Chromatography Separates components in mixture Based on polarity
Chromatography Separates components in mixture: Based on - polarity - boiling point - ionic strength - size

Chromatography • Mobile phase: phase which sample is dissolved in may be gas, liquid, or supercritical fluid • Stationary phase: phase which mobile phase is forced through • Mobile and stationary phases are chosen so the analyte will distribute itself between the two phases
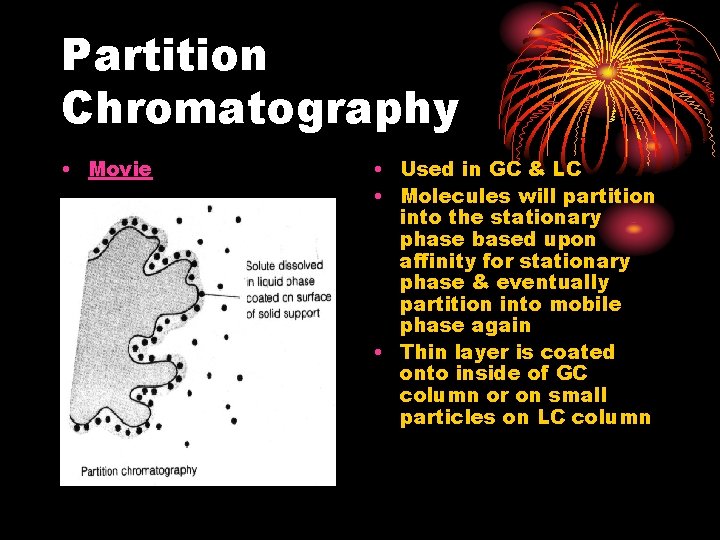
Partition Chromatography • Movie • Used in GC & LC • Molecules will partition into the stationary phase based upon affinity for stationary phase & eventually partition into mobile phase again • Thin layer is coated onto inside of GC column or on small particles on LC column

