CI 4 6 BornHaber Cycle C JHUDSON 2005
CI 4. 6 – Born-Haber Cycle (C) JHUDSON 2005

What do we mean by lattice enthalpy? For an ionic compound the lattice enthalpy is the enthalpy change when one mole of solid in its standard state is formed from its ions in the gaseous state. - + - + - The lattice enthalpy cannot be measured directly and so we make use of other known enthalpies and link them together with an enthalpy cycle. This enthalpy cycle is the Born-Haber cycle.
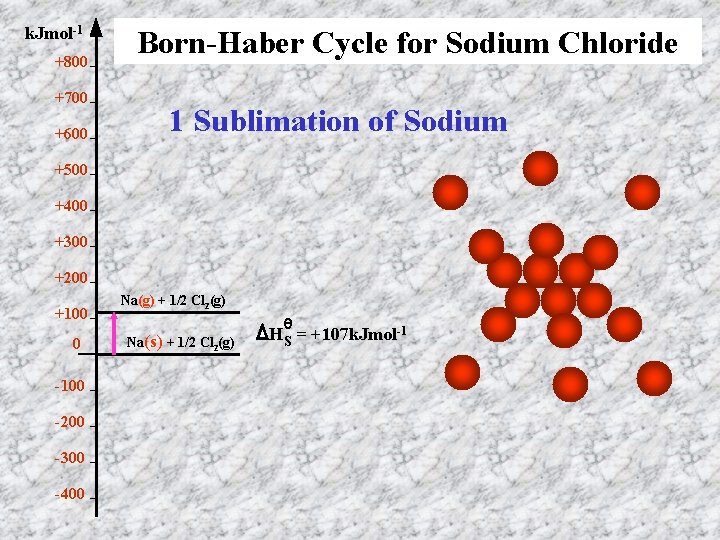
k. Jmol-1 +800 +700 +600 Born-Haber Cycle for Sodium Chloride 1 Sublimation of Sodium +500 +400 +300 +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) θ HS = +107 k. Jmol-1
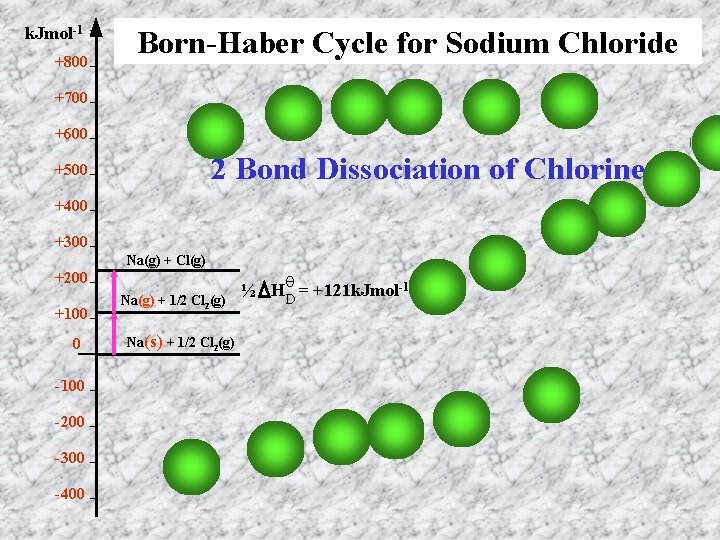
k. Jmol-1 +800 Born-Haber Cycle for Sodium Chloride +700 +600 2 Bond Dissociation of Chlorine +500 +400 +300 Na(g) + Cl(g) +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) ½ HθD = +121 k. Jmol-1
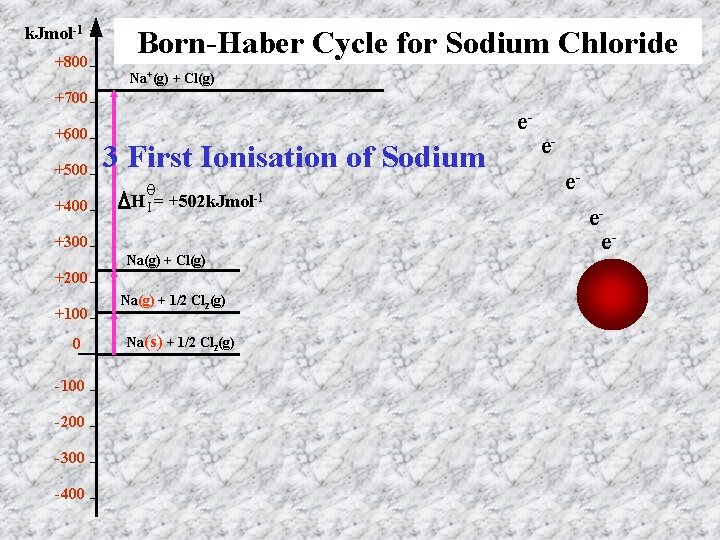
