Chronic Lymphocytic Leukemia Risk factors and complications By

Chronic Lymphocytic Leukemia: Risk factors and complications By Olfat M. Hendy Professor of Clinical Pathology (Hematology & Immunology), NLI- Menoufia University Egypt


Objectives Understand the CLL pathogenesis and causes w Discuss risk factors of CLL and how affecting diagnosis and treament outcome w Discuss the complication of CLL w

Chronic Lymphocytic leukemia The World Health Organization (WHO) includes “lymphocytic leukemias” and “lymphoma” within one classification. w However, Lymphoma begins in a lymph node, or another lymphatic structure in the skin, gastrointestinal tract, or some other site in the body. Lymphocytic leukemia develops in the lymphatic tissue within the bone marrow. w

What is CLL? Chronic lymphocytic leukemia (CLL) results from an acquired (not present at birth) mutation in the DNA of a single marrow cell that develops into a lymphocyte. w In 95 percent of people with CLL, the change occurs in a B lymphocyte. w In the other 5 percent of people with CLL, the cell that transforms from normal to leukemic has the features of a T lymphocyte or a natural killer (NK) cell w
The Course of CLL Once the marrow cell undergoes the leukemic change, it multiplies into many cells. w The result is uncontrolled growth of CLL cells in the marrow, leading to an increase in the number of CLL cells in the peripheral blood. w

The leukemic cells that accumulate in the marrow in people with CLL do not prevent normal blood cell production. w So, this is the reason for the generally less severe early course of CLL. w CLL takes different forms; some people have disease that is slow growing (stable disease) and some have faster growth. w

People with minimal changes in their blood cell counts (a little or no decrease in the number of red blood cells, normal neutrophil and platelet counts) may have stable disease for years. w Other people with CLL have a faster-growing form of the disease due to marked increase of number of lymphocytes leading to the CLL cells accumulate in the bone marrow and blood, and there is a significant decrease in the numbers of red blood cells and platelets. w

People with faster-growing CLL may have; A severe immunoglobulin deficiency, sometimes coupled with a low neutrophil count, which can lead to recurrent infections. w Enlarged lymph nodes: that can lead to compression of neighboring organs (eg; enlarged lymph nodes in the abdomen can interfere with the functions of the GIT and/or the urinary tract). w An enlarged spleen which can press on the stomach causing early fullness and also discomfort in the left upper part of the abdomen. w

Diagnosis and Risk Factors Incidence: CLL is more common in people who are 70 years and older w The incidence of the disease increases from less than one per 100, 000 in individuals aged 40 to 44 years to more than 30 per 100, 000 in individuals aged 80 and older. w Older patients tend to have a worse outcome due to being diagnosed with a more aggressive CLL and the inability to tolerate treatment and symptoms of the disease. w

Common Tests Used to Evaluate CLL: Laboratory tests: w Complete blood count and blood chemistry studies. w Immunophenotyping by Flow cytometry. w Immunohistochemistry. w Fluorescence in-situ hybridization (FISH) and conventional chromosomal analysis. w Polymerase chain reaction (PCR) for mutational status Other tests: w Biopsies: Lymph node biopsy ( when clinically indicated), BM examination and aspiration (not necessarily performed to diagnose CLL). w Imaging tests: X-ray, CT and PET (positron emission tomography) scan (not routinely performed)