Adsorption Chromatography • Very similar to partition chromatography • Adsorption just on surface, partition into thin layer • Not used as widely as partition used mainly in TLC & very small particles in LC • Movie
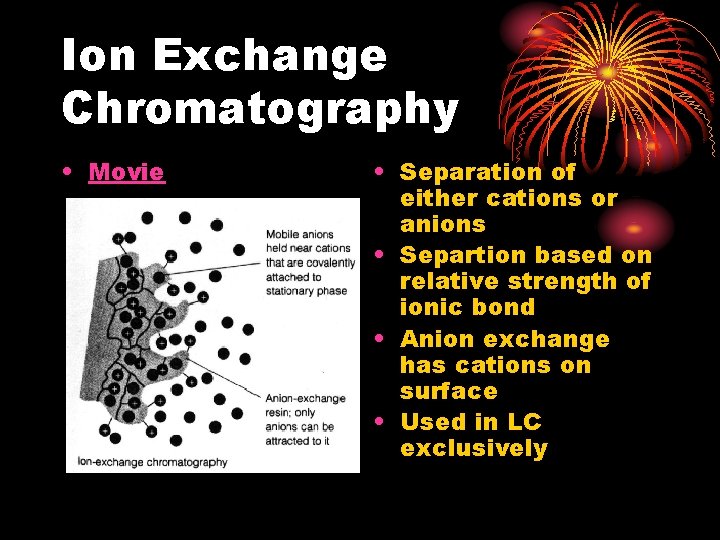
Ion Exchange Chromatography • Movie • Separation of either cations or anions • Separtion based on relative strength of ionic bond • Anion exchange has cations on surface • Used in LC exclusively
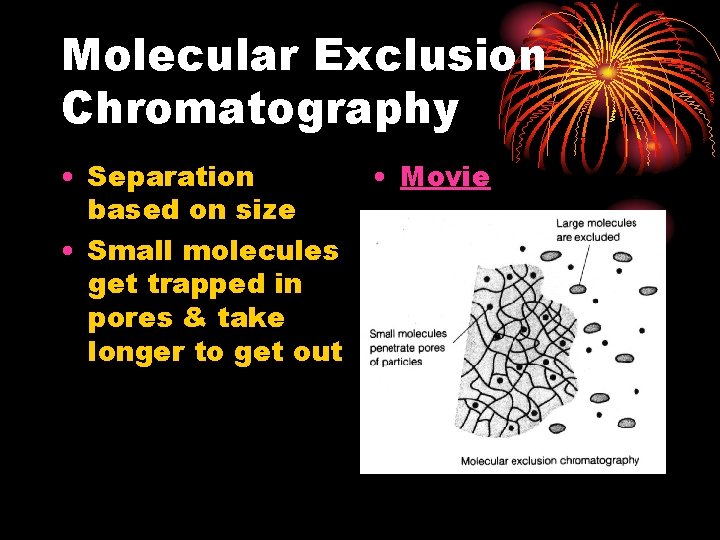
Molecular Exclusion Chromatography • Separation based on size • Small molecules get trapped in pores & take longer to get out • Movie
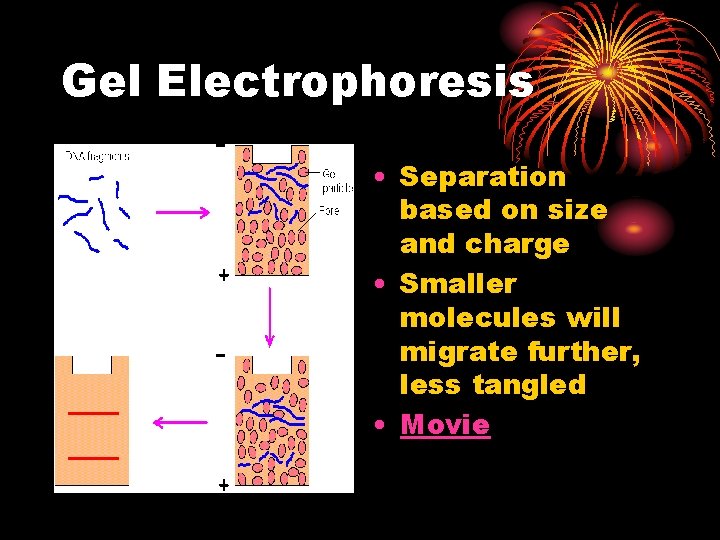
Gel Electrophoresis • Separation based on size and charge • Smaller molecules will migrate further, less tangled • Movie

Affinity Chromatography • Very selective • Specific binding site is used to concentrate analyte on column • Used a lot in biological applications • Movie
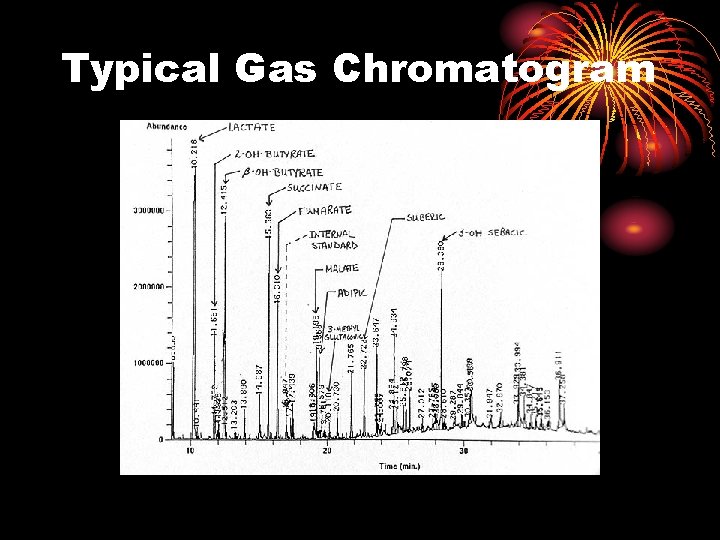
Typical Gas Chromatogram
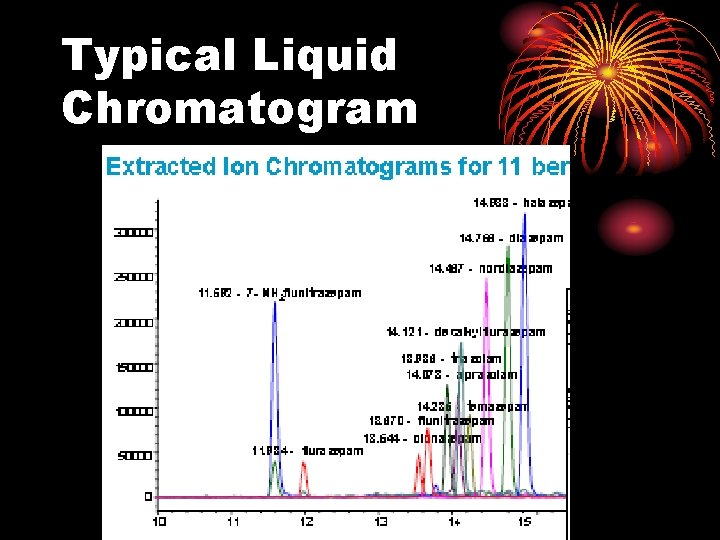
Typical Liquid Chromatogram