k. Jmol-1 +800 Born-Haber Cycle for Sodium Chloride Na+(g) + Cl(g) +700 +600 +500 +400 e- 3 First Ionisation of Sodium θ HI = +502 k. Jmol-1 +300 Na(g) + Cl(g) +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) eeee+
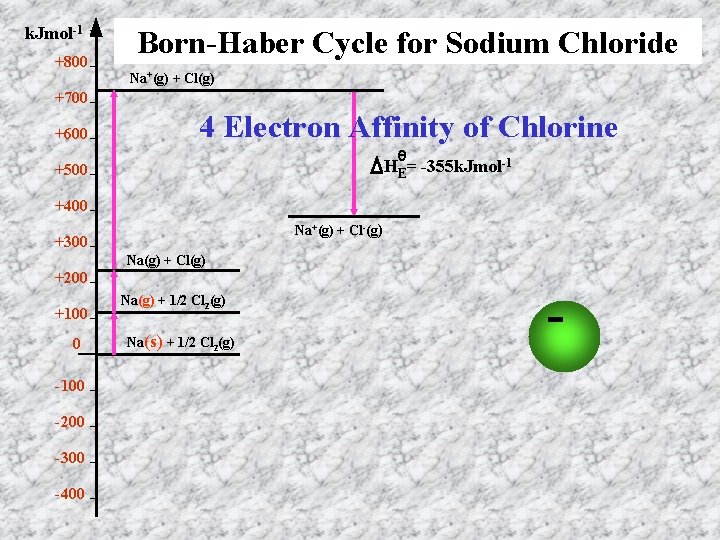
k. Jmol-1 +800 Born-Haber Cycle for Sodium Chloride Na+(g) + Cl(g) +700 +600 4 Electron Affinity of Chlorine θ HE= -355 k. Jmol-1 +500 +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) -e-
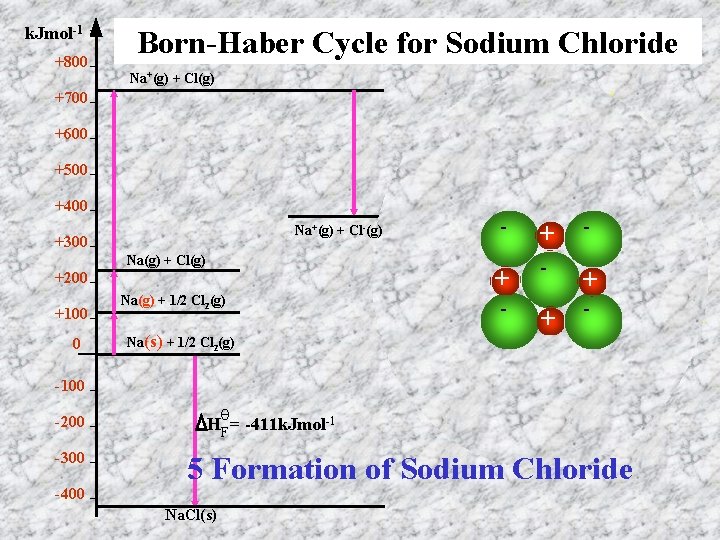
k. Jmol-1 +800 Born-Haber Cycle for Sodium Chloride Na+(g) + Cl(g) +700 +600 +500 +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) - + - + - Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 θ HF = -411 k. Jmol-1 5 Formation of Sodium Chloride Na. Cl(s)
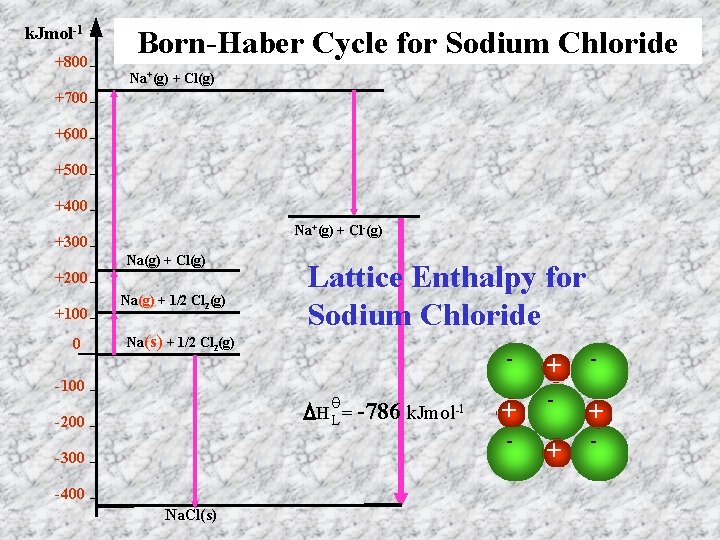
k. Jmol-1 +800 Born-Haber Cycle for Sodium Chloride Na+(g) + Cl(g) +700 +600 +500 +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) -100 Lattice Enthalpy for Sodium Chloride θ H L = -786 k. Jmol-1 -200 -300 -400 Na. Cl(s) - + - + -
- Slides: 8