Diagnosis: Briefly; w 1 - Blood Cell Count and Examination. w A patient with CLL will have increased numbers of lymphocyte, low platelet counts and low RBCs counts may also be present.
2 - Bone Marrow Examination: a BM aspiration and biopsy generally are not needed to make a diagnosis of CLL provided RBCs and platelets are normal. w However, these tests are recommended before treatment as a baseline data that can rule out other diseases. w They can also be used later on to evaluate the effects of therapy. w The bone marrow biopsy results will also show one of four patterns characteristic of CLL: nodular, interstitial, mixed or diffuse. w
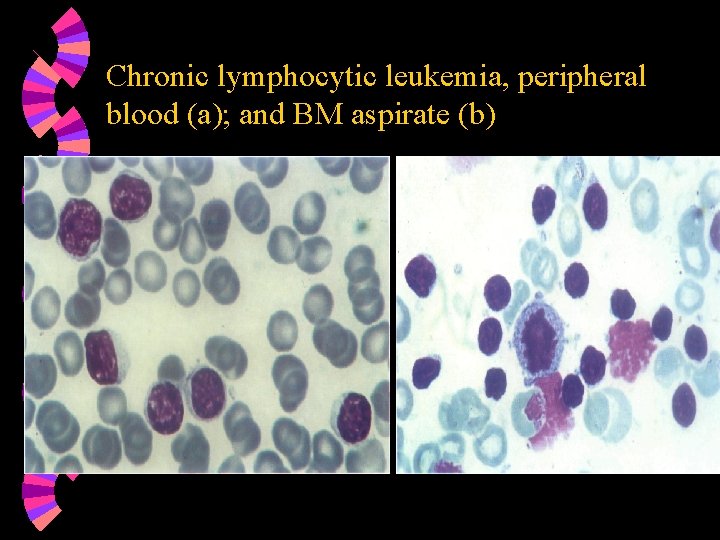
Chronic lymphocytic leukemia, peripheral blood (a); and BM aspirate (b)
3 - Immunophenotyping. (or flow cytometry) of lymphocytes is an important process used to diagnose CLL, and other types of leukemia and lymphoma. w Immunophenotyping also determines whether the CLL cells are from a change in either B-cell or T-cell development. CLL: making the diagnosis if; w Persistent lymphocytosis > 5× 109/L with chracteristic morphology and monoclonal phenotype: CD 5+, 19+, 22+, 20+, 23+, Sm. Ig (dim), CD 79 b low/-, CD 10 w
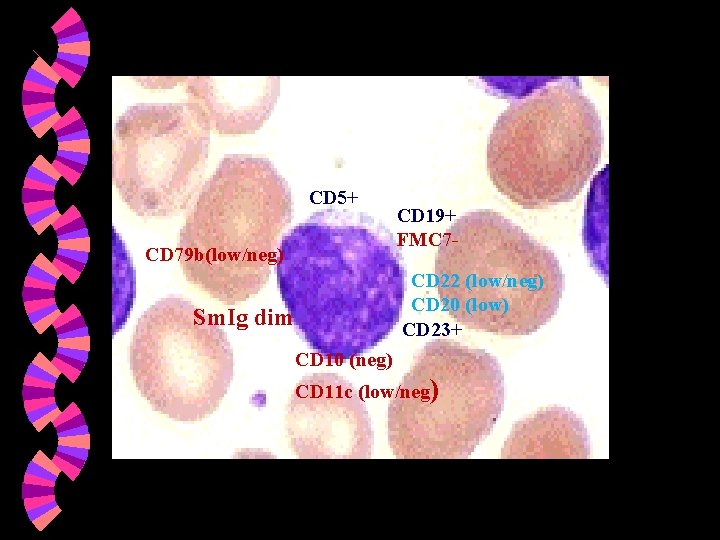
CD 5+ CD 79 b(low/neg) CD 19+ FMC 7 CD 22 (low/neg) CD 20 (low) CD 23+ Sm. Ig dim CD 10 (neg) CD 11 c (low/neg)

4 - Immunoglobulin Levels. - The measurement of the concentration of immunoglobulins (gamma globulins) in the blood is another important test. - People with CLL often have low levels of immunoglobulins because CLL cells do not make effective antibodies. w 5 - B 2 microglobulin level. 6 - LDH serum level.