Introduction to Chromatography - Theory General Relationships 1. Distribution constant a. Craig counter current experiment 2. Retention time 3. Relationship between distribution constant and retention time 4. Capacity factor k’ 5. Selectivity factor a

Introduction to Chromatography - Theory Peak Broadening 1. Shapes 2. Column efficiency a. plate height b. number of plates 3. Kinetic factors – Van Deemter equation

Craig counter current • movie
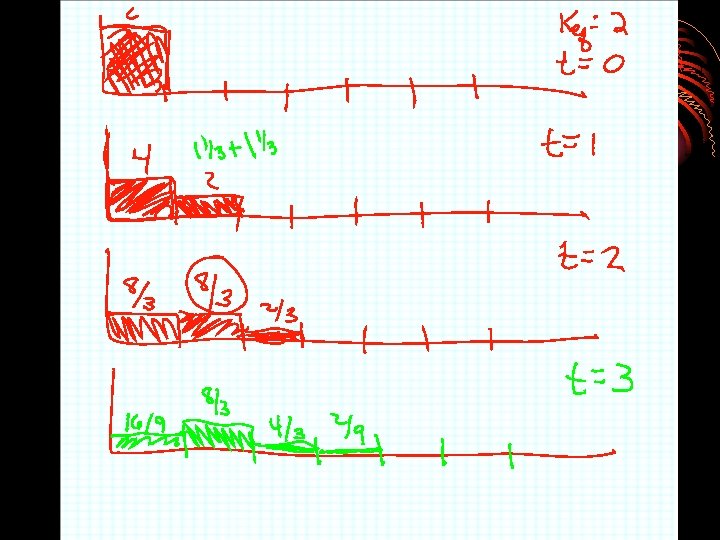
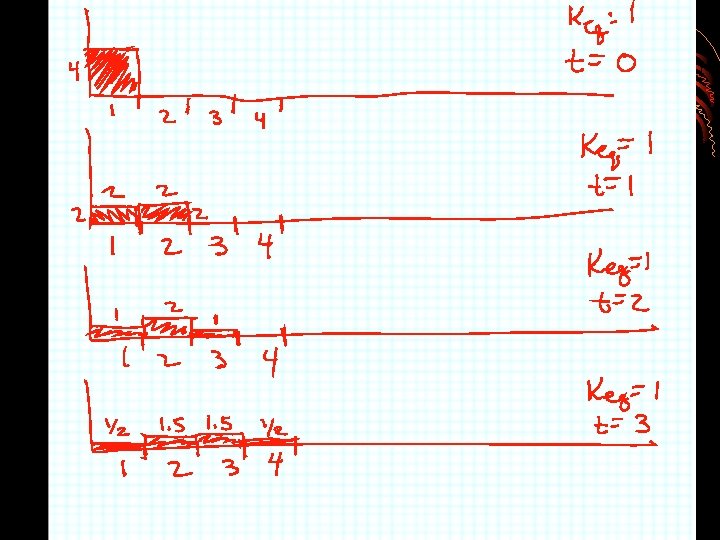
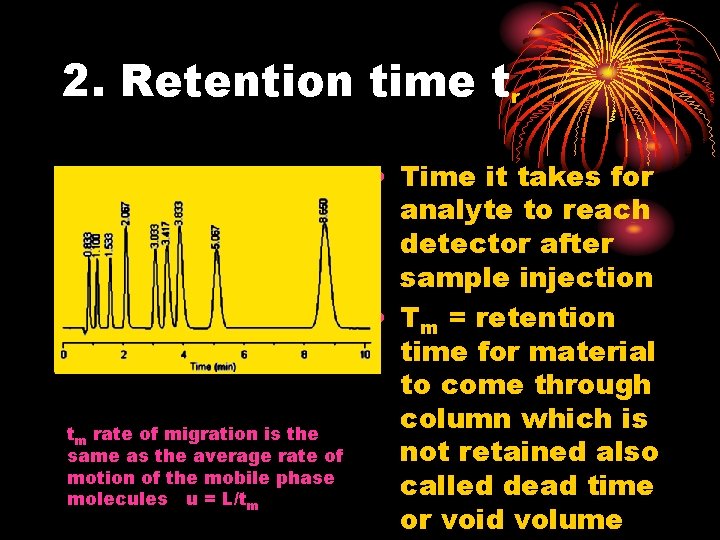
2. Retention time tr tm rate of migration is the same as the average rate of motion of the mobile phase molecules u = L/tm • Time it takes for analyte to reach detector after sample injection • Tm = retention time for material to come through column which is not retained also called dead time or void volume

