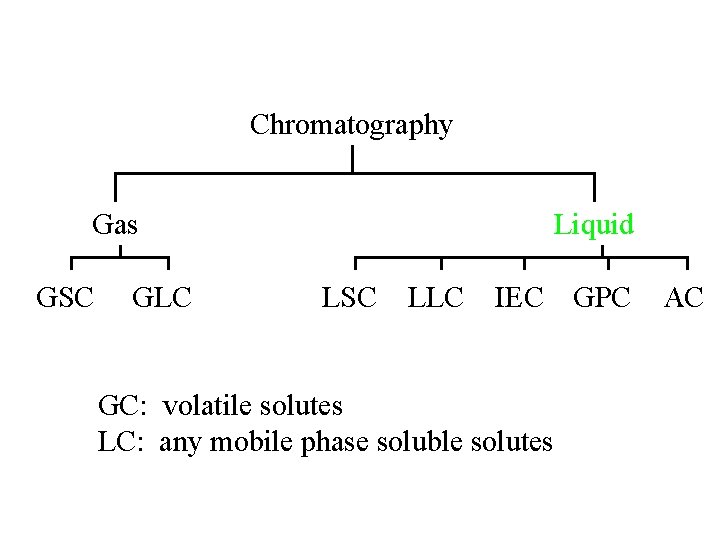
Chromatography Gas GSC GLC Liquid LSC LLC IEC



















- Slides: 61

Chromatography Gas GSC GLC Liquid LSC LLC IEC GPC GC: volatile solutes LC: any mobile phase soluble solutes AC

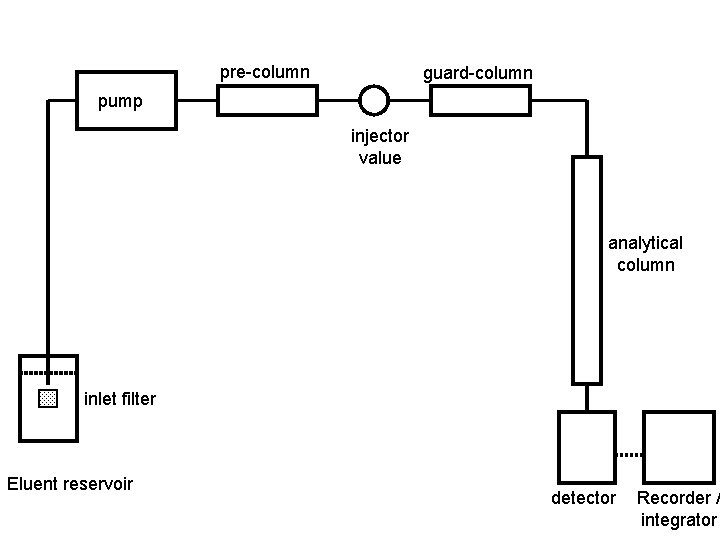
pre-column guard-column pump injector value analytical column inlet filter Eluent reservoir detector Recorder / integrator

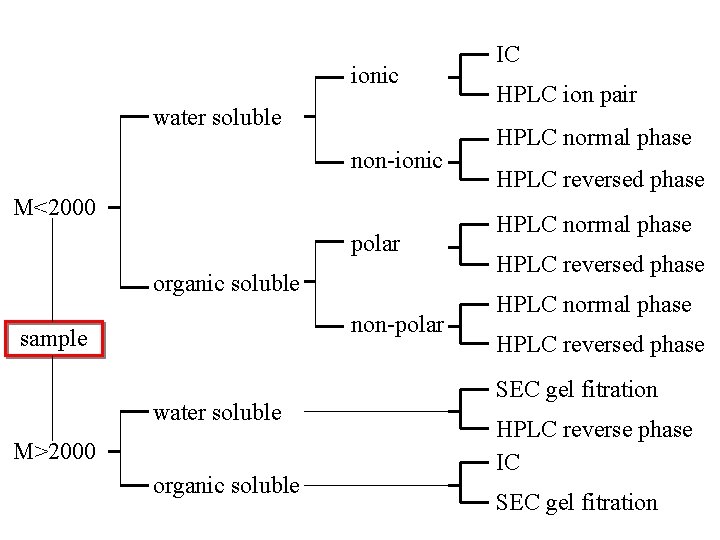
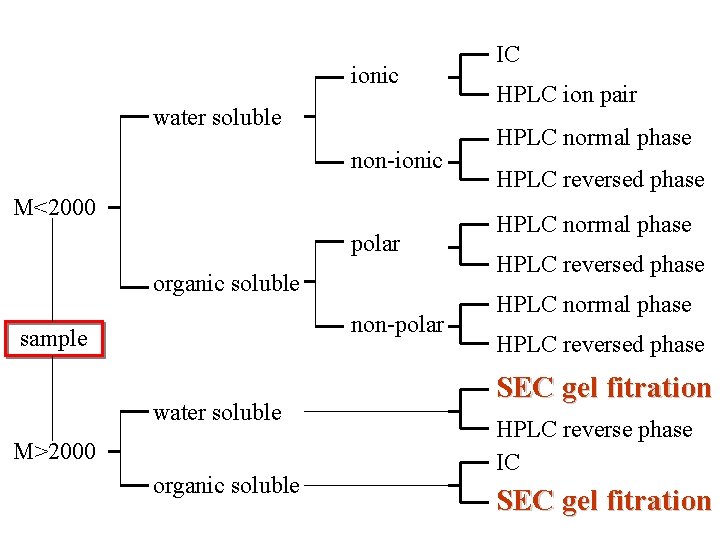
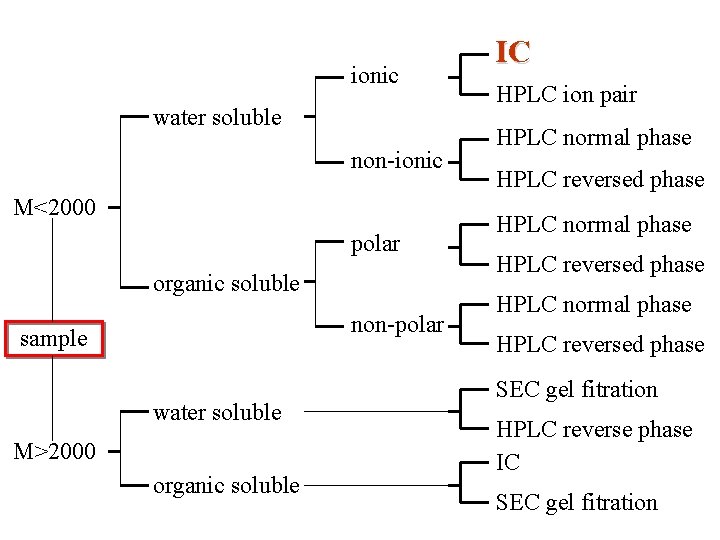
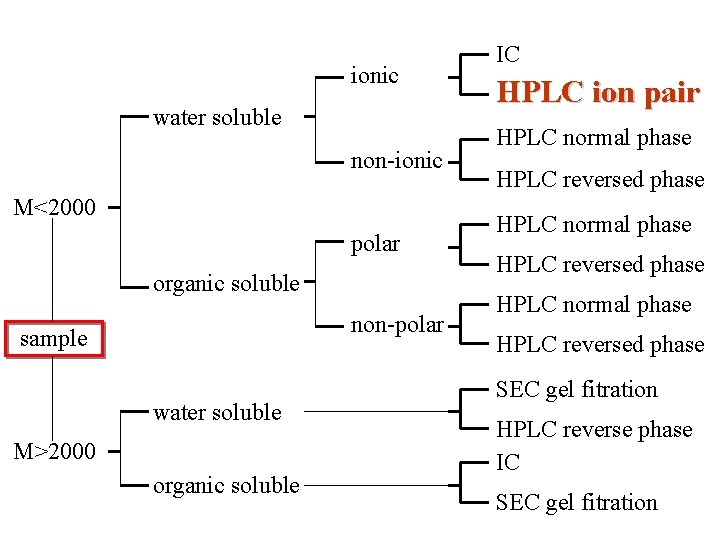
ionic water soluble non-ionic M<2000 polar organic soluble non-polar sample water soluble M>2000 organic soluble IC HPLC ion pair HPLC normal phase HPLC reversed phase SEC gel fitration HPLC reverse phase IC SEC gel fitration

