Classification of carbohydrates Carbohydrates are mainly classified into

Classification of carbohydrates Carbohydrates are mainly classified into four different groups: Monosachharides Disaccharides Oligosaccharides Polysaccharides

MONOSACCHARIDES Monosaccharides are those carbohydrates which Cannot be Hydrolyzed further into more simple carbohydrates. Thus, they are the Simplest form of Carbohydrates. Familiar examples are: Glucose, Fructose, Ribose & Galactose.

Monosaccharides are further classified on the basis of: Aldehyde or Ketone Group: Aldomonosaccharides (Aldoses). Ketomonosaccharides (Ketoses). Carbon Chain Length. Trioses. Tetroses. Pentoses. Hexoses. Heptoses.
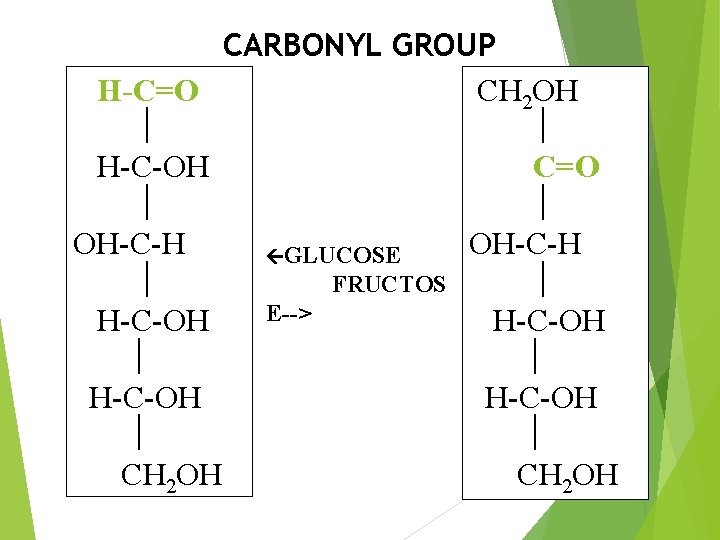
CARBONYL GROUP H-C=O CH 2 OH H-C-OH C=O OH-C-H GLUCOSE FRUCTOS E--> H-C-OH CH 2 OH
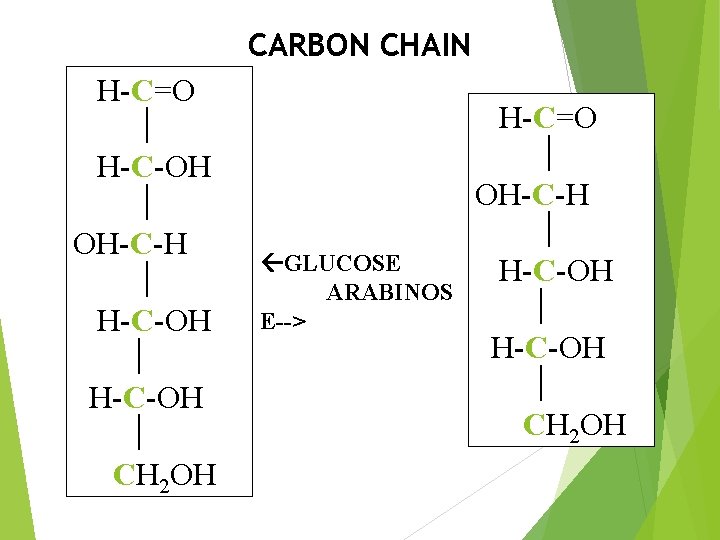
CARBON CHAIN H-C=O H-C-OH OH-C-H H-C-OH CH 2 OH GLUCOSE ARABINOS E--> H-C=O OH-C-H H-C-OH CH 2 OH