3. Distribution constant & retention time v = u x moles of analyte in mobile phase total moles of analyte v=ux Cm. Vm + Cs. Vs v=ux 1 1 + KVs/Vm = 1 1 + Cs. Vs/Cm. Vm
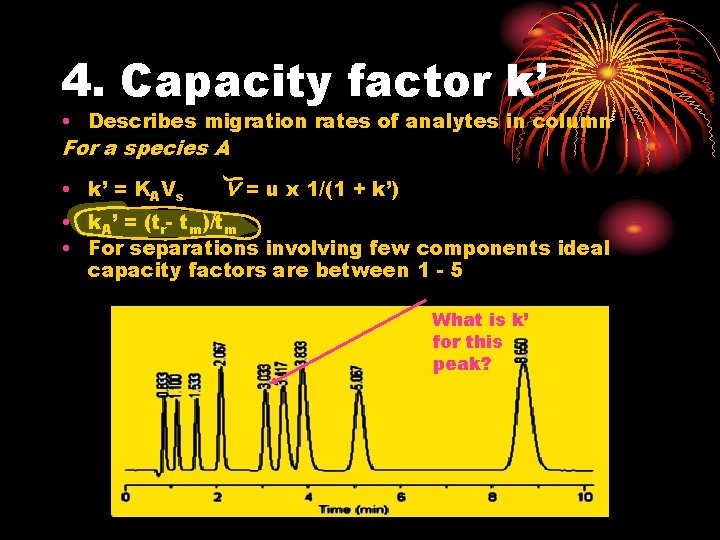
4. Capacity factor k’ • Describes migration rates of analytes in column For a species A • k’ = KAVs v = u x 1/(1 + k’) • k. A’ = (tr- tm)/tm • For separations involving few components ideal capacity factors are between 1 - 5 What is k’ for this peak?
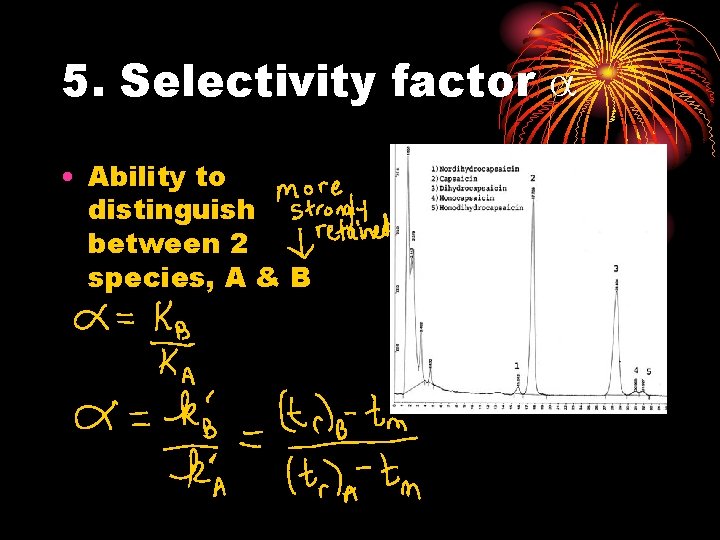
5. Selectivity factor a • Ability to distinguish between 2 species, A & B
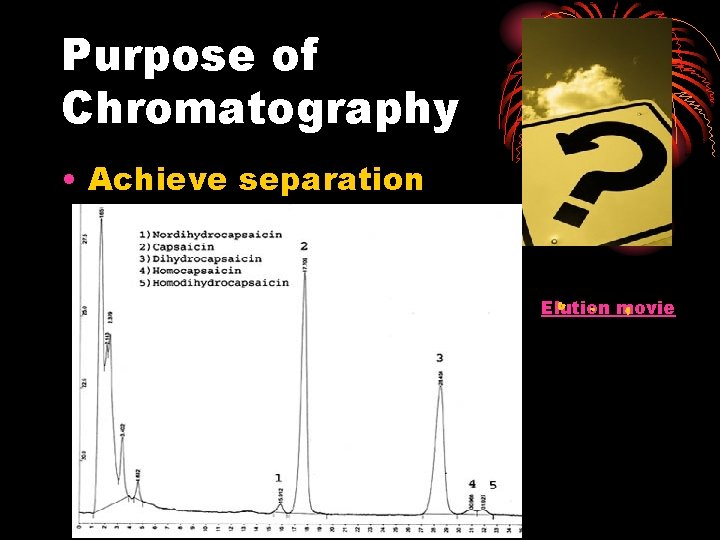
Purpose of Chromatography • Achieve separation Elution movie