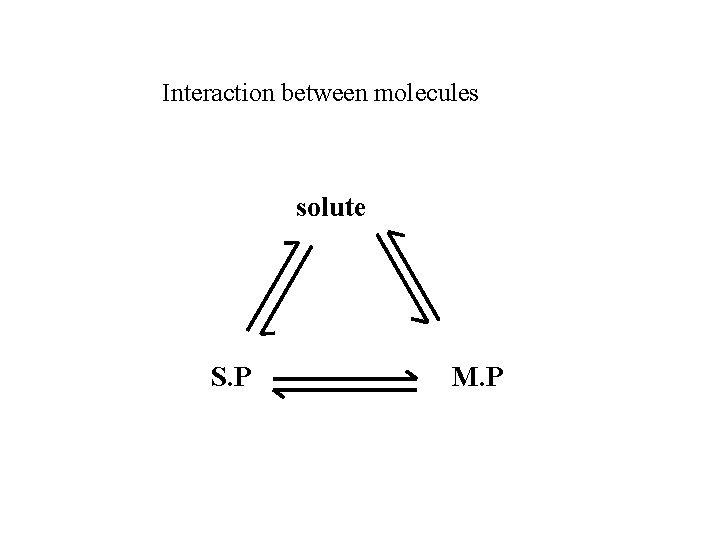
Interaction between molecules solute S. P M. P

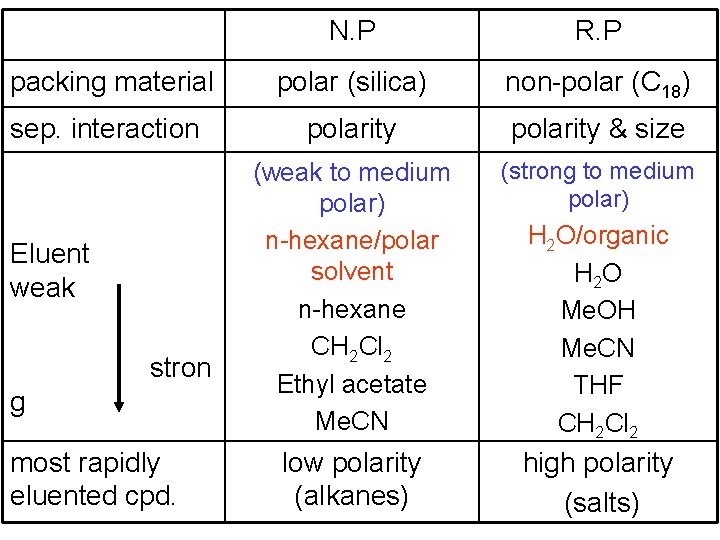
N. P R. P packing material polar (silica) non-polar (C 18) sep. interaction polarity & size (weak to medium polar) n-hexane/polar solvent n-hexane CH 2 Cl 2 Ethyl acetate Me. CN (strong to medium polar) low polarity (alkanes) high polarity (salts) Eluent weak stron g most rapidly eluented cpd. H 2 O/organic H 2 O Me. OH Me. CN THF CH 2 Cl 2

