Circular Dichroism Optical Rotatory Dispersion Compiled by Dr

Circular Dichroism & Optical Rotatory Dispersion Compiled by Dr. A. John Maria Xavier Department of Chemistry Loyola College Chennai

Introduction Chiral structure can be distinguished/ characterized by polarized light. Optical rotation: rotation the rotation of linearly polarized light by sample. the Optical rotary dispersion: dispersion the variation of optical rotation as a function of wavelength. The spectrum of optical rotation. Circular Dichroism: Dichroism the difference in absorption of left and right circularly light. (i. e. , L R) 10/31/2020 Dr. John M Xavier 2

Intro… "Dichroism" is used to denote direction-dependent light absorption. Linear Dichroism refers to the differential absorption of light polarized parallel or perpendicular to the some reference axis. "Birefringence" refers to the direction-dependent index of refraction 10/31/2020 Dr. John M Xavier 3

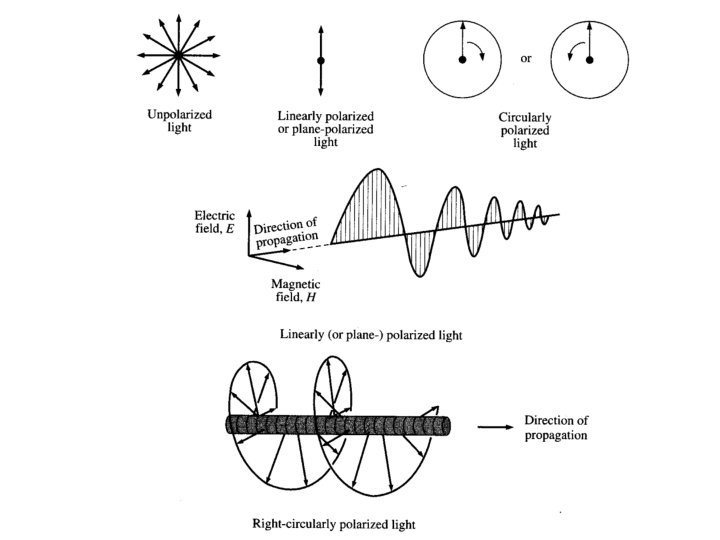
• Plane polarized light consists two circularly polarized components of equal intensity • Two circularly polarized components are like left- and right-handed springs/helices • As observed by looking at the source, right-handed circularly polarized light rotates clockwise • Frequency of rotation is related to the frequency of the light • Can be resolved into its two circularly polarized components 10/31/2020 Dr. John M Xavier 4
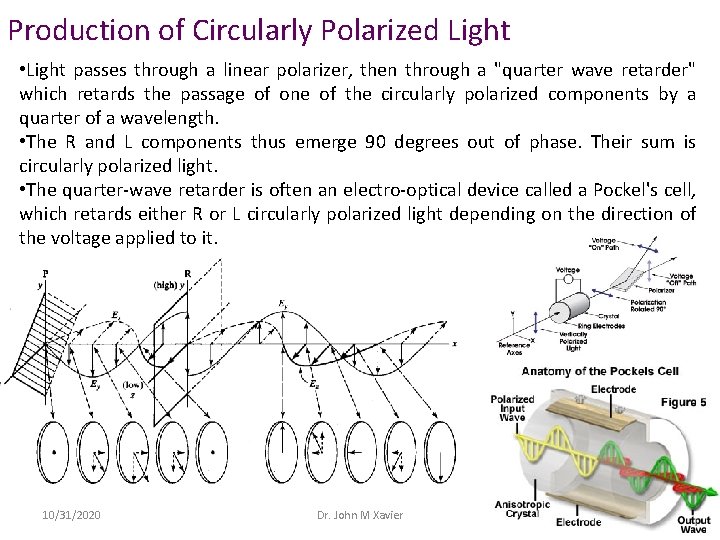
Production of Circularly Polarized Light • Light passes through a linear polarizer, then through a "quarter wave retarder" which retards the passage of one of the circularly polarized components by a quarter of a wavelength. • The R and L components thus emerge 90 degrees out of phase. Their sum is circularly polarized light. • The quarter-wave retarder is often an electro-optical device called a Pockel's cell, which retards either R or L circularly polarized light depending on the direction of the voltage applied to it. 10/31/2020 Dr. John M Xavier 6