EXAMPLES OF MONOSACCHARIDES NO. OF CARBON ATOMS ALDO KETO 3 C TRIOSE GLYCERALDEHYDE DIHYDROXYACETONE 4 C TETROSE ERYTHRULOSE 5 C PENTOSE RIBOSE, XYLOSE RIBULOSE, XYLULLOSE 6 C HEXOSE GLUCOSE, GALACTOSE, MANNOSE FRUCTOSE
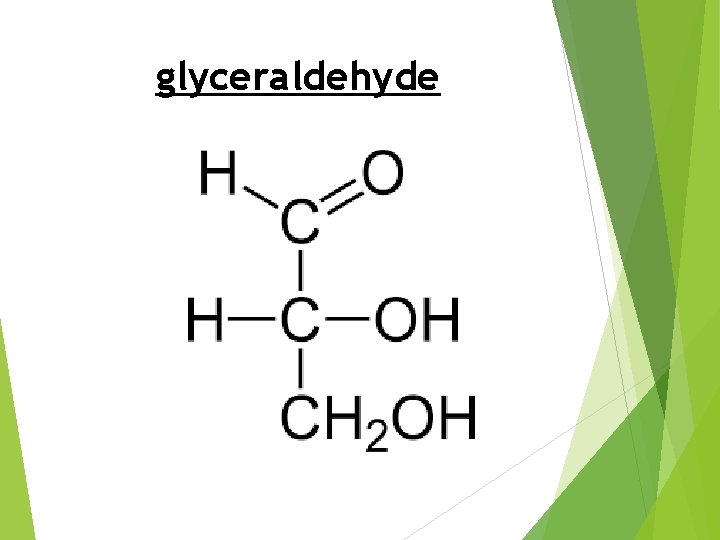
glyceraldehyde

GLYCERALDEHYDE IS THE SIMPLEST MONOSACCHARIDE(PARTICULARLY ALDOSES) IT CANNOT BE HYDROLYZED FURTHER. IT IS THE SMALLEST POSSIBLE STRUCTURE THAT IS A CARBOHYDRATE CANNOT HAVE LESS THAN 3 CARBON ATOMS. GENERAL FORMULA OF MONOSACCHARIDE: (C • H 2 O)N (WHERE N IS ANY NUMBER OF THREE OR GREATER)

isomerism Isomers are basically molecules that have the same chemical formula but they differ in their chemical structures. Asymmetric Carbon is an important determinant of Isomerism. Asymmetric Carbon is that Carbon which is attached with four different groups.
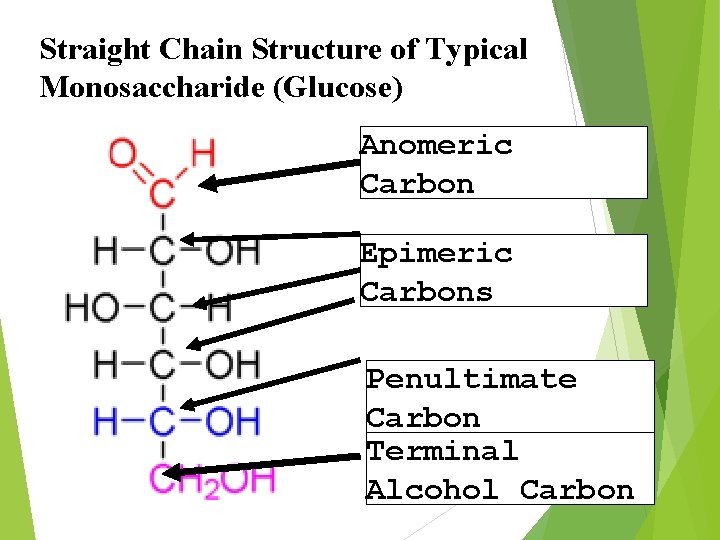
Straight Chain Structure of Typical Monosaccharide (Glucose) Anomeric Carbon Epimeric Carbons Penultimate Carbon Terminal Alcohol Carbon
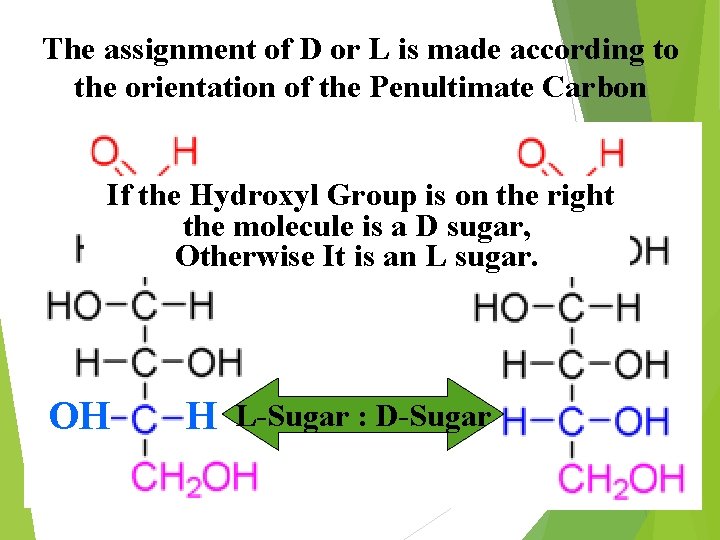
The assignment of D or L is made according to the orientation of the Penultimate Carbon If the Hydroxyl Group is on the right the molecule is a D sugar, Otherwise It is an L sugar. OH H L-Sugar : D-Sugar

