg 2+, Al 3+ Anions: Nonmetal atoms gain valence electrons and become negatively charged ions called Anions. Ex: Cl-, O 2 -, S 2 -, N 3 Naming Metal ions: 1) Name of parent metal followed by ion 2) Transition metals can form more than on cation (Fe 2+, Fe 3+) In this case, it is necessary to specify the charge. As per IUPAC the cation can be named for the parent metal followed by its charge by Roman numerals in parentheses. Ex: Iron(II) ion for Fe 2+, Copper(II) ion for Cu 2+ 3) Naming transition metals with their charge in parentheses is called stock system. Naming of. Ag+, Zn 2+ and Cd 2+ are not required to follow the Stock system

Naming of Metal cations by Latin system The Latin system of naming involves the Latin name of metal and suffix –ous or –ic If the metal produces two cations, the lower charge ion is received –ous and higher charge ion is received –ic Ex: Latin name of ion is ferrum. Ferrous ion is F 2+ and Ferric ion is Fe 3+ Latin Name for Copper is Cuprum. Cuprous is for Cu+ and Cupric is for Cu 2+ Mercury(I) ion is exception for naming with Latin name (Hydrargyrum is difficult to pronounce) Hg 22+ : Mercurous (Hg+ becomes Hg 2+ for stability reasons) Hg 2+ : Mecuric:
Monoatomic Cations
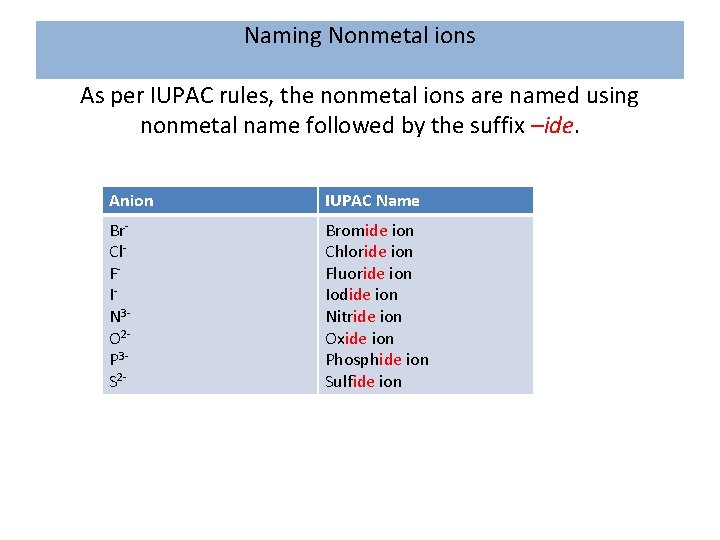
Naming Nonmetal ions As per IUPAC rules, the nonmetal ions are named using nonmetal name followed by the suffix –ide. Anion IUPAC Name Br. Cl. FIN 3 O 2 P 3 S 2 - Bromide ion Chloride ion Fluoride ion Iodide ion Nitride ion Oxide ion Phosphide ion Sulfide ion
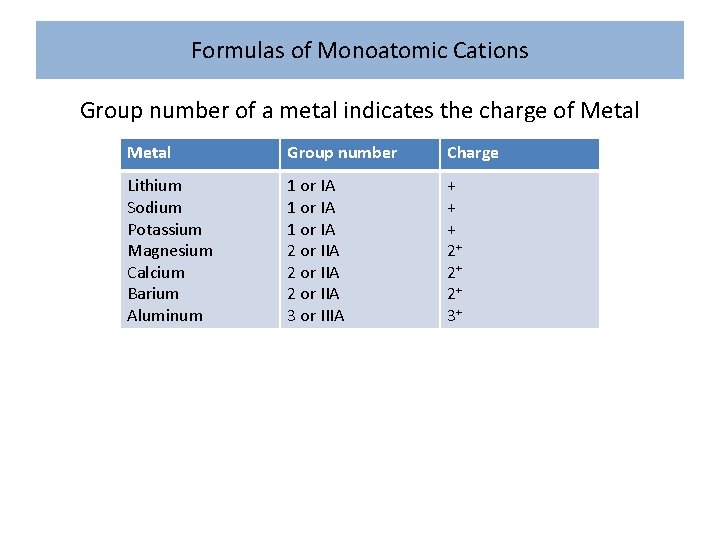
Formulas of Monoatomic Cations Group number of a metal indicates the charge of Metal Group number Charge Lithium Sodium Potassium Magnesium Calcium Barium Aluminum 1 or IA 2 or IIA 3 or IIIA + + + 2+ 2+ 2+ 3+