Risk Factor and outcome of patients with CLL depends on: 1 - CLL Staging. w 2 - Chromosomal Changes and Mutational Status. w

1 - Staging: Staging for CLL helps doctors to both assess how the disease is expected to progress over time and also to develop a treatment plan Staging systems for CLL take into account: w The elevation of blood and marrow leukemic lymphocyte counts. w The size and distribution of lymph nodes w The spleen size. w The degree of anemia and the extent of decreased blood platelet counts. w

Table 1. Commonly Used CLL Staging Systems : Rai Staging System (at Diagnosis) Binet Staging System (at Diagnosis) Low Risk— 0: Abnormal increase in the number of circulating lymphocytes in blood and BM A: Abnormal increase in the number of circulating lymphocytes in the blood and less than 3 areas of palpable enlarged lymphoid tissue Intermediate Risk—I & II: Abnormal increase in the number of lymphocytes in the blood and BM with enlarged lymph nodes, Or Abnormal increase in the number of Lymphocytes in the blood and marrow and enlarged spleen and/or liver B: Abnormal increase in the number of the circulating lymphocytes in blood and greater than 3 areas of palpable enlarged lymphoid tissue High Risk—III & IV: - Abnormal increase in the number of lymphocytes in the blood and BM and anemia (hemoglobin <11 g/d. L), Or - Abnormal increase in the number of lymphocytes in blood and BM and low platelet count (platelet <100, 000/μL) C: Same as B with anemia (hemoglobin <11 g/d. L in men or hemoglobin <10 g/d. L in women) or low platelet count (platelets <100, 000/μL)

2 - Chromosomal Changes: CLL patients who are tested with “fluorescence in situ hybridization (FISH)” are found to have chromosomal abnormalities. Del(13 q) - Deletions on the long arm of chromosome 13, del(13 q), are the most common. - Del(13 q) with no other chromosomal abnormalities is associated with a relatively more favorable outcome. Trisomy 12 - About 10 to 20 percent of patients have CLL cells with three copies of chromosome 12 (trisomy 12) instead of the expected two chromosomes. - Trisomy 12 is associated with intermediate-risk CLL. - Trisomy 12 with other chromosomal abnormalities is associated with a higher risk than trisomy 12 alone.

Del(11 q) - Up to 20% of people with CLL have deletions in CLL cells in the long arm of chromosome 11, del(11 q). - The proportion of CLL patients with del 11 q tend to be younger with large lymph nodes and have high-risk disease. Chromosome 14 Structural abnormalities of chromosome 14 or or Chromosome 6 in CLL cells indicate higher-risk 6 disease Del(17 p) - About 5% of people with CLL at diagnosis have deletions in the short arm of chromosome 17, del(17 p). The critical TP 53 gene in this region is typically deleted. - People who have CLL with del(17 p) tend to have higher-risk disease and usually do not respond as well to standard initial therapy. - Their CLL treatment needs to be approached in a different manner.

CD 38 The expression of CD 38 on CLL cells may be an indicator of higher-risk CLL. ZAP-70 - ZAP-70 (zeta-associated protein 70), when increased, may be associated with higher-risk disease. - The National Comprehensive Cancer Network (NCCN) guidelines state that the evaluation of ZAP 70 expression by flow cytometry can be challenging and is not recommended outside of a clinical trial.

Blood lymphocyte doubling People with CLL whose lymphocyte number doubles in one year have higher-risk CLL and need closer follow up; a lymphocyte number that remains stable indicates a relatively lower risk. beta 2 - A higher level of serum B 2 M, a protein that is microglobulin shed from CLL cells, is associated with a greater (B 2 M) extent of disease. - Several studies have found that B 2 M and other serum markers, such as CD 23, may help predict survival or progression-free survival. Unmutated Ig. Hv - The unmutated immunoglobulin heavy chain variable region gene (Ig. Hv) suggests the likelihood of higher-risk disease. - Forty percent of CLL patients at diagnosis will have this, whereas 60 % will have the more favorable Ig. Hv-mutated disease.

