classical mechanics Newton Sir Isaac Newton 25 December

양자역학의 배경 ü 고전역학 (classical mechanics, Newton 역학)의 종말 Sir Isaac Newton (25 December 1642 – 20 March 1726/7) Philosophiæ Naturalis Principia Mathematica ("Mathematical Principles of Natural Philosophy"), first published in 1687 우리가 잘못 알고 있는 물리 상식 바로 가기
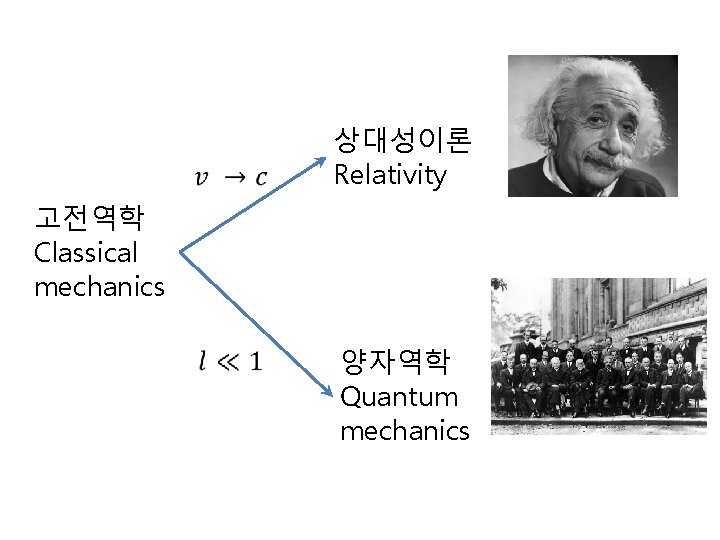
상대성이론 Relativity 고전역학 Classical mechanics 양자역학 Quantum mechanics

Atom, a-tom Astro Boy (Tetsuwan Atomu, "Mighty Atom" "Iron Arm Atom") first published in 1952 and television program first broadcast in 1963, by Osamu Tezuka (1928 -1989). The name atom comes from the Greek ἄτομος (atomos, "indivisible") from ἀ- (a-, "not") and τέμνω (temnō, "I cut").

Democritus (Δημόκριτος, Dēmokritos, "chosen of the people") (ca. 460 BC – ca. 370 BC) was an Ancient Greek philosopher born in Abdera, Thrace, Greece. He was an influential pre-Socratic philosopher and pupil of Leucippus, who formulated an atomic theory for the universe. Pythagoras of Samos ( Πυθαγόρας ὁ Σάμιος [Πυθαγόρης in Ionian Greek] Pythagóras ho Sámios "Pythagoras the Samian", or simply Πυθαγόρας; about 570 – about 495 BC) was an Ionian Greek philosopher, mathematician, and founder of the religious movement called Pythagoreanism.

Thales of Miletus (Θαλῆς ὁ Μιλήσιος c. 624 BC–c. 546 BC) was a pre-Socratic Greek philosopher from Miletus in Asia Minor, and one of the Seven Sages of Greece. Many, most notably Aristotle, regard him as the first philosopher in the Greek tradition. According to Bertrand Russell, "Western philosophy begins with Thales. “He was also the first to define general principles and set forth hypotheses, and as a result has been dubbed the "Father of Science", though it is argued that Democritus is actually more deserving of this title. Thales' most famous belief was his cosmological thesis, which held that the world started from water. Aristotle considered this belief roughly equivalent to the later ideas of Anaximenes, who held that everything in the world was composed of air. The Ancient Greek aphorism "Know thyself", (γνῶθι σεαυτόν, English phonetics pronunciation: gnōthi seauton (also. . . σαυτόν. . . sauton with the ε contracted), was inscribed in the pronaos (forecourt) of the Temple of Apollo at Delphi.