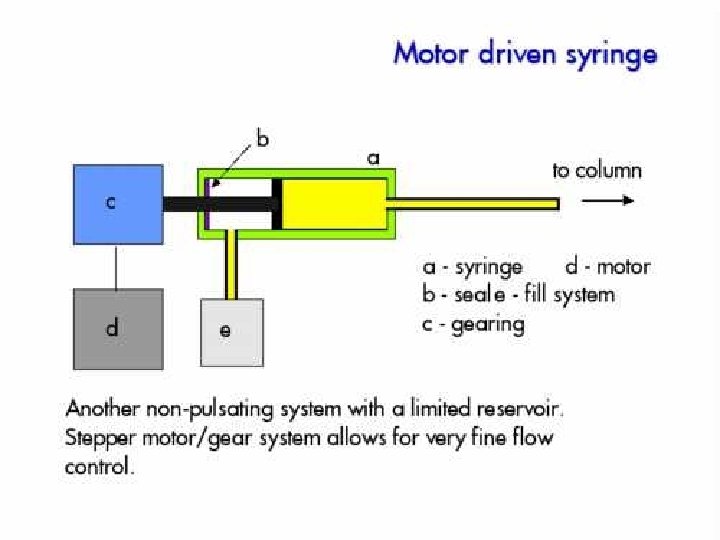
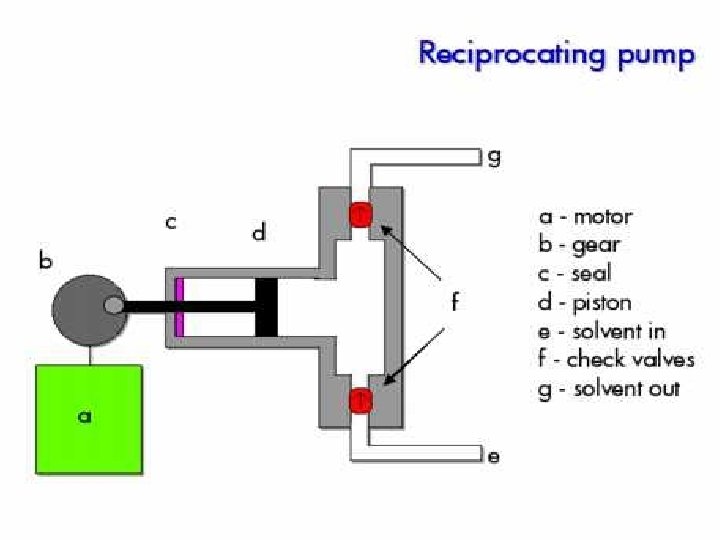
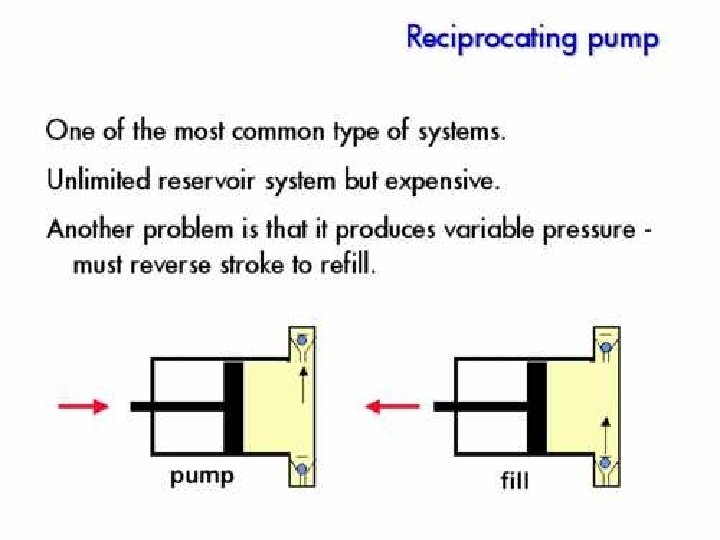
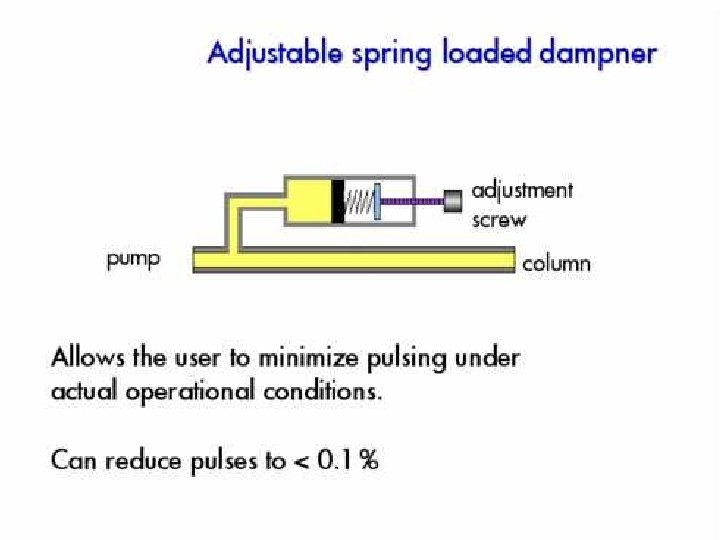
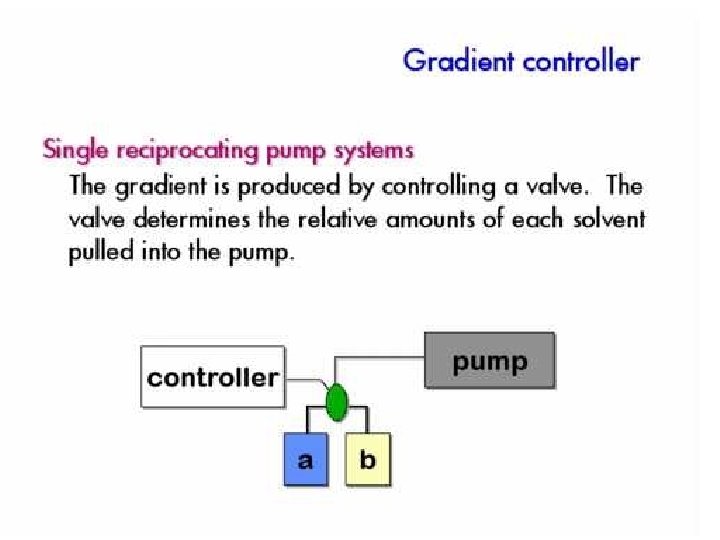
Pump • Motor driven syringe • Piston – Reciprocating – Multiple reciprocating • Advantage and disadvantage – Solvent reservoir limited? – Pressure pulse produced? – Gradient can be produced?







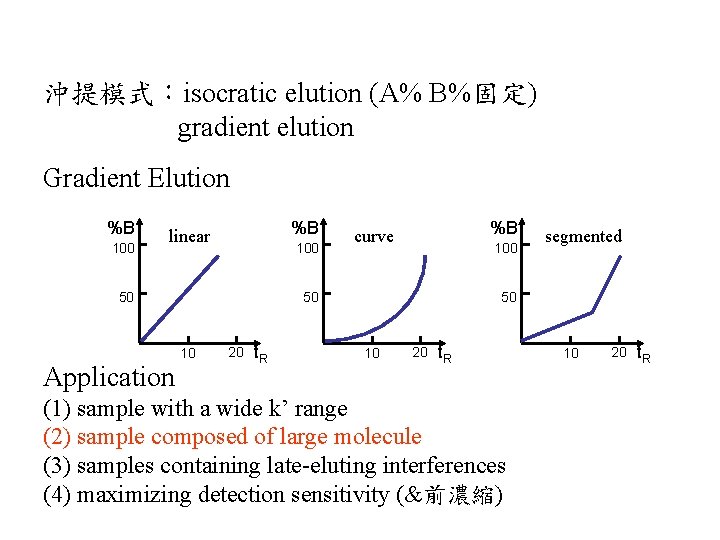
沖提模式:isocratic elution (A% B%固定) gradient elution Gradient Elution %B 100 %B linear 100 50 Application %B curve 100 50 10 20 t. R segmented 50 10 20 t. R (1) sample with a wide k’ range (2) sample composed of large molecule (3) samples containing late-eluting interferences (4) maximizing detection sensitivity (&前濃縮) 10 20 t. R



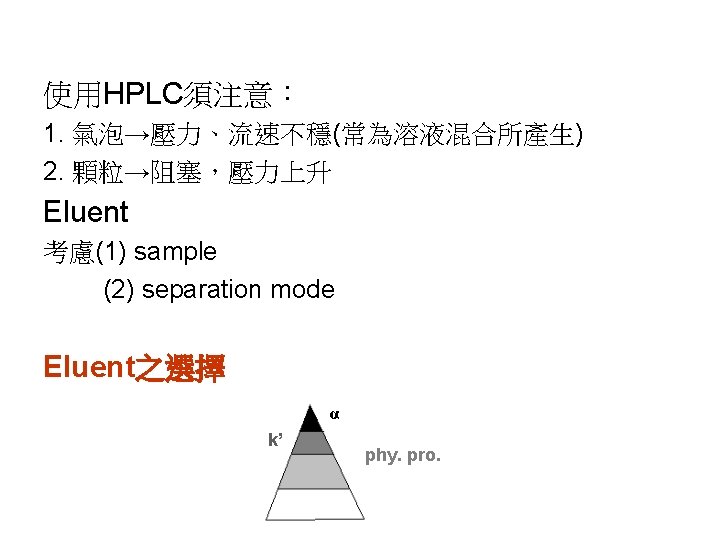
(1) phy. properties b. p. 、viscosity、UV cutoff、毒性、純度、價格 UV cutoff: (2) 1 ≤ k’ ≤ 10 for simple components 0. 5 ≤ k’ ≤ 25 for multiple components solute/eluent interaction↑ k’↓ solute S. P M. P


4 major interaction (a) dispersion interaction (b) dipole interaction (c) H-bonding interaction (d) dielectric interaction dispersion force: x (非極性分子) s Temporacy dipole moment from x will polarize the e in adjacent molecule s (R. I. 值大表示分散力大)

eluent strength solvent polarity N. P. solvent polarity ↑ k’↓ R. P. solvent polarity ↑ k’↓ N. P. P’:polarity R. P.