NOTCH 1 gene mutations SF 3 B 1 gene mutations - Notch 1 is a gene involved in the development of different type of blood cells. In CLL, about 10 -15% of patients have mutations of this gene causing it to be more active than it should be. -The CLL patients who have NOTCH 1 gene mutations may progress more quickly, requiring therapy and have a shorter overall survival. - Some commercial laboratories do this test, but it is not recommended by the NCCN guidelines. - The SF 3 B 1 gene is involved in the forming of select proteins in CLL and other blood cancers. - In CLL, approximately 10 to 15% of patients have mutations of this gene, resulting in dysfunctional protein processing. -The CLL patients who have SF 3 B 1 gene mutations may progress more quickly, requiring therapy and have a shorter remission and overall survival. -It is not included in the NCCN guidelines

TP 53 gene - The TP 53 gene is viewed as the gatekeeper to mutations protecting the DNA of cells from damage. Mutated DNA of cancer cells lead to increased cancer growth and resistance to chemotherapy. Mutation of the TP 53 gene is commonly seen in patients who also have del(17 p). - Some patients just have mutation of the TP 53 gene and, these patients have a higher likelihood of progressing more quickly, requiring therapy, not responding well to traditional therapies and having an overall shorter survival. CD 49 d - CD 49 d plays a critical role in leucocyte trafficking, expression activation, and survival, and also facilitates interactions between leucocytes and stromal cells in the BM or LN, and can serve as a signaling receptor that influences Bcell survival via upregulation of Bcl-2 family member. -CLL patients with high expression of CD 49 d have a robust adverse prognostic marker, short overall survival.

Some Factors That Influence the Decision to Treat Patients Who Have CLL: w w w w Enlarging lymph nodes Enlarging spleen Worsening anemia Absolute lymphocyte count (>300, 000/ul) Falling platelet count CLL symptoms (such as fatigue, night sweats, weight loss, fever, etc. ) The presence of risk factors.
Complications: CLL or CLL Treatment 1 - Infections are a common complication for people with CLL. Because of this high risk of infections, immediate vaccination for pneumococcal pneumonia (repeated every 5 years) and a yearly flu vaccine is recommended. A higher risk of infection is caused by: w - The inability of the person’s CLL cells to make antibodies needed to fight infections. w - The effect of chemotherapy, which causes reduced cell counts for certain infection-fighting white blood cells in the blood, specifically neutrophils and monocytes. w

Management of infection; Antibiotic and antifungal therapy are usually required to treat bacterial or fungal infections during the course of the disease. w People who get recurrent infections may also receive gamma globulin injections to correct the immune deficiency. w CLL-related low blood counts are often therapy related are corrected by blood component therapy. w White blood cell growth factors (G-CSF, GMCSF) may used in some situations. w

2 - Anemia is a common side effect of chemotherapy and corrected by blood transfusions. 3 - Richter Transformation. - In about 3 to 5% of people with CLL, the disease transforms into an aggressive lymphoma because of a change in the characteristics of the CLL cells. - This is much more common in Ig. Hv-unmutated CLL. - People with this type of CLL may have significantly enlarged lymph nodes, and may have fevers and weight loss. - Richter transformation is treated with aggressive chemotherapy and reduced-intensity allogeneic transplantation, if feasible. - Outcome for patients with Richter transformation is generally poor unless it is diagnosed prior to receiving CLL therapy.
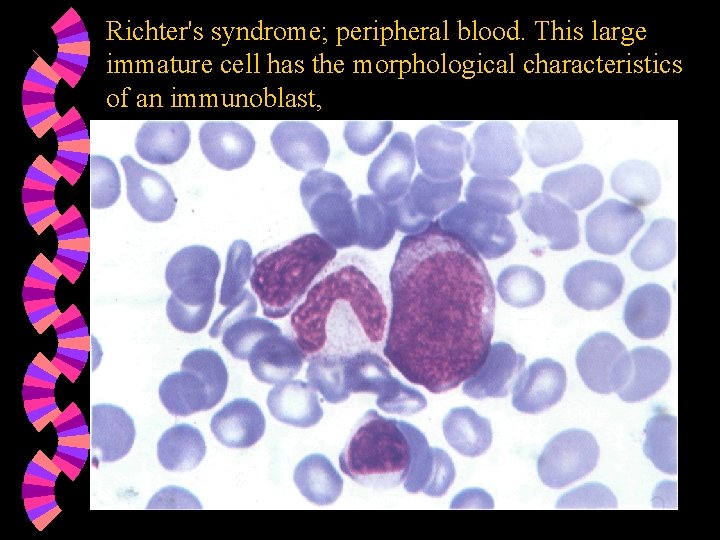
Richter's syndrome; peripheral blood. This large immature cell has the morphological characteristics of an immunoblast,