The E vector of linearly polarized light (also called plane-polarized light has a constant direction and a modulated amplitude. By contrast, the E vector of circularly polarized light has a constant amplitude but a modulated direction. 10/31/2020 Dr. John M Xavier 7
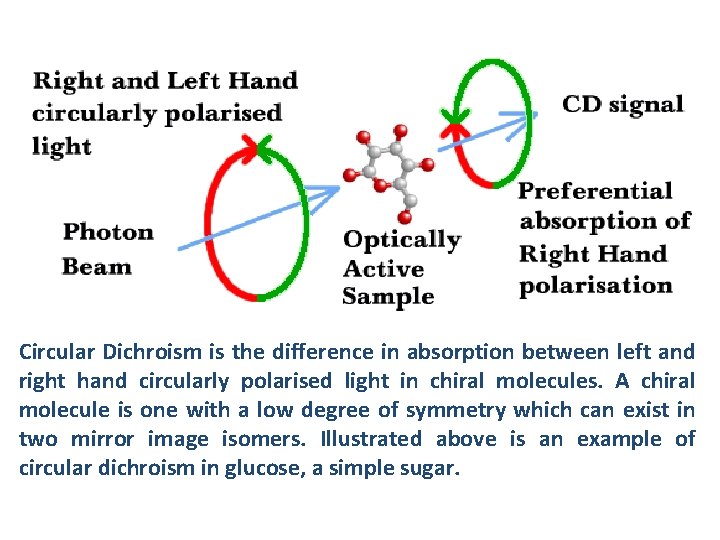
Circular Dichroism is the difference in absorption between left and right hand circularly polarised light in chiral molecules. A chiral molecule is one with a low degree of symmetry which can exist in two mirror image isomers. Illustrated above is an example of circular dichroism in glucose, a simple sugar.
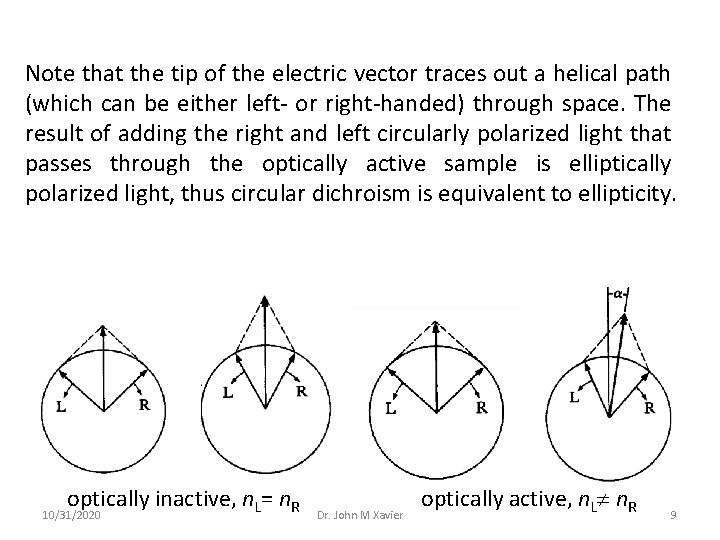
Note that the tip of the electric vector traces out a helical path (which can be either left- or right-handed) through space. The result of adding the right and left circularly polarized light that passes through the optically active sample is elliptically polarized light, thus circular dichroism is equivalent to ellipticity. optically inactive, n. L= n. R 10/31/2020 Dr. John M Xavier optically active, n. L n. R 9

Optical rotary dispersion • If the refractive indices of the sample for the left and right handed polarized light are different, when the components are recombined, the planepolarized radiation will be rotated through an angle • nl, nr are the indices of the refraction for lefthanded and right-handed polarized light • is in radians per unit length (from )