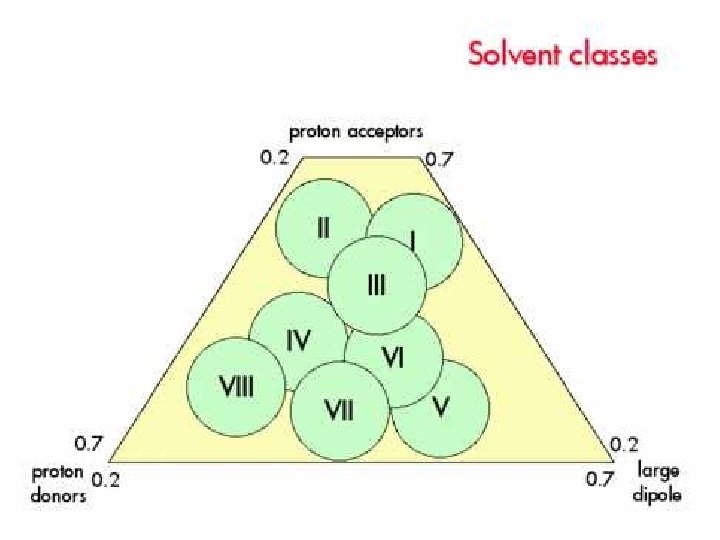
solvent mixture :volume ratio lf solvent a, b 30% Me. OH 70% H 2 O 40% Me. OH 60% H 2 O (3) solvent selectivity proton donor 由proton acceptor 將solvent分類 dipole moment


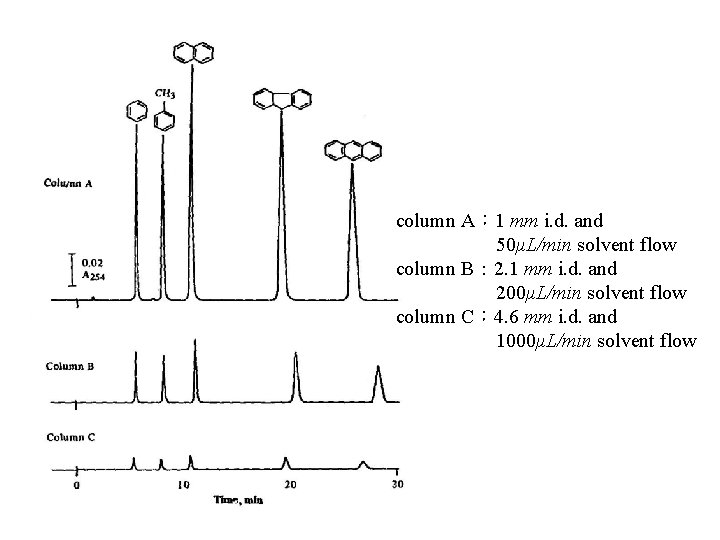
由physical size分類 column I. D. L 1. Analytical column: 2. 1~5. 0 mm 10, 15, 25 cm 2. microbore column: 1. 0~2. 0 mm ‘’ ‘’ 3. capillary column: ‘’ ‘’ 同樣濃度,peak大小: A>B>C A: 1 mm B: 2. 1 mm C: 4. 6 mm 50 μL/min 0. 2 m. L/min 1 m. L/min

column A: 1 mm i. d. and 50μL/min solvent flow column B: 2. 1 mm i. d. and 200μL/min solvent flow column C: 4. 6 mm i. d. and 1000μL/min solvent flow


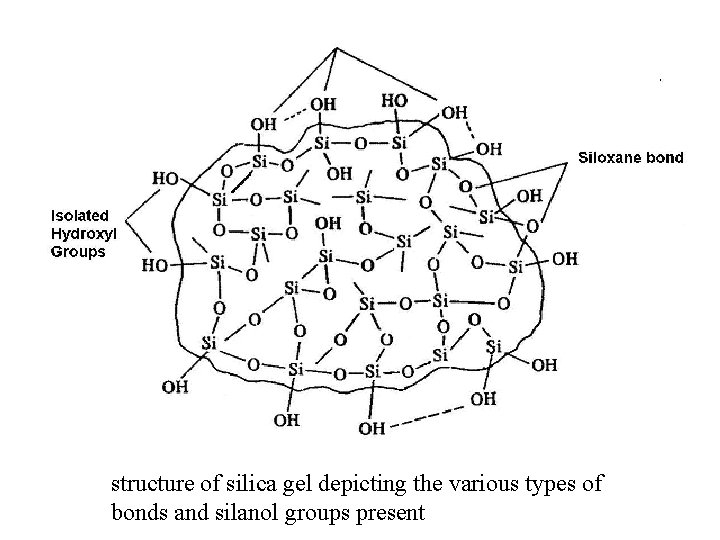
structure of silica gel depicting the various types of bonds and silanol groups present

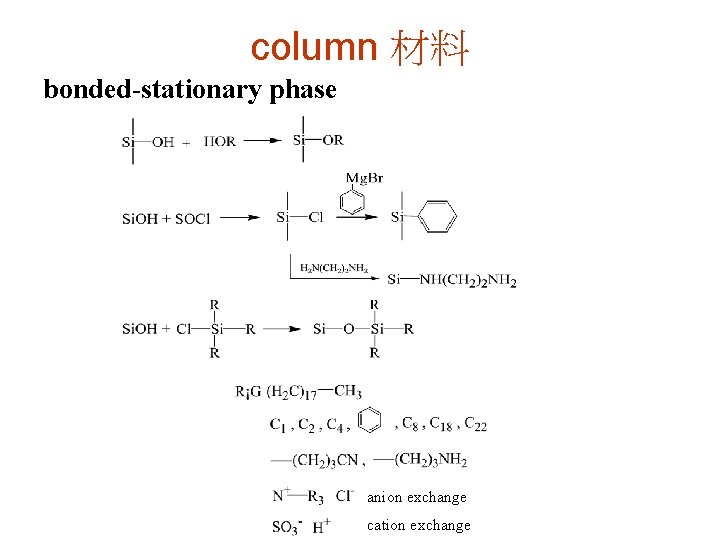
column 材料 bonded-stationary phase anion exchange cation exchange