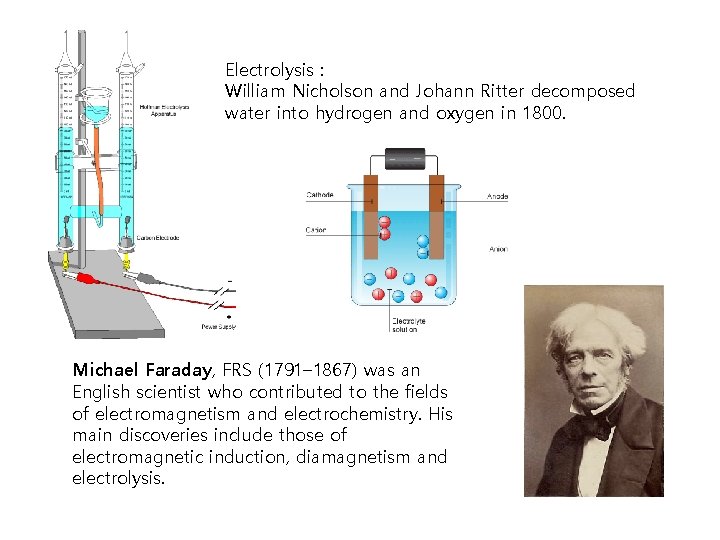
Electrolysis : William Nicholson and Johann Ritter decomposed water into hydrogen and oxygen in 1800. Michael Faraday, FRS (1791– 1867) was an English scientist who contributed to the fields of electromagnetism and electrochemistry. His main discoveries include those of electromagnetic induction, diamagnetism and electrolysis.

John Dalton FRS (1766 -1844) was an English chemist, meteorologist and physicist. He is best known for his pioneering work in the development of modern atomic theory, and his research into colour blindness (sometimes referred to as Daltonism, in his honour). Five main points of Dalton's atomic theory 1. Elements are made of extremely small particles called atoms. 2. Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. 3. Atoms cannot be subdivided, created, or destroyed. 4. Atoms of different elements combine in simple whole-number ratios to form chemical compounds. 5. In chemical reactions, atoms are combined, separated, or rearranged.

Dmitri Ivanovich Mendeleev ( Дми трий Ива нович Менделе eв, 1834 – 1907) was a Russian chemist and inventor. He is credited as being the creator of the first version of the periodic table of elements in 1869. Using the table, he predicted the properties of elements yet to be discovered.
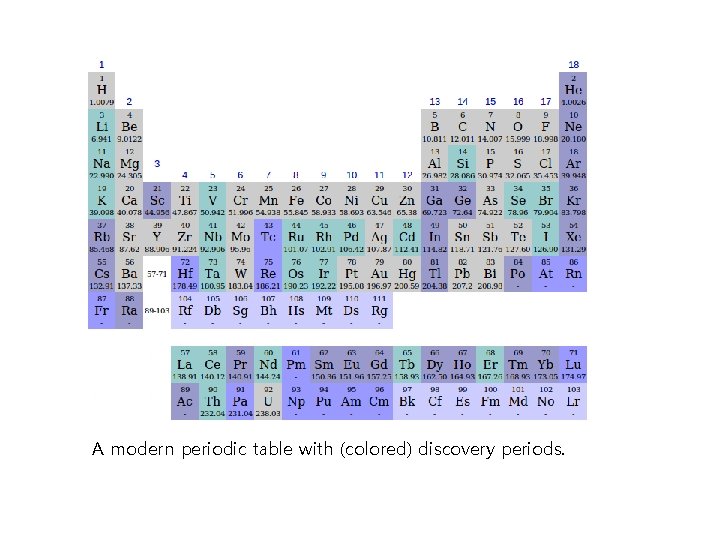
A modern periodic table with (colored) discovery periods.

Discovery of electron Electron was discovered in 1897 by Sir Joseph John "J. J. " Thomson, (1856 -1940), 1906 Nobel laureate in physics. In 1896, the British physicist J. J. Thomson, with his colleagues John S. Townsend and H. A. Wilson, performed experiments indicating that cathode rays really were unique particles, rather than waves, atoms or molecules as was believed earlier. Thomson made good estimates of both the charge e and the mass m, finding that cathode ray particles, which he called "corpuscles, " had perhaps one thousandth of the mass of the least massive ion known: hydrogen. He showed that their charge to mass ratio, e/m, was independent of cathode material. He further showed that the negatively charged particles produced by radioactive materials, by heated materials and by illuminated materials were universal. The name electron was again proposed for these particles by the Irish physicist George F. Fitzgerald, and the name has since gained universal acceptance. Both electric and electricity are derived from the Latin ēlectrum (also the root of the alloy of the same name), which came from the Greek word for amber, ἤλεκτρον (ēlektron).