Peak Broadening
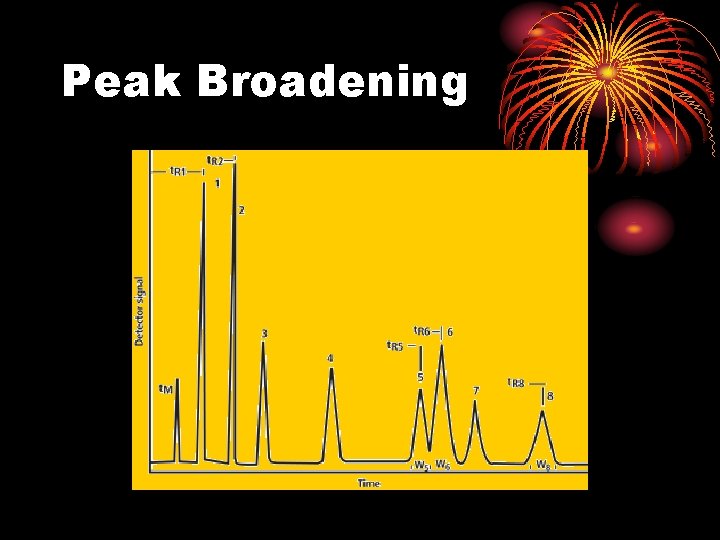
Peak Broadening

• Is peak broadening a good or bad thing? BAD Why?
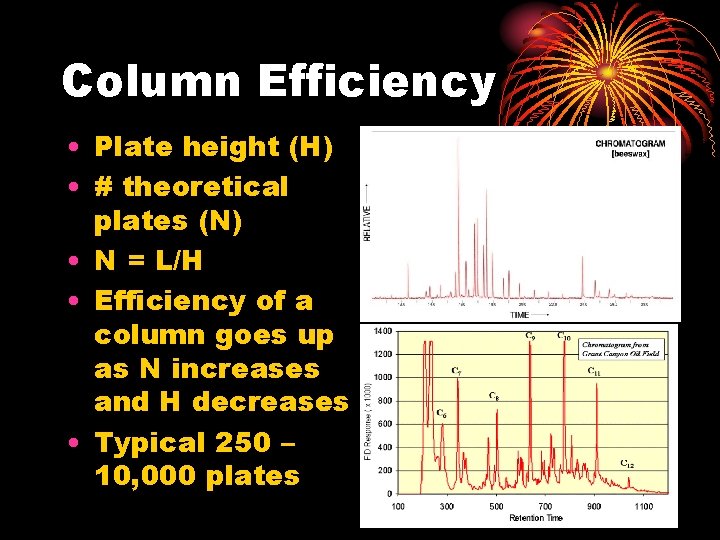
Column Efficiency • Plate height (H) • # theoretical plates (N) • N = L/H • Efficiency of a column goes up as N increases and H decreases • Typical 250 – 10, 000 plates