D and L isomerism produce mirror images
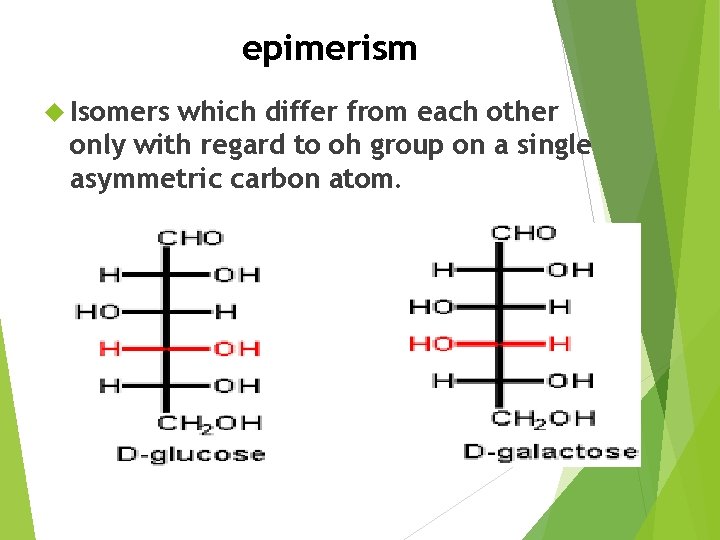
epimerism Isomers which differ from each other only with regard to oh group on a single asymmetric carbon atom.

Optical isomers When a beam of polarized light is passed through sugars, they will be rotated either towards right or left. Right Rotation (dextrorotatory) E. g. DGlucose Left Rotation (levorotatory) E. g. DFructose

Monosaccharide Derivatives of Biological Importance Oxidation Products (Sugar Acids) Reduction Products (Sugar Alcohols). Amino Sugars. Phosphates.