A Crookes tube is an early experimental electrical discharge tube, invented by English physicist William Crookes and others around 1869 -1875, in which cathode rays, streams of electrons, were discovered. Crookes and his glowing tubes gained notoriety, as shown by this 1902 caricature in Vanity Fair. The caption read "ubi Crookes ibi lux", which in Latin means roughly, "Where there is Crookes, there is light”. Maltese (Amalfi) cross Crookes railway (paddlewheel) tube
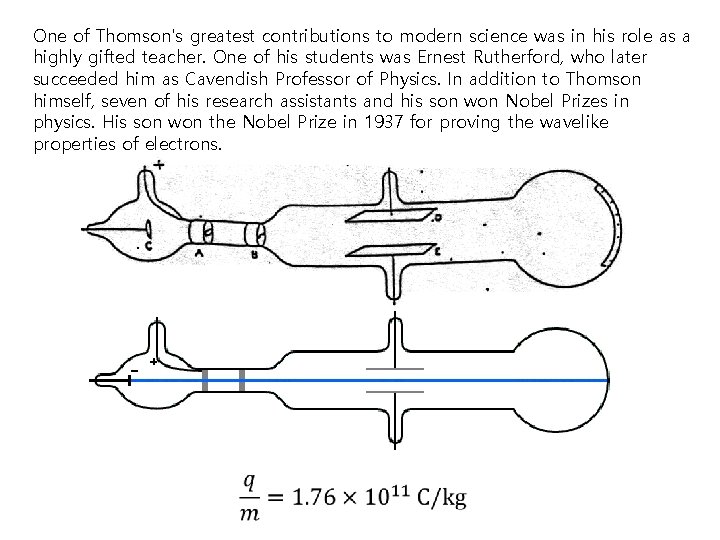
One of Thomson's greatest contributions to modern science was in his role as a highly gifted teacher. One of his students was Ernest Rutherford, who later succeeded him as Cavendish Professor of Physics. In addition to Thomson himself, seven of his research assistants and his son won Nobel Prizes in physics. His son won the Nobel Prize in 1937 for proving the wavelike properties of electrons.

Robert A. Millikan (March 22, 1868 – December 19, 1953) was an American experimental physicist honored with the Nobel Prize for Physics in 1923 for his measurement of the elementary electronic charge (1909) and for his work on the photoelectric effect.

Discovery of nucleus Atom is made of nucleus and electrons ! Atomic Nucleus was discovered in 1911 as a result of Ernest Rutherford's interpretation of the famous 1909 Rutherford experiment performed by Hans Geiger and Ernest Marsden, under the direction of Rutherford was awarded Nobel Prize in Chemistry in 1908 "for his investigations into the disintegration of the elements, and the chemistry of radioactive substances". . Top: Expected results: alpha particles passing through the plum pudding model of the atom undisturbed. Bottom: Observed results: a small portion of the particles were deflected, indicating a small, concentrated charge. Note that the image is not to scale; in reality the nucleus is vastly smaller than the electron shell.
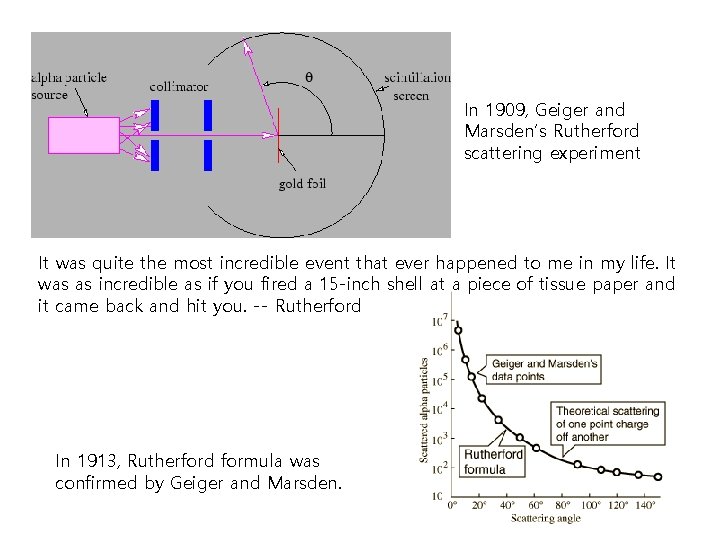
In 1909, Geiger and Marsden’s Rutherford scattering experiment It was quite the most incredible event that ever happened to me in my life. It was as incredible as if you fired a 15 -inch shell at a piece of tissue paper and it came back and hit you. -- Rutherford In 1913, Rutherford formula was confirmed by Geiger and Marsden.