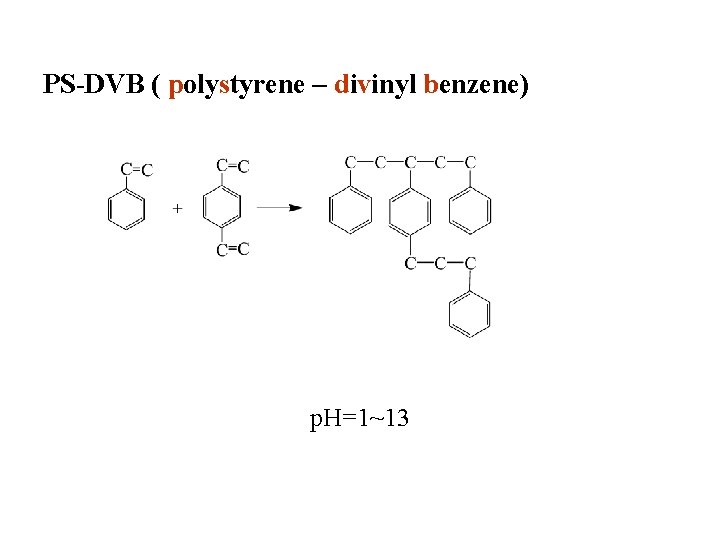
PS-DVB ( polystyrene – divinyl benzene) p. H=1~13

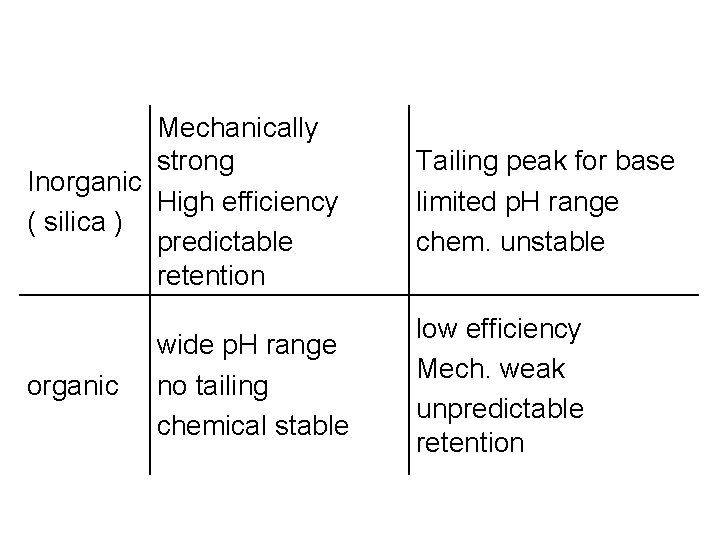
Mechanically strong Inorganic High efficiency ( silica ) predictable retention organic wide p. H range no tailing chemical stable Tailing peak for base limited p. H range chem. unstable low efficiency Mech. weak unpredictable retention

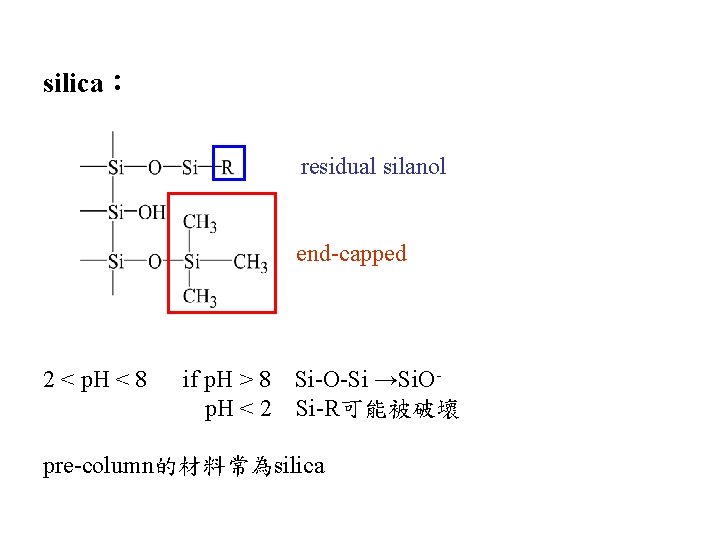
silica: residual silanol end-capped 2 < p. H < 8 if p. H > 8 Si-O-Si →Si. Op. H < 2 Si-R可能被破壞 pre-column的材料常為silica

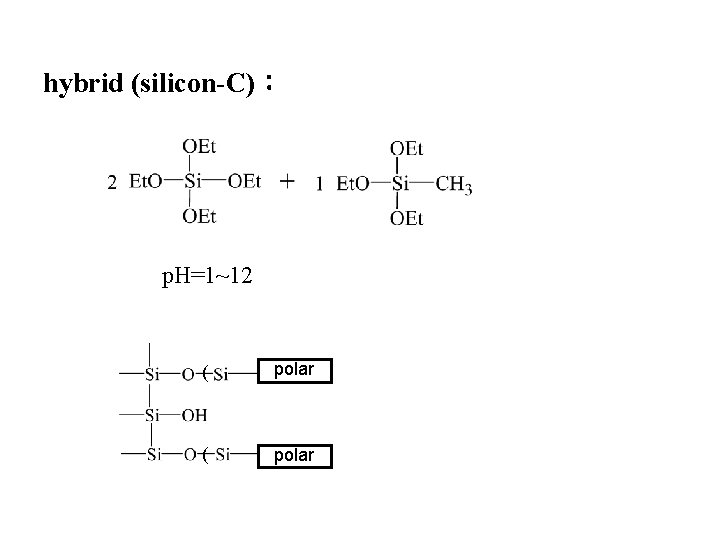
hybrid (silicon-C): p. H=1~12 ( polar

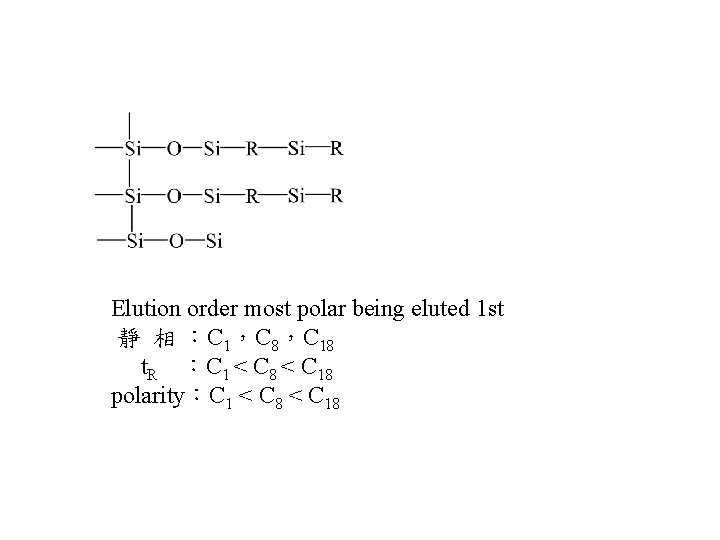
Elution order most polar being eluted 1 st 靜 相 :C 1,C 8,C 18 t. R :C 1 < C 8 < C 18 polarity:C 1 < C 8 < C 18