Oxidation Products (Sugar Acids) When oxidized under proper conditions, Aldoses may form three types of acids (Sugar Acids): Uronic Acids. Aldonic acids. Saccharic Type Acids. of sugar Acid produced depends on which carbon is oxidized.
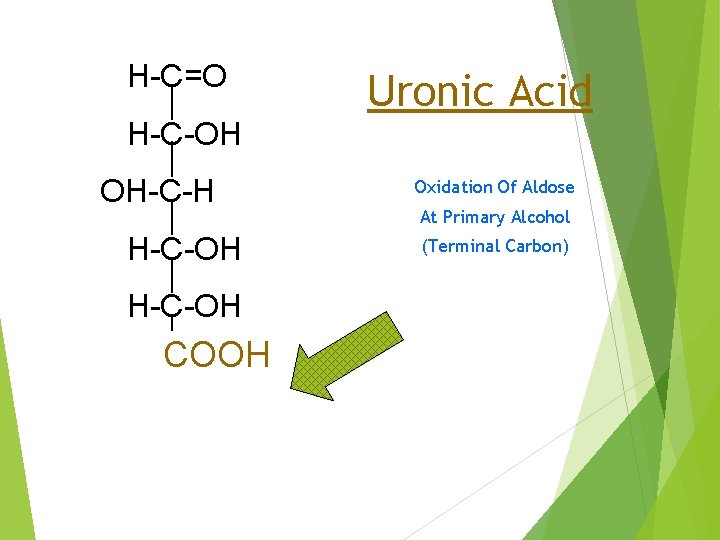
H-C=O H-C-OH OH-C-H H-C-OH COOH CH 2 OH Uronic Acid Oxidation Of Aldose At Primary Alcohol (Terminal Carbon)
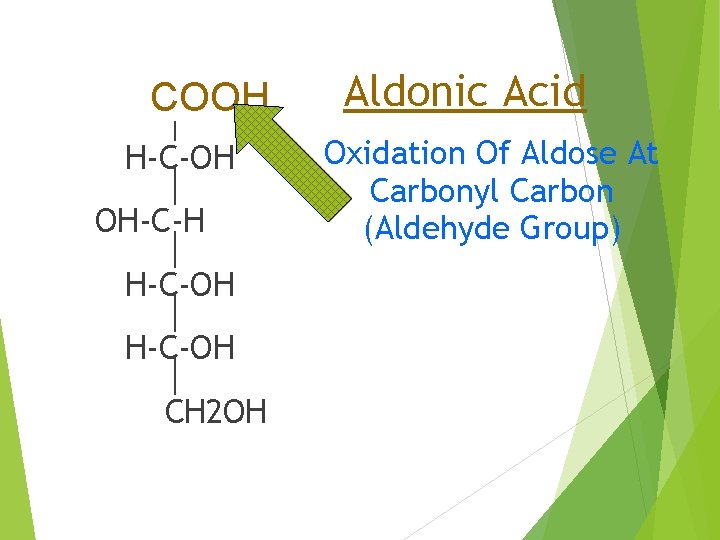
H-C=O COOH H-C-OH OH-C-H H-C-OH CH 2 OH Aldonic Acid Oxidation Of Aldose At Carbonyl Carbon (Aldehyde Group)
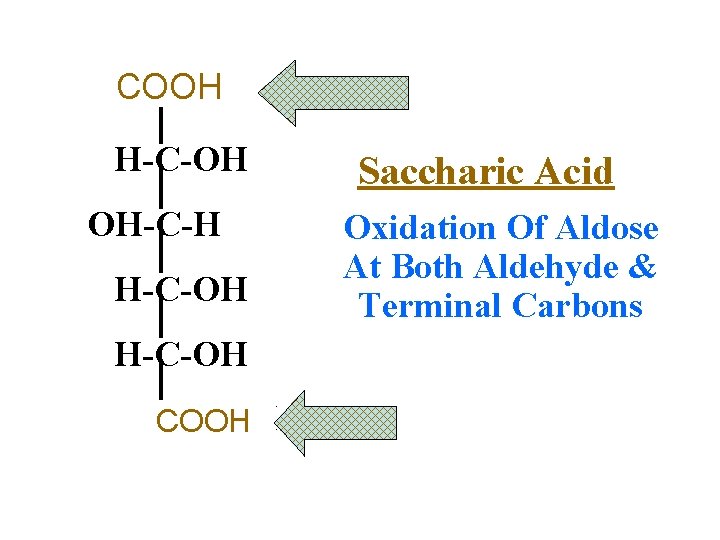
COOH H-C=O H-C-OH Saccharic Acid OH-C-H Oxidation Of Aldose At Both Aldehyde & H-C-OH Terminal Carbons H-C-OH CH 2 OH COOH

Examples of Aldoses with their Corresponding Uronic Acids are: Sugar Glucose ------Mannose ------Galactose ------- Uronic Acids Glucuronic acid, Iduronic acid -----------Mannuronic acid -----------Galacturonic acid ------------

Examples of Aldoses with their Corresponding Aldonic Acids are: Sugar Aldonic Acids Glucose ------Mannose ------Galactose ------- Gluconic acid -----------Mannonic acid -----------Galactonic acid ------------

Amino Sugar: H-C=O NH 2 H-C-OH OH-C-H H-C-OH CH 2 OH Substitution of Amino Group for a Hydroxyl Group of a Sugar Results in the Formation of an Amino Sugar. The Amino Group is attached to Carbon 2.