4 - Prolymphocytic leukemia (PLL): About 15% of people with CLL have leukemia cells that are a mix of lymphocytes and another type of white blood cell, called a prolymphocyte”. w Most people with this type of CLL follow a similar course to that of other people with CLL. w The spleen may enlarge further, and the disease may become less responsive to treatment. w
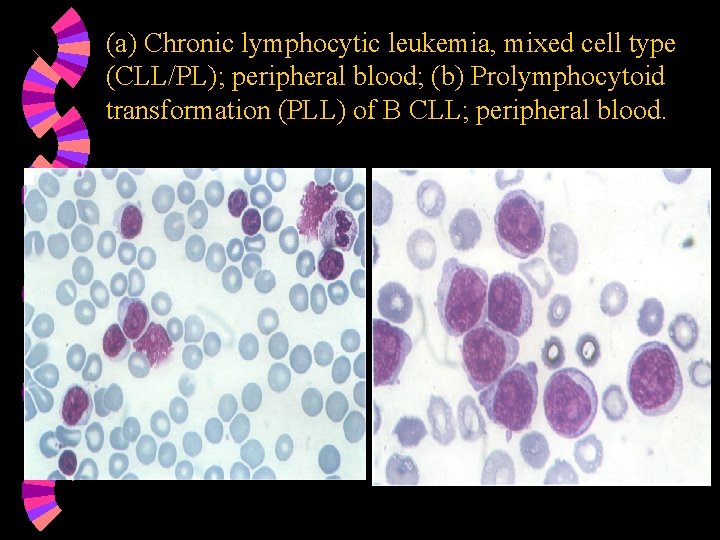
(a) Chronic lymphocytic leukemia, mixed cell type (CLL/PL); peripheral blood; (b) Prolymphocytoid transformation (PLL) of B CLL; peripheral blood.

5 - Autoimmune Hemolytic Anemia. Some people with CLL produce “autoantibodies” thet are usually directed against the patient’s red blood cells and causes them to be removed rapidly from the blood. w This condition, called “autoimmune hemolytic anemia, ” can worsen the effects of already low red blood cell counts. w The “antiglobulin test” or “Coombs test” is used to identify the autoantibodies. w Less often, the antibody works against the platelets can cause an “immune thrombocytopenia, ” leading to a significant decreased platelet counts. w

6 - Second Cancers. w w w People with CLL have a higher risk of developing a second cancer. The second cancers that frequently developed are melanoma, soft tissue sarcoma, colorectal cancer, lung cancer, squamous cell skin cancer and basal cell carcinoma. Both treated and untreated people with CLL can develop AML or myelodysplastic syndromes. This complication is more common after treatment with fludarabine and cyclophosphamide or fludarabine/ cyclopho-sphamide/ rituximab. It is important to follow up with patients on a regular basis during and after therapy.

Take Home Message 1 -For diagnosing B-CLL: identify >5. 000 monoclonal B-cells in peripheral blood with a compatible immunophenotype. w 2 - The characteristic immunophenotype: CD 19+, CD 5+, CD 23+, CD 20/low, CD 22 low/neg, CD 79 b low/neg, s. Ig/low, FMC 7 -, CD 10 -, CD 11 c+low/neg, CD 103 -. w 3 - Including markers related to prognosis: CD 38, CD 49 d, Zap 70, detection of chromosomal changes at chromosome most commonly 11, 12, 13, 17 and less commonly 14, 6. w 5 - The risk factors in CLL depend upon the chromosomal changes at chromosomes above, unmutated Ig. Hv, and P 53 gene mutations. w 6 - The most common complications of CLL are: infections, anemia, Richter transformation, autoimmune HA, PLL and development of second cancers. w


Thank You
- Slides: 38