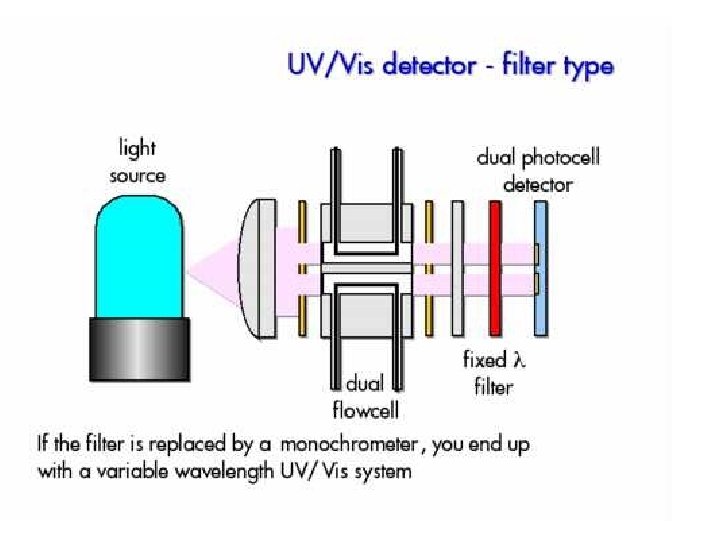
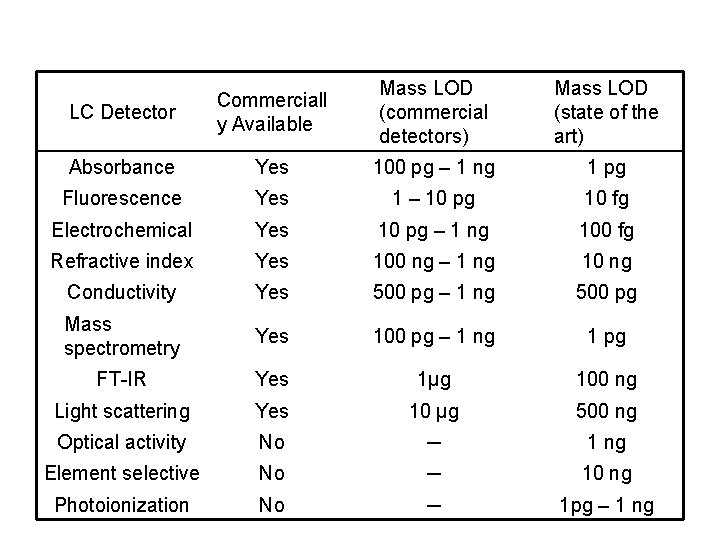
HPLC detectors • Bulk property detectors – respond to a mobile phase bulk property – refractive index, dielectric constant, or density • Solute property detectors – respond to some property of solutes – UV absorbance, fluorescence, or diffusion current


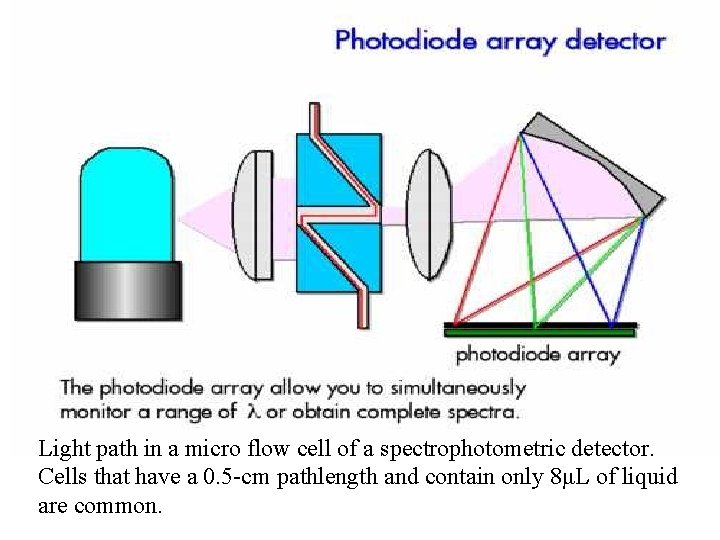
Light path in a micro flow cell of a spectrophotometric detector. Cells that have a 0. 5 -cm pathlength and contain only 8μL of liquid are common.


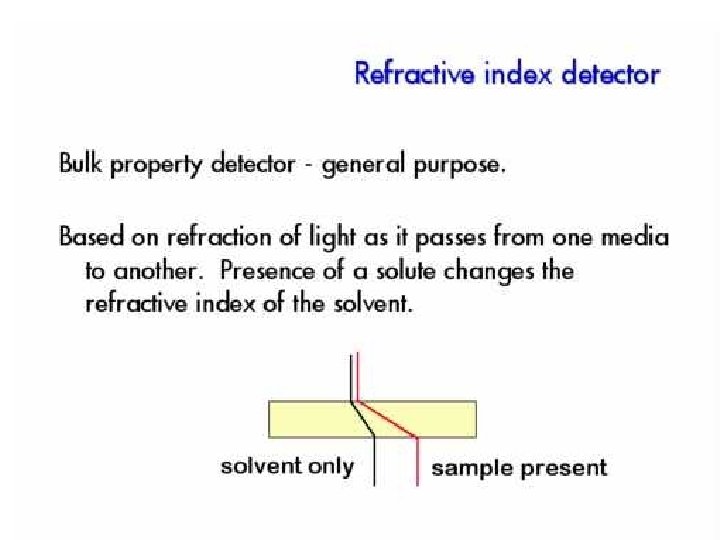
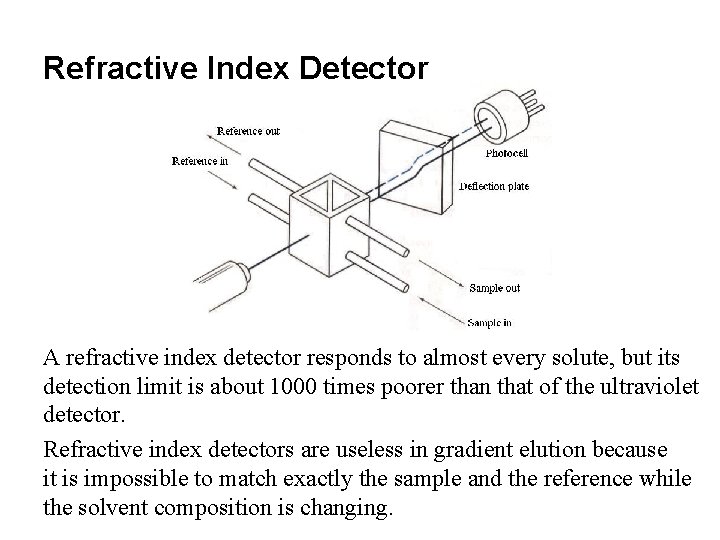
Refractive Index Detector A refractive index detector responds to almost every solute, but its detection limit is about 1000 times poorer than that of the ultraviolet detector. Refractive index detectors are useless in gradient elution because it is impossible to match exactly the sample and the reference while the solvent composition is changing.

Fluorescence detectors excite the eluant with a laser and measure the fluorescence. These detectors are very sensitive but respond only to the few analytes that fluoresce.