Nucleus is made of protons and neutrons !! Proton : theorized by William Prout (1815), discovered by Ernest Rutherford (1917 -1919), named by him (1920) Neutron : theorized by Ernest Rutherford (1920), discovered by James Chadwick (1932) And the story goes on !!!

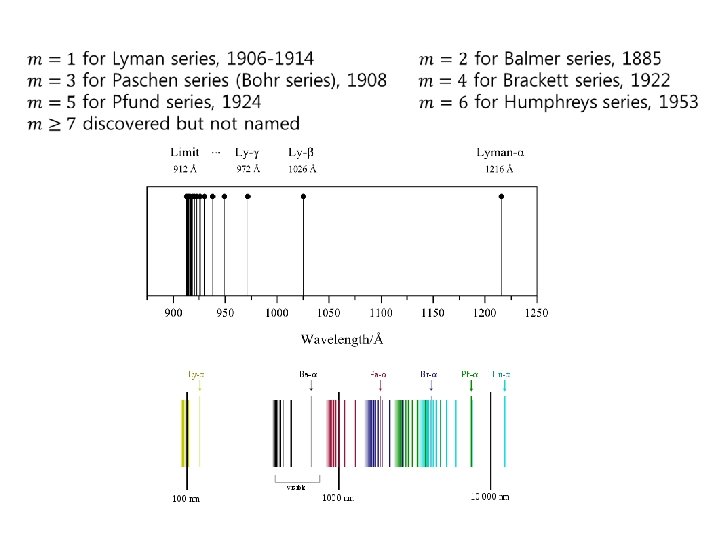
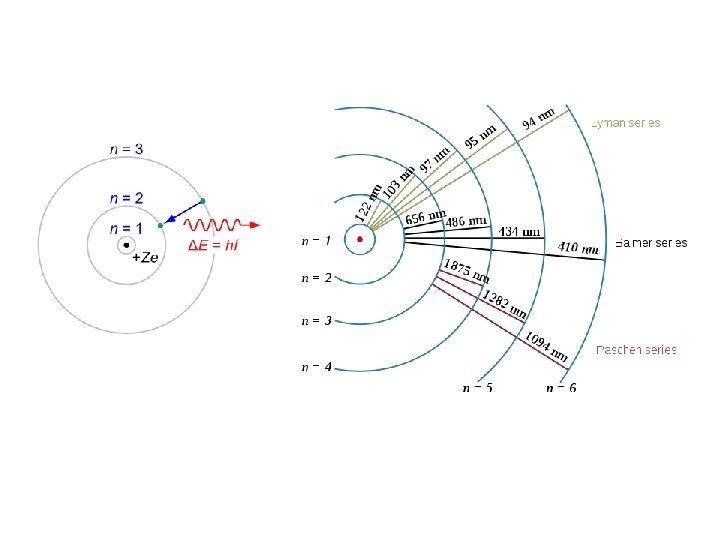
Hydrogen line spectra Balmer referred to the Balmer constant as the "fundamental number of hydrogen. " Johann Jakob Balmer (May 1, 1825 – March 12, 1898) was a Swiss mathematician and mathematical physicist. He taught at a school for girls in Basel. He also lectured at the University of Basel. His major contribution (made at the age of sixty, in 1885) was an empirical formula for the visible spectral lines of the hydrogen atom

Rydberg formula Johannes (Janne) Robert Rydberg (November 8, 1854 – December 28, 1919) was a Swedish physicist mainly known for devising the Rydberg formula, in 1888.