Physics of CD and ORD • Linear polarized light can be viewed as a superposition of opposite circular polarized light of equal amplitude and phase. • A projection of the combined amplitudes perpendicular to the propagation direction thus yields a line. When this light passes through an optically active sample with a different absorbance A for the two components, the amplitude of the stronger absorbed component will be smaller than that of the less absorbed component. • The consequence is that a projection of the resulting amplitude now yields an ellipse instead of the usual line. • The occurrence of ellipticity is called Circular Dichroism. • Rotation of the polarization plane (or the axes of the dichroic ellipse) by a small angle a occurs when the phases for the 2 circular components become different, which requires a difference in the refractive index n. The change of optical rotation with wavelength is called optical rotary dispersion, ORD. 10/31/2020 Dr. John M Xavier 11
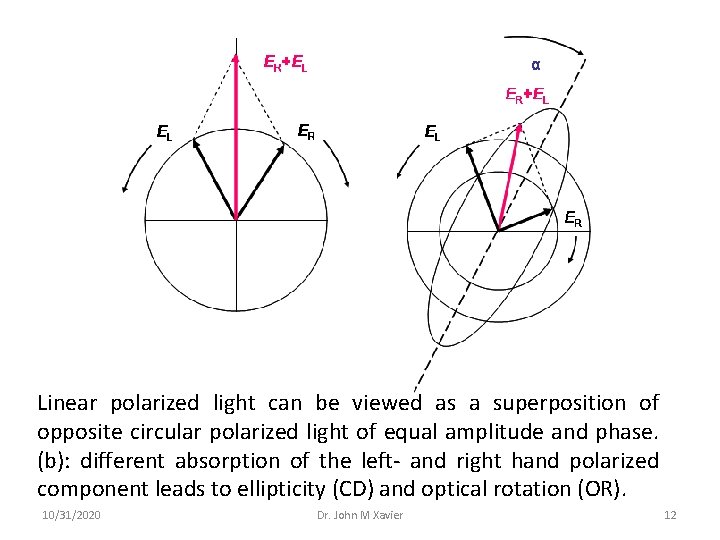
Linear polarized light can be viewed as a superposition of opposite circular polarized light of equal amplitude and phase. (b): different absorption of the left- and right hand polarized component leads to ellipticity (CD) and optical rotation (OR). 10/31/2020 Dr. John M Xavier 12

CD data analysis As mentioned in the introduction, the difference in absorption to be measured is very small. The differential absorption is usually a few 1/100 ths to a few 1/10 th of a percent, but it can be determined quite accurately. The raw data plotted on the chart recorder represent the ellipticity of the sample in radians. To be able to compare these ellipticity values we need to convert into a normalized value. The unit most commonly used (in protein and peptide work) is the mean molar ellipticity per residue. We need to consider path length l, concentration c , molecular weight M and number of residues 10/31/2020 Dr. John M Xavier 13

Circular Dichroism Circular dichroism (CD) spectroscopy measures differences in the absorption of left-handed polarized light versus right-handed polarized light which arise due to structural asymmetry. The absence of regular structure results in zero CD intensity, while an ordered structure results in a spectrum which can contain both positive and negative signals. Jasco J-810 Circular Dichroism System
Polarimetry What happens if light interacts with chiral molecules? Enantiomeric molecules interact differently with circular polarized light. Polarizability a depends on direction of rotation of incoming circular polarized light. Optically active substances exhibit different refractive indices for right n. R and left n. L polarized light n. R ≠ n. L
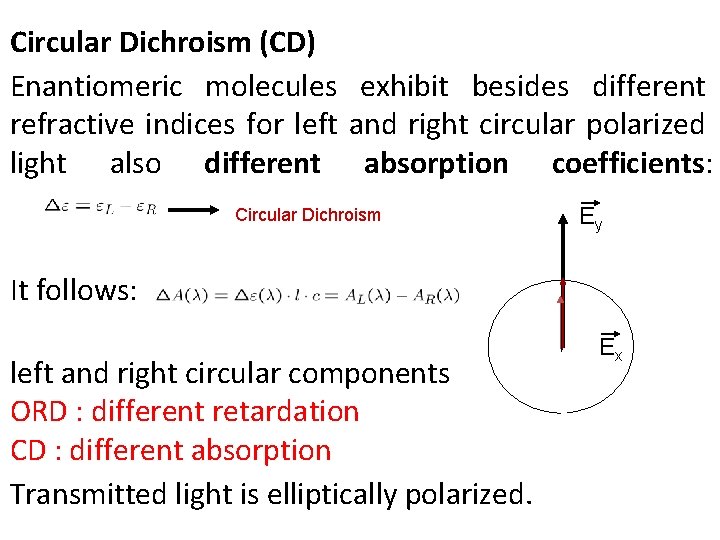
Circular Dichroism (CD) Enantiomeric molecules exhibit besides different refractive indices for left and right circular polarized light also different absorption coefficients: Circular Dichroism Ey It follows: left and right circular components ORD : different retardation CD : different absorption Transmitted light is elliptically polarized. Ex
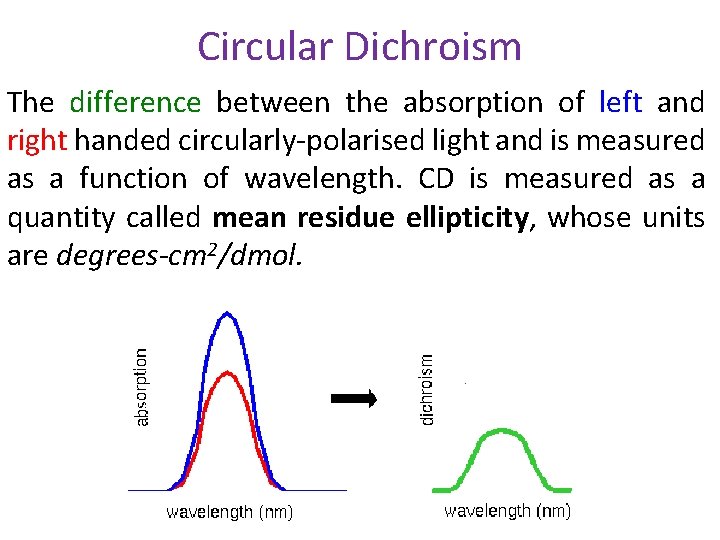
Circular Dichroism The difference between the absorption of left and right handed circularly-polarised light and is measured as a function of wavelength. CD is measured as a quantity called mean residue ellipticity, whose units are degrees-cm 2/dmol.