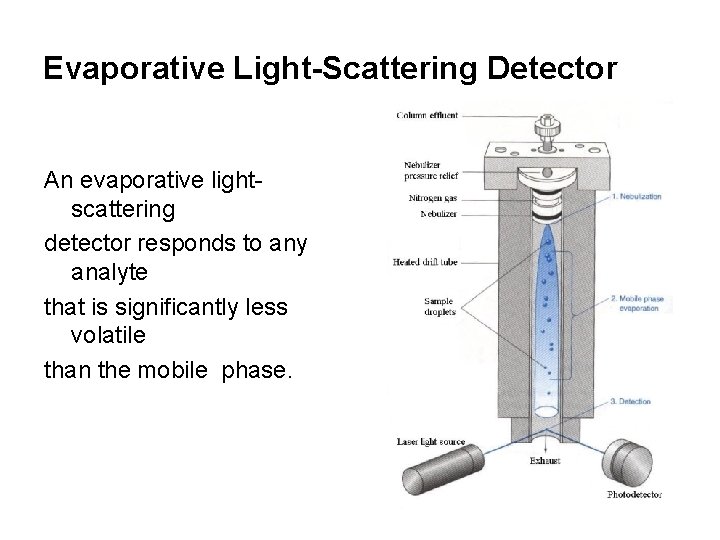
Evaporative Light-Scattering Detector An evaporative lightscattering detector responds to any analyte that is significantly less volatile than the mobile phase.

LC Detector Commerciall y Available Mass LOD (commercial detectors) Mass LOD (state of the art) Absorbance Yes 100 pg – 1 ng 1 pg Fluorescence Yes 1 – 10 pg 10 fg Electrochemical Yes 10 pg – 1 ng 100 fg Refractive index Yes 100 ng – 1 ng 10 ng Conductivity Yes 500 pg – 1 ng 500 pg Mass spectrometry Yes 100 pg – 1 ng 1 pg FT-IR Yes 1μg 100 ng Light scattering Yes 10 μg 500 ng Optical activity No ─ 1 ng Element selective No ─ 10 ng Photoionization No ─ 1 pg – 1 ng

ionic water soluble non-ionic M<2000 polar organic soluble non-polar sample water soluble M>2000 organic soluble IC HPLC ion pair HPLC normal phase HPLC reversed phase SEC gel fitration HPLC reverse phase IC SEC gel fitration

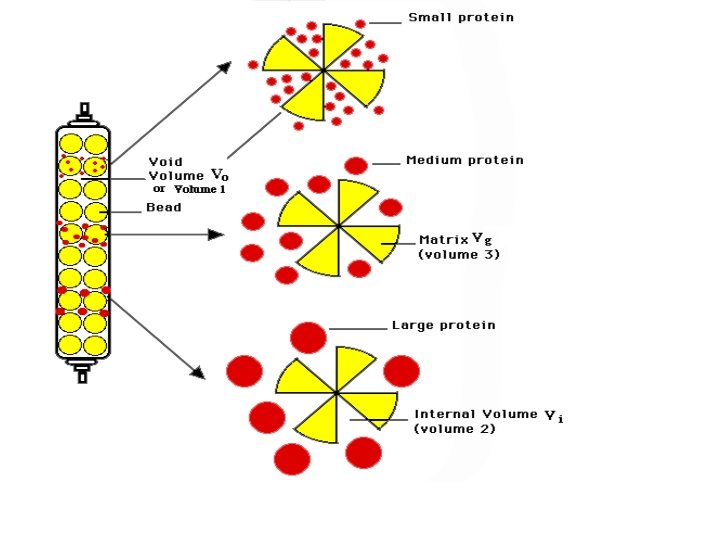
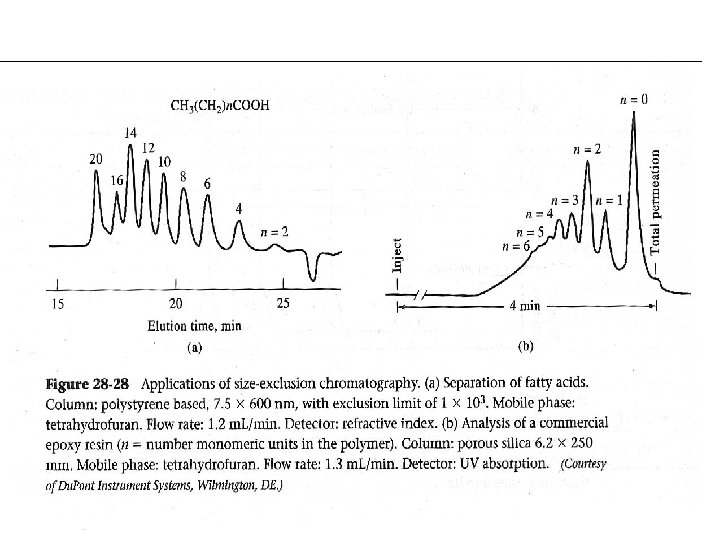
Size-Exclusion Chromatography (SEC) ‧固定相︰ silica or polymer particles with pore size 102 -106 Å ‧ Type︰ -Gel Filtration Chromatography (GFC) stationary phase: hydrophilic mobile phase: aqueous 適用: protein, amino acid, peptide. -Gel Permeation Chromatography (GPC) stationary phase: hydrophobic mobile phase: nonpolar solvent 適用: oligomers


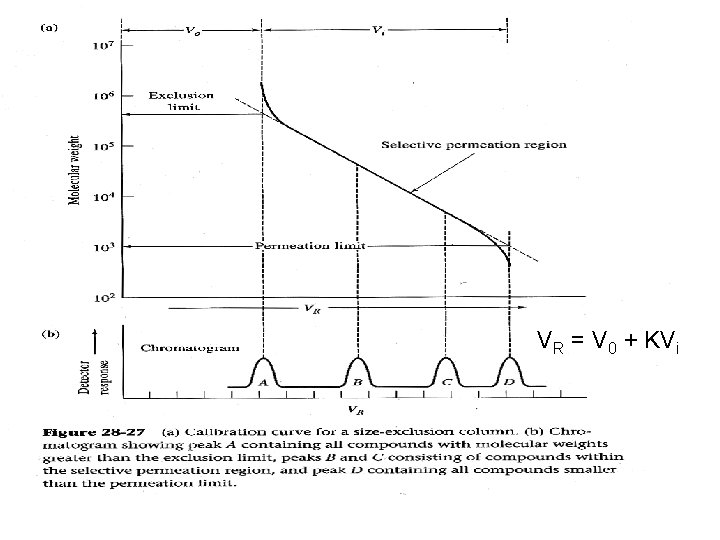
VR = V 0 + KVi


Applications: 1. Molecular weight determination 2. Group fraction

ionic water soluble non-ionic M<2000 polar organic soluble non-polar sample water soluble M>2000 organic soluble IC HPLC ion pair HPLC normal phase HPLC reversed phase SEC gel fitration HPLC reverse phase IC SEC gel fitration