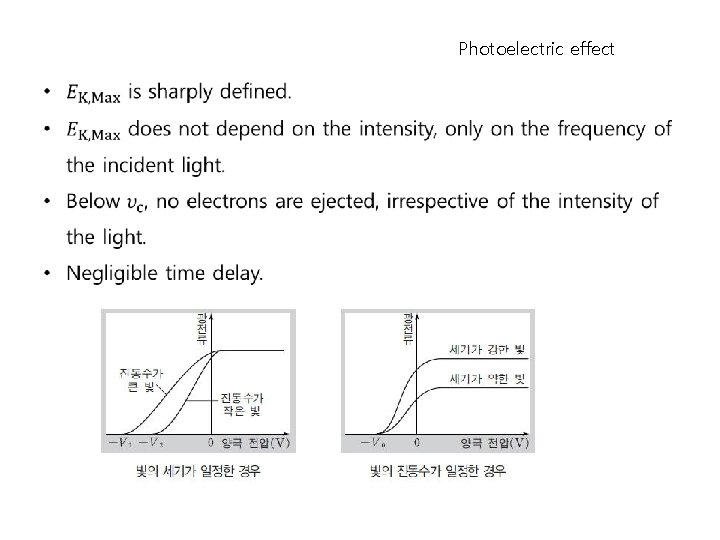
Photoelectric effect
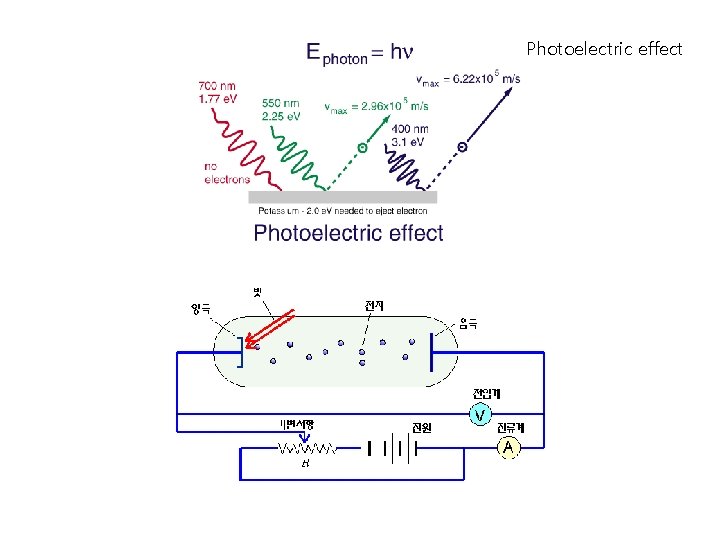
Photoelectric effect
Photoelectric effect
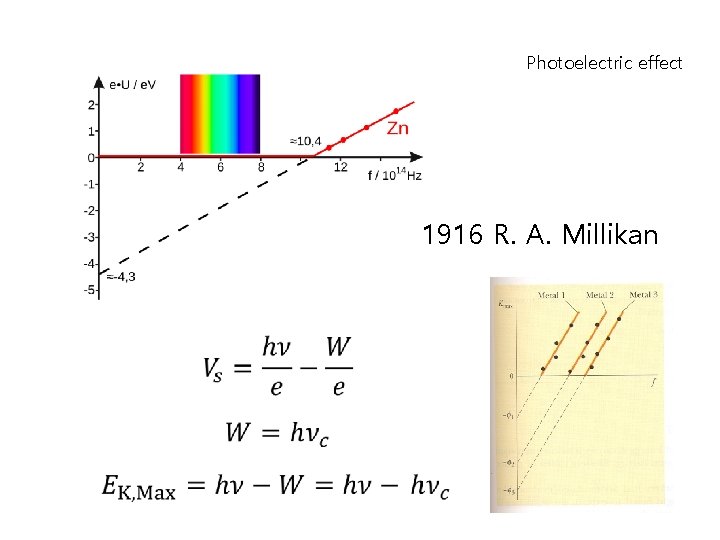
Photoelectric effect 1916 R. A. Millikan
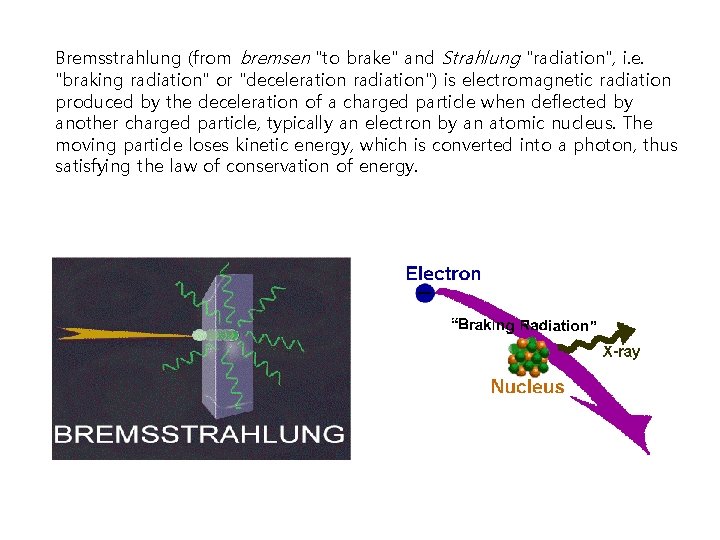
Bremsstrahlung (from bremsen "to brake" and Strahlung "radiation", i. e. "braking radiation" or "deceleration radiation") is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typically an electron by an atomic nucleus. The moving particle loses kinetic energy, which is converted into a photon, thus satisfying the law of conservation of energy.
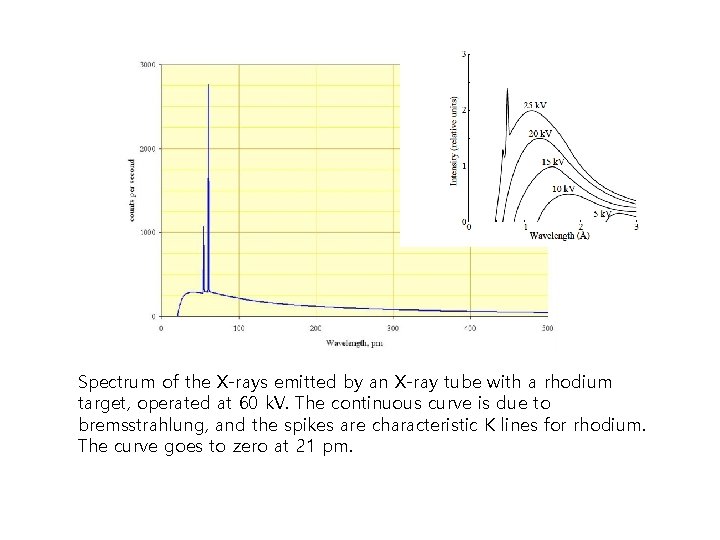