Formulas of Monoatomic Anions Group VA or 15: Group VIA or 16: Group VIIA or 17: Gains 3 electrons Gains 2 electrons Gains 1 electron N 3 O 2 F- Requirement of electrons is the key to predict the negative charge on nonmetals The elements in Group VA need 3 electrons, Group VIA need 2 electrons, Group VII need 1 one electron to get isoelectronic structure with the nearest noble gas Predict Charge of Barium ion, Cobalt(II) ion, Choride ion, Phosphide ion, Iodide ion

Polyatomic Ions (Anions) 1) Generally one or more elements combine with oxygen to produce ions These oxyions have names ending with –ate Ex: Nitrate ion of NO 33 -, Sulfate for SO 422) Some oxyions have names ending with –ite Ex: Nitrite for NO 22 -, Sulfite for SO 32 The ions with name ending with -ite, will have one less oxygen atom than the corresponding –ate Chlorate ion Cl. O 3 - Chlorite ion Cl. O 2 - (Distinguish between monatomic anion and polyatomic anion)
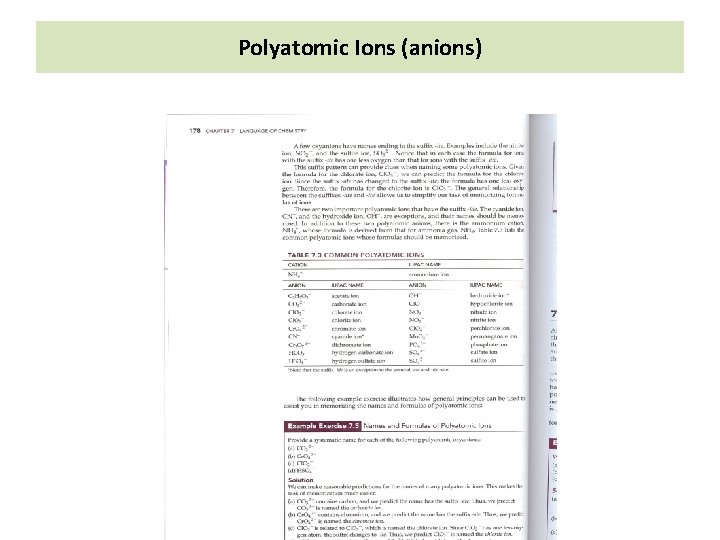
Polyatomic Ions (anions)

Writing Chemical Formulas 1) Ionic compound contains positive and negative ions. Ex: Na. Cl 2) Formula unit is the simplest representative particle of ionic compound 3) Since the formula unit is neutral, the total positive charge must be equal to total negative charge. 4) Sodium Chloride: Na. Cl is composed of Na+ and Cl. Calcium Chloride: Ca 2+, Cl. To have neutral charge, it is necessary to have 2 Cl- ions Therefore, calcium chloride has the formula of Ca. Cl 2 (Ca 2+ and 2 Cl-) Exercise: Copper(I) oxide Cu+ Cadmium oxide Cd 2+ Cobalt(III)oxide Co 3+ Aluminum oxide Al 3+ and O 2 and O 2 -

Formula Units Containing Polyatomic Ions “The rules apply same as in the case of monoatomic ions” The charge of positive ions and negative ions should be equal NH 4+ and SO 42 - : Two NH 4+ ions are needed to give neutral formula. Therefore the formula is (NH 4)2 SO 4 Practice examples: Ca 2+ and CO 32 Ca 2+ and OHCa 2+ and PO 43 -

Binary Ionic Compounds The net charge must be zero to the ionic compound. This principle applies to every ionic compound including Binary ionic compounds. Practice: Determine the charge of iron in Fe 2 O 3, Cu 3 P and Co. N

Naming Binary Ionic Compounds 1) The metal cation is named first followed by nonmetal anion 2) The suffix –ide should be added to the nonmetal anion Mg. O: Magnesium oxide Sn. F 2: Stock system- Tin(II) fluoride, Latin system: Stannous fluoride Practice: Systematic names of Mn 3 P 2 and Fe 2 S 3

Predicting Formulas of Binary Ionic Compounds 1) Use periodic table to predict the formula 2) Group number is the charge of the metal ion (cation) Practice: Sodium chloride, Aluminum oxide Magnesium chloride Aluminum sulfide Gallium oxide

Transition metal in Ternary ionic Compounds 1) Transition metals have variable charges 2) The charge must be determined first before attempting the name Determine the charge and give the correct name for: 1) Fe 3(PO 4)2 2) Hg(OH)2 3) Co(Cl. O 3)3