Plate Height

3. Kinetic Factors: The Van Deemter Equation Reality: column efficiency is affected by kinetic factors

What variable do you think are important in determining the efficiency of a separation?
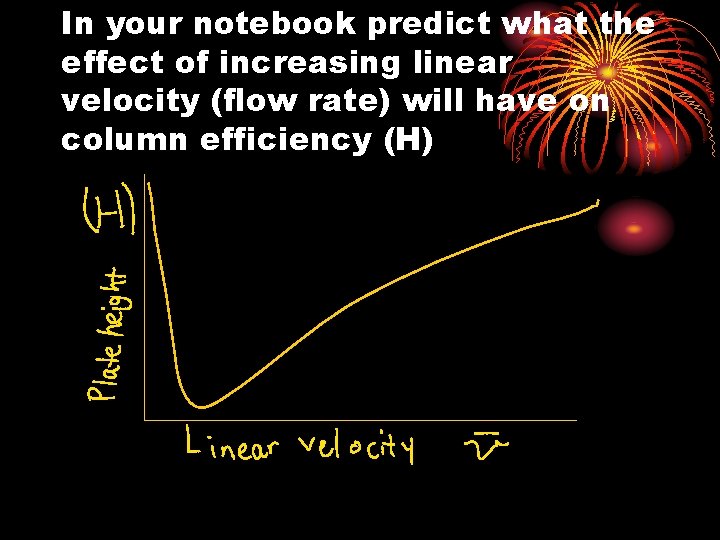
In your notebook predict what the effect of increasing linear velocity (flow rate) will have on column efficiency (H)
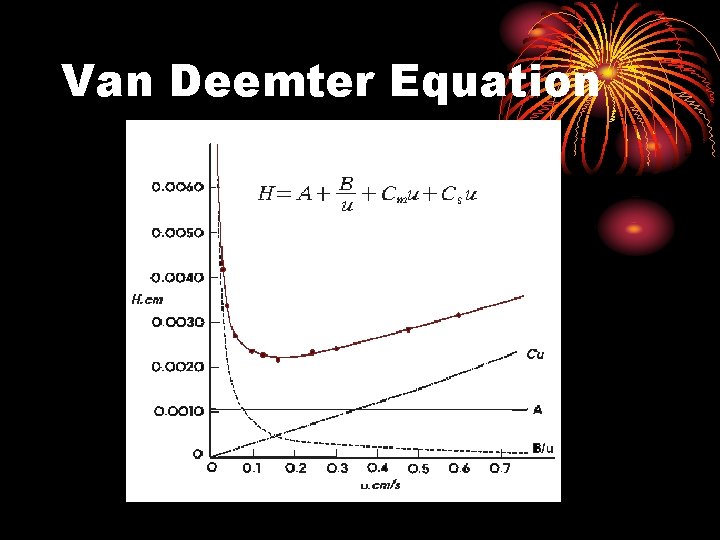
Van Deemter Equation

Van Deemter Equation H = A + B/u + Cu A = Eddy diffusion: Due to different paths molecules can take as they go through particles B/u = longitudnal diffusion Band diffuses in and against direction of mobile phase movement Cu often broken into 2 terms Csu + Cmu Mass transfer coefficient: Time it takes for analyte to diffuse into stationary phase

How can band broadening be reduced? (and thus column efficiency be enhanced) 1. Decrease particle diameter 2. Decrease column width 3. Lowering temperature in GC (reduces diffusion coefficient) 4. Minimize thickness of liquid stationary phase

Resolution This is called General Elution Problem Rs = 2((tr)B – (tr)A) w. A + w. B
- Slides: 33