Cl Chloride HF Hydroflouric acid HBr Hydrobromic acid



�Cl � Chloride HF = Hydroflouric acid HBr = Hydrobromic acid HI = Hydroiodic acid �HCl � Hydro-chlor-ic acid

�Cl. O 4 – � Perchlorate �HCl. O 4 � Perchlor-ic acid

�Cl. O 3 - � Chlorate �HCl. O 3 � Chlor-ic acid

�Cl. O 2 - � Chlorite �HCl. O 2 � Chlor-ous acid

�Cl. O � Hypochlorite �HCl. O � Hypo-chlor-ous acid

�NO 3 - � Nitrate �HNO 3 � Nitric acid

�NO 2 - � Nitrite �HNO 2 � Nitrous acid

�SO 4 2 - � Sulfate �H 2 SO 4 � Sulfuric acid

�SO 3 2 - � Sulfite �H 2 SO 3 � Sulfurous acid

�PO 4 3 - � Phosphate �H 3 PO 4 � Phosphoric acid

�PO 3 3 - � Phosphite �H 3 PO 3 � Phosphorous acid
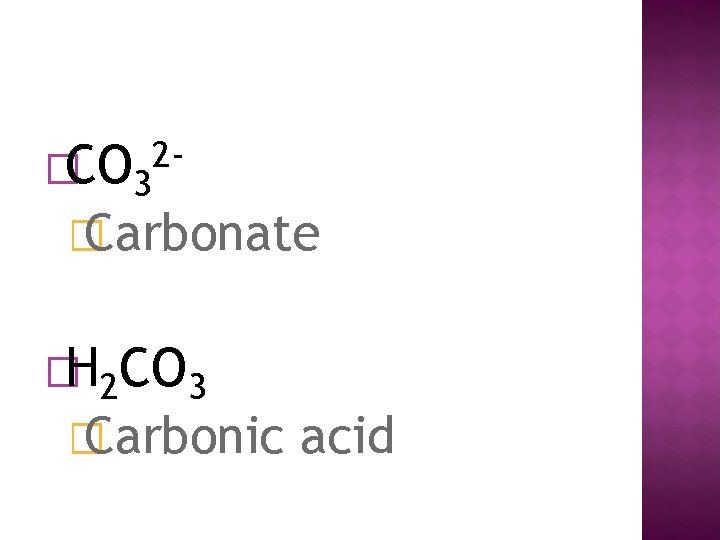
�CO 3 2 - � Carbonate �H 2 CO 3 � Carbonic acid

�Cr. O 4 2 - � Chromate �H 2 Cr. O 4 � Chromic acid

�Cr 2 O 7 2 - � Dichromate �H 2 Cr 2 O 7 � Dichromic acid

�Mn. O 4 - � Permanganate �HMn. O 4 � Permanganic acid

�SCN � Thiocyanate �HCN � Thiocyanic acid

Treated like a Standard Ion �CN � Cyanide �HCN � Hydrocyanic acid
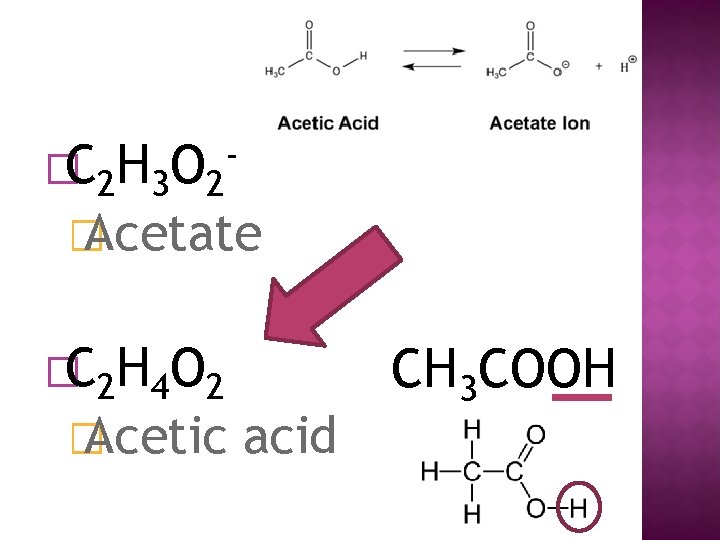
�C 2 H 3 O 2 - � Acetate �C 2 H 4 O 2 � Acetic acid CH 3 COOH

�OH Li. OH = Lithium hydroxide � Hydroxide KOH = Potassium hydroxide Rb. OH = Rubidium hydroxide Cs. OH = Cesium hydroxide �Na. OH � Sodium Ca(OH)2 = Calcium hydroxide

�NH 2 - � Amide �NH 3 � Ammonia �NH 4 + � Ammonium

�Completely dissociate �Group I and II � Hydroxylated �Cation + OH�Li. OH, etc. �Mg(OH)2, etc.

�Completely dissociate �Strongly EN anions � Protonated �H+ HF is an exception. It + Anion is NOT as strong acid. �HCl, HBr, HI �HCl. O 4, HNO 3, H 2 SO 4
- Slides: 24