Spectrum of the X-rays emitted by an X-ray tube with a rhodium target, operated at 60 k. V. The continuous curve is due to bremsstrahlung, and the spikes are characteristic K lines for rhodium. The curve goes to zero at 21 pm.
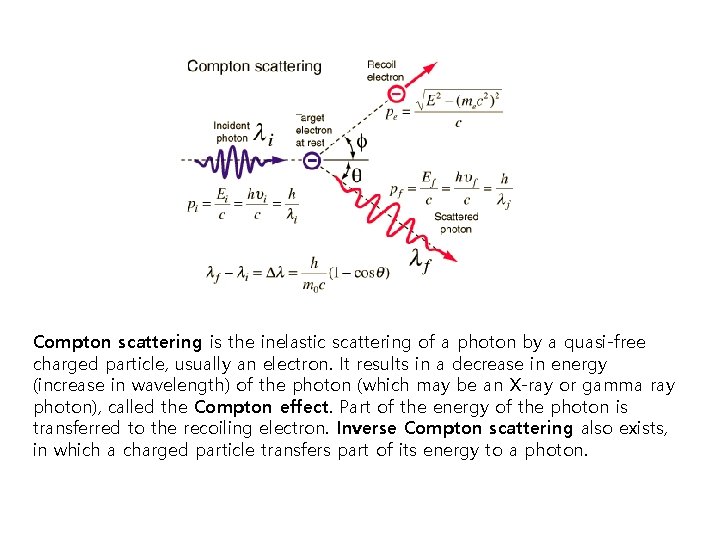
Compton scattering is the inelastic scattering of a photon by a quasi-free charged particle, usually an electron. It results in a decrease in energy (increase in wavelength) of the photon (which may be an X-ray or gamma ray photon), called the Compton effect. Part of the energy of the photon is transferred to the recoiling electron. Inverse Compton scattering also exists, in which a charged particle transfers part of its energy to a photon.