Circular dichroism • Measurement of how an optically active compound absorbs right- and left-handed circularly polarized light • All optically active compounds exhibit CD in the region of the appropriate absorption band • CD is plotted as l- r vs • For CD, the resulting transmitted radiation is not plane-polarized but elliptically polarized 10/31/2020 Dr. John M Xavier 18

Circular dichroism • is therefore the angle between the initial plane of polarization and the major axis of the ellipse of the resultant transmitted light, α = [180/λ](n. L-n. R) • A quantity is defined such that tan is the ratio of the major and minor axis of the ellipse of the transmitted light • ’ approximates the ellipticity • When expressed in degrees, ’ can be converted to a specific ellipticity [ ] or a molar ellipticity [ ] • CD is usually plotted as [ ] 10/31/2020 Dr. John M Xavier 19

If RCP component travels faster, the medium is dextrorotatory (α is +ve) If LCP component travels faster, the medium is Levorotatory (α is –ve) 10/31/2020 Dr. John M Xavier 20

ORD and CD curves: Cotton effects • CD plots are Gaussian rather than S-shaped • Positive or negative deflections depend on the sign of or [ ] and corresponds to the sign of the Cotton effect • Maximum of the CD occurs at the absorption max • When more than one overlapping Cotton effect, the CD may be easier to interpret than the ORD with overlapping S-shaped bands 10/31/2020 Dr. John M Xavier 21

• One of the important uses of CD and ORD is to determine the absolute configuration of ketones and aldehydes. • This is accomplished by application of a semiempirical generalization known as the octant rule. • The rule predicts the sign of the n → π* Cotton effect of a ketone due to the contributions of substituents in each of eight octant sectors surrounding the carbonyl chromophore. • The rule allows one to determine the absolute configuration and also the likely conformation of a molecule from the sign of the Cotton effect. 10/31/2020 Dr. John M Xavier 22