Common Examples of Amino Sugars (Aminosaccharides) Galactosamine Glucosamine

Sugar Alcohols; Reduction Products Sugar Alcohols are obtained by reduction of Monosaccharides (Both Aldoses and Ketoses) and disaccharides. Sugar alcohols, also known as Polyols, Polyhydric Alcohols, or Polyalcohols.
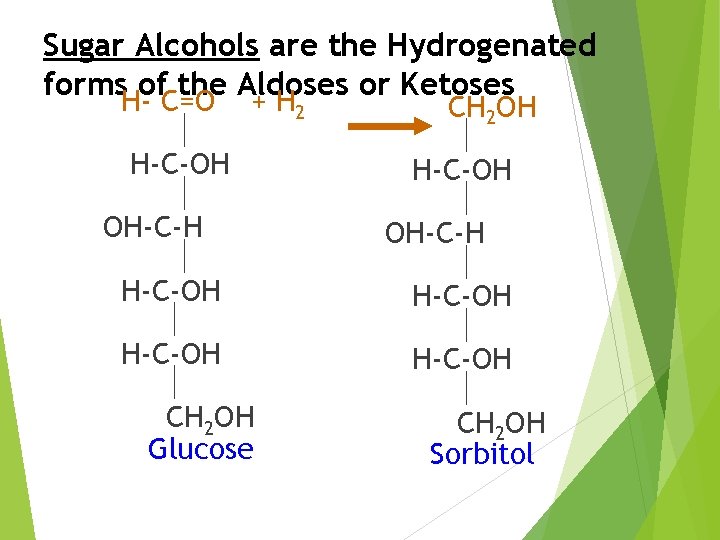
Sugar Alcohols are the Hydrogenated forms of the Aldoses or Ketoses H- C=O + H 2 H-C-OH OH-C-H H-C-OH CH 2 OH Glucose CH 2 OH H-C-OH OH-C-H H-C-OH CH 2 OH Sorbitol

Sugars with their Corresponding Alcohols Glyceraldehyde & Dihydroxyacetone Glucose Mannose Fructose Galactose Ribose Erythrose Xylose Lactose Maltose Glycerol Sorbitol (Glucitol) Mannitol Sorbitol & Mannitol Dulcitol Ribitol Erythritol Xylitol Lactitol Maltitol

Sugar Phosphates Phosphate Esters formed by various Monosaccharides are of Great Importance in Metabolic Reactions. Sugar phosphates of biological importance. Ribose and Deoxyribose in Nucleic Acids are Phosphate Esters of these monosaccharides. Phosphates of Glucose, Fructose and Glyceraldehyde etc. are important intermediate compounds in carbohydrate metabolism of almost all the cells.

Disaccharides Two Joined Monosaccharides. Sucrose: Glucose + Fructose Maltose: Glucose + Glucose Lactose: Glucose + Galactose

Oligosaccharides Composed Of: Three To Ten Monosaccharide Units. E. g. Fructooligosaccharides

Polysaccharides Larger Than Ten Monosaccharide Units. Can Reach Many Thousands Of Units. Homopolysaccharides. Heteropolysaccharides.

Homopolysaccharides Similar Types of Monosaccharide Units. Starch Glycogen Cellulose

Heteropolysaccharides Different Types of Monosaccharide Units. Mucopolysaccharides (Glycosaminoglycans that contain galactose and amine sugars) Agar (Contains various sugars like Glucose, Galactose)
- Slides: 32