Arthur Holly Compton (September 10, 1892 – March 15, 1962) was an American physicist who won the Nobel Prize in Physics in 1927 for his discovery of the Compton effect, which demonstrated the particle nature of electromagnetic radiation. It was a sensational discovery at the time: the wave nature of light had been well-demonstrated, but the idea that light had both wave and particle properties was not easily accepted. He is also known for his leadership of the Manhattan Project's Metallurgical Laboratory, and served as Chancellor of Washington University in St. Louis from 1945 to 1953. 1936년 1월 13일 타임지 표지의 아서 콤프턴

Bohr atom Rutherford-Bohr atom Niels Henrik David Bohr (1885 -1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum mechanics, for which he received the Nobel Prize in Physics in 1922. He was part of the British team of physicists working on the Manhattan Project. One of his sons, Aage Bohr, grew up to be an important physicist who in 1975 also received the Nobel Prize. Bohr has been described as one of the most influential scientists of the 20 th century. As a post-doctoral student, Bohr first conducted experiments under J. J. Thomson, of Trinity College, Cambridge and Cavendish Laboratory. In 1912 he met and later joined Ernest Rutherford at Manchester University, where on and off he spent four fruitful years in association with the older physics professor.

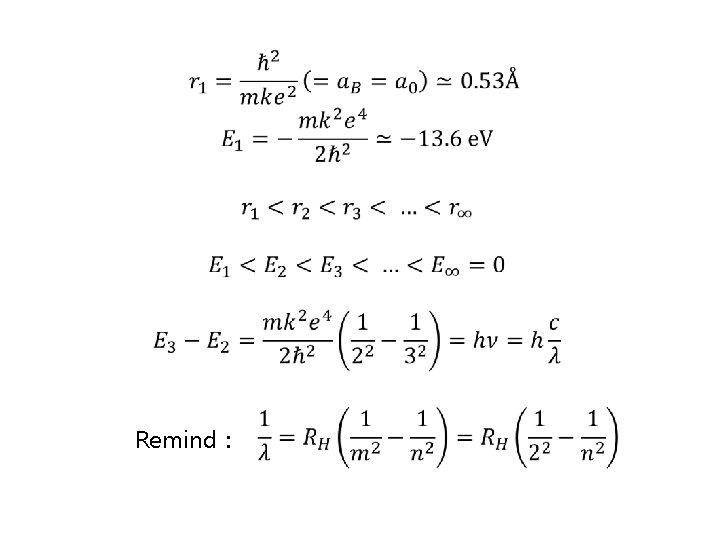
Remind :


Otto Stern (17 February 1888 – 17 August 1969) was a German physicist and Nobel laureate in physics. As an experimental physicist Stern contributed to the discovery of spin quantization in the Stern–Gerlach experiment with Walther Gerlach in February 1922 at the Physikalischer Verein in Frankfurt am Main. He was awarded the 1943 Nobel Prize in Physics, the first to be awarded since 1939. He was the sole recipient in Physics that year, and the award citation omitted mention of the Stern–Gerlach experiment, as Gerlach had remained active in Nazi-led Germany.

Max Theodor Felix von Laue (9 October 1879 – 24 April 1960) was a German physicist who won the Nobel Prize in Physics in 1914 for his discovery of the diffraction of X-rays by crystals. In addition to his scientific endeavors with contributions in optics, crystallography, quantum theory, superconductivity, and theory of relativity, he had a number of administrative positions which advanced and guided German scientific research and development during four decades. A strong objector to National Socialism, he was instrumental in re-establishing and organizing German science after World War II.
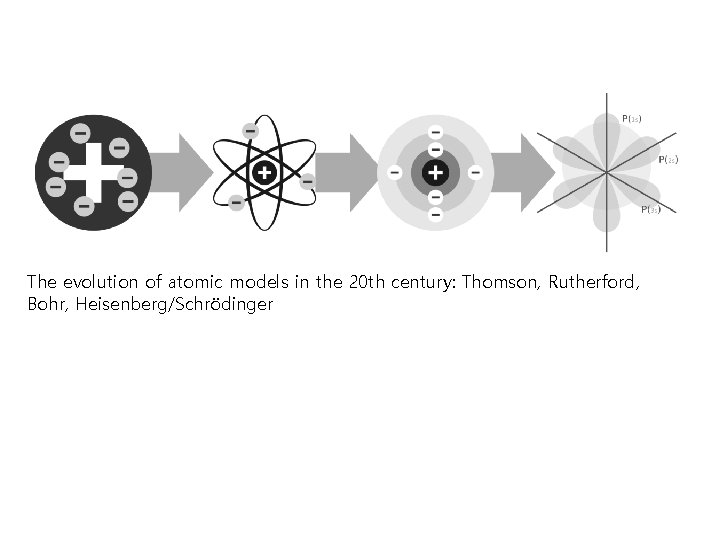
The evolution of atomic models in the 20 th century: Thomson, Rutherford, Bohr, Heisenberg/Schrödinger
- Slides: 36