Naming Ternary Ionic Compounds Name cation first followed by anion Ca. CO 3 - Calcium carbonate Cu. SO 4: Stock system: Copper(II) sulfate Latin System: Cupric sulfate Practice: 1) KMn. O 4, Ca(NO 3)2, Hg(NO 3)2, Ba. Cr. O 4, Cu(NO 2)2 2) Write the formula for Mercury(I) nitrite, iron(III) sulfate 3) Identify binary and ternary ionic compounds Li. Cl, Li. Cl. O 2

Predicting Formulas of Ternary Ionic Compounds 1) With the help of Periodic table, predict the charge of the metal 2) If the anion is familiar, use the charge of the anion to predict the formula 3) Take help from the similar formula. Identify the group of the metal or nonmetal and predict formula Examples: Calcium carbonate is Ca. CO 3. Predict a) Barium carbonate and Calcium silicate. Potassium chlorate is KCl. O 3. Predict Lithium chlorate, Potassium bromate and Sodium bromate

Binary Molecular Compounds 1) Compounds of two nonmetals 2) Molecule is the simplest representative in a binary molecules 3)Order of writing: C, P, N, H, S, I, Br, Cl, O, F. Ex: CH 4, PH 3, NH 3, H 2 S, HI, HBr, HCl, H 2 O, HF

Naming of Binary Molecular Compounds 1) First element named first followed by second element with suffix –ide. 2) The number of atoms of each element will be indicated by Greek prefixes (mono, di, tri etc) The prefix mono is always omitted unless it is necessary EX: CO and CO 2 are named as Carbon monoxide and Carbon dioxide
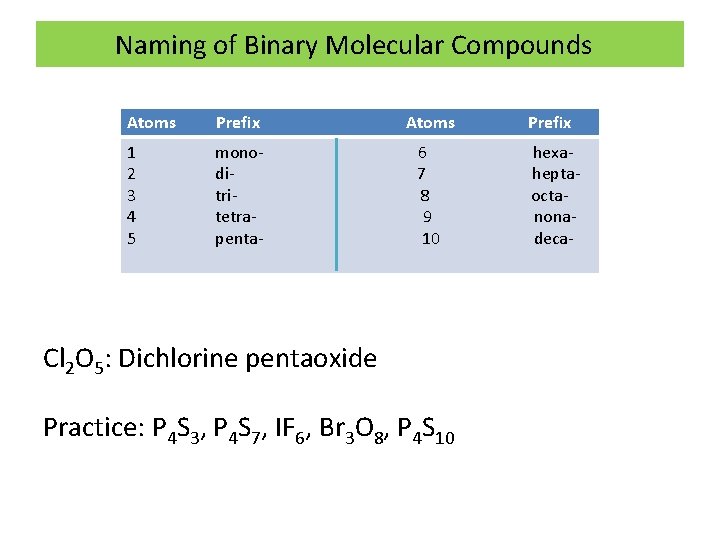
Naming of Binary Molecular Compounds Atoms Prefix Atoms 1 2 3 4 5 monoditritetrapenta- 6 7 8 9 10 Cl 2 O 5: Dichlorine pentaoxide Practice: P 4 S 3, P 4 S 7, IF 6, Br 3 O 8, P 4 S 10 Prefix hexaheptaoctanonadeca-

Binary Acids “Aqueous solution of a compound containing hydrogen and a nonmetal” Ex: HCl, HBr Named by prefix hydro- followed by nonmetal stem and adding the suffix –ic acid Muriatic Acid: Aqueous hydrogen chloride Chemical name is hydrochloric acid, HCl(aq) Practice: HF(aq), HBr(aq), H 2 S(aq)

Ternary Oxyacids “An aqueous solution of a compound containing hydrogen and an oxyanion” Naming: Attach –ic acid or -ous acid to the nonmetal stem HNO 3 (aq) : Nitr + ic acid HNO 2 (aq): Nitr + ous acid SO 42 -: Sulfate (sodium sulfate) SO 32 -: Sulfite (sodium sulfite) H 2 SO 4(aq): Sulfuric acid H 2 SO 3(aq): Sulfurous acid

Ternary acids and related compounds Formula Ternary acid Ternary compound Formula HCl. O 2 HCl. O 3 HCl. O 4 Hypochlorous acid Chloric acid Perchloric acid Sodium hypochlorite Sodium chlorate Sodium perchlorate Na. Cl. O 2 Na. Cl. O 3 Na. Cl. O 4 Exercise: H 3 PO 4(aq), H 3 PO 3(aq), HBr. O 2(aq), HBr. O 3(aq)
- Slides: 27