Cotton Effect • Positive Cotton effect is where the peak is at a higher wavelength than the trough. • Negative Cotton effect is the opposite • Optically pure enantiomers always display opposite Cotton effect ORD curves of identical magnitude • Zero crossover point between the peak and the trough closely corresponds to the normal UV max
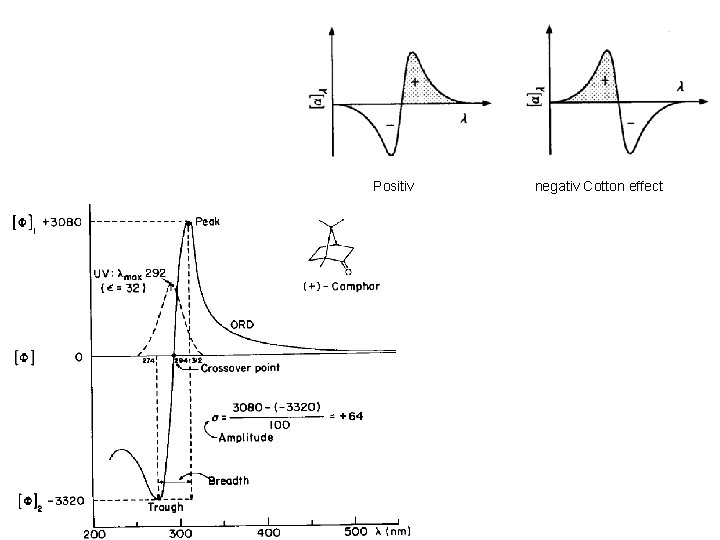
Positiv negativ Cotton effect
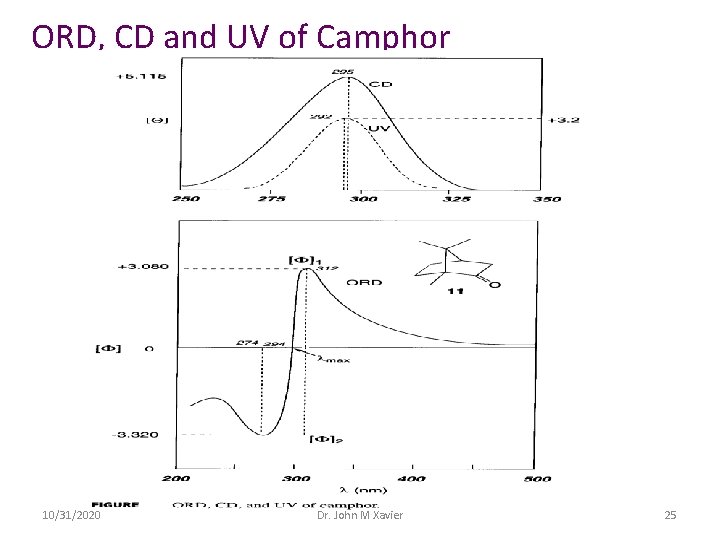
ORD, CD and UV of Camphor 10/31/2020 Dr. John M Xavier 25

The ORD curve or anomalous curve or Cotton curve is said to exhibit a CE which is characterized by a) amplitude and b) width If the peak is at higher wavelength than the trough the CE is positive whereas if the trough is at higher wavelength, CE is negative Plain curve: it is represented mathematically by the following equation in which λ is the wavelength at which the specific rotation is measured, λ° corresponds to the wavelength close to the absorption maximum, k is empirical constant. [α] = k/(λ 2 - λ° 2) The above equation is known as Drude equation 10/31/2020 Dr. John M Xavier 26

Carbonyl compounds • Most important application of ORD-CD data is for organic compounds • C=O has a weak n * transition ~ 280 nm, but it can be easily observed by ORD-CD on dilute samples (10 -2 – 10 -6 M) • Semi-empirical rules have been developed to derive conclusions about C=O location, ring conformational properties, and absolute stereochemistry • The constitution, conformation or configuration can be determined if two of these three things are known 10/31/2020 Dr. John M Xavier 27
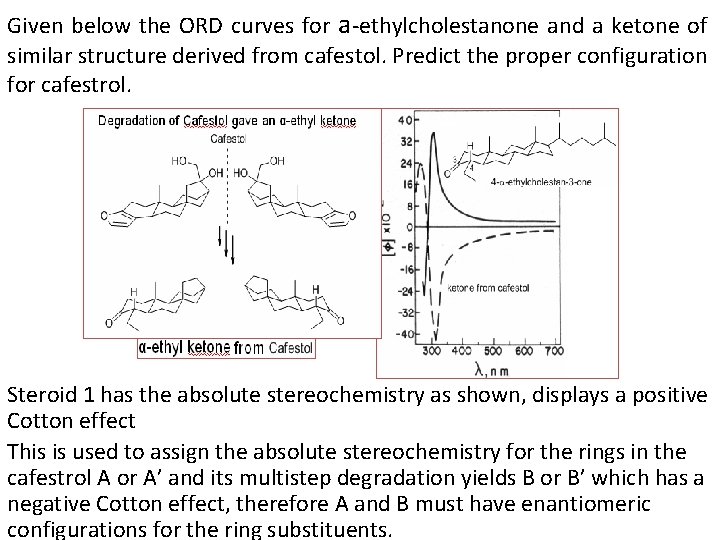
Given below the ORD curves for a-ethylcholestanone and a ketone of similar structure derived from cafestol. Predict the proper configuration for cafestrol. Steroid 1 has the absolute stereochemistry as shown, displays a positive Cotton effect This is used to assign the absolute stereochemistry for the rings in the cafestrol A or A’ and its multistep degradation yields B or B’ which has a negative Cotton effect, therefore A and B must have enantiomeric configurations for the ring substituents.
- Slides: 28