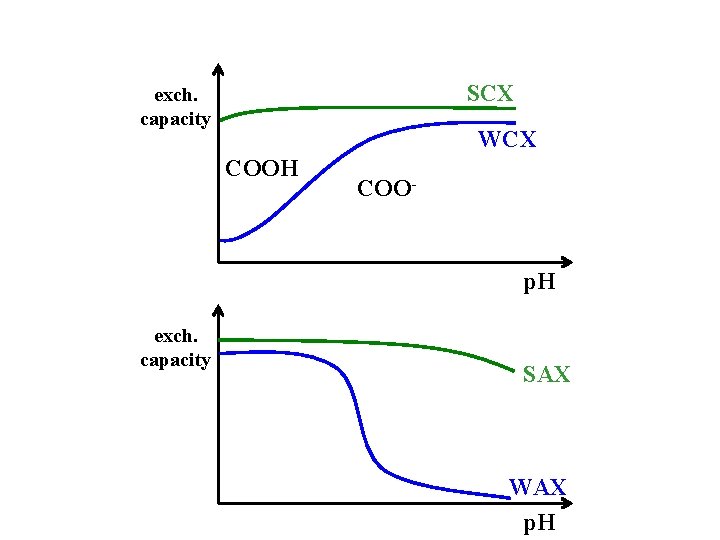
Ion-exchange chromatography sample ion:X mobile phase:Y charged stationary phase:R X- + R + Y- Y- + R + X- anion exchange X + + R -Y + + R -X + cation exchange SCX:strong cation exchange –SO 3 -H+ WCX:weak cation exchange –COOH SAX:strong anion exchange –NR 3+OHWAX:weak anion exchange –NH 2

SCX exch. capacity WCX COOH COO- p. H exch. capacity SAX WAX p. H

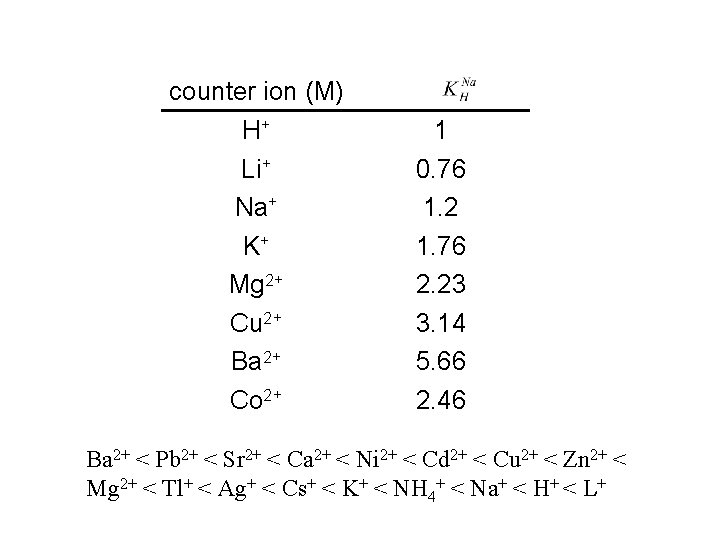
selectivity R-SO 3 -H+(R) + Na+ (S)+ Cl- R-SO 3 -Na+(R) + H+ (S)+ Cl- selectivity coefficient:

counter ion (M) H+ Li+ Na+ K+ Mg 2+ Cu 2+ Ba 2+ Co 2+ 1 0. 76 1. 2 1. 76 2. 23 3. 14 5. 66 2. 46 Ba 2+ < Pb 2+ < Sr 2+ < Ca 2+ < Ni 2+ < Cd 2+ < Cu 2+ < Zn 2+ < Mg 2+ < Tl+ < Ag+ < Cs+ < K+ < NH 4+ < Na+ < H+ < L+

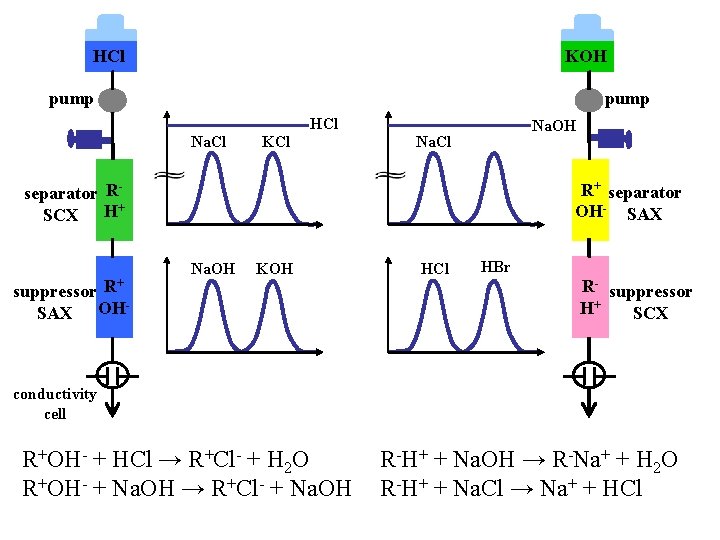
HCl KOH pump Na. Cl KCl HCl Na. OH Na. Cl separator RSCX H+ R+ R+ separator OH- SAX Na. OH KOH suppressor SAX OH HCl HBr R- suppressor H+ SCX conductivity cell R+OH- + HCl → R+Cl- + H 2 O R+OH- + Na. OH → R+Cl- + Na. OH R-H+ + Na. OH → R-Na+ + H 2 O R-H+ + Na. Cl → Na+ + HCl

ionic water soluble non-ionic M<2000 polar organic soluble non-polar sample water soluble M>2000 organic soluble IC HPLC ion pair HPLC normal phase HPLC reversed phase SEC gel fitration HPLC reverse phase IC SEC gel fitration

Ion-pair chromatography (IPC) IPC & RP-HPLC share several features: column and mobile phase and similar Compounds:weak acids, bases, ionic compound Concept:solvent extraction Aaq- + Baq- (A+B- )aq A+B-org ion-pair aq:aqueous phase org:organic phase →Ionized compound Aaq+ in water add suitable counter ion Baqion-pair A+B- similar to non-ionic compound →In solvent extraction constant

capacity (reaction) factor

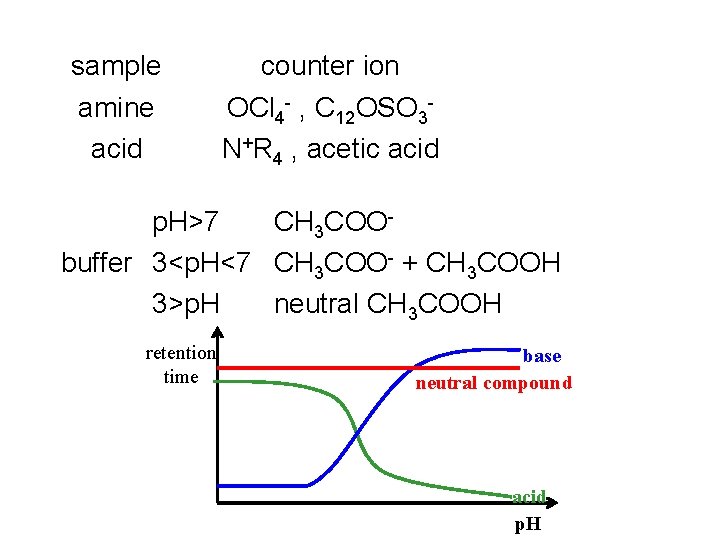
sample amine acid counter ion OCl 4 - , C 12 OSO 3 N+R 4 , acetic acid p. H>7 CH 3 COObuffer 3<p. H<7 CH 3 COO- + CH 3 COOH 3>p. H neutral CH 3 COOH retention time base neutral compound acid p. H

Electrochemical Detector An electrochemical detector responds to analytes that can be oxidized or reduced. The detector is very sensitive to flow rate and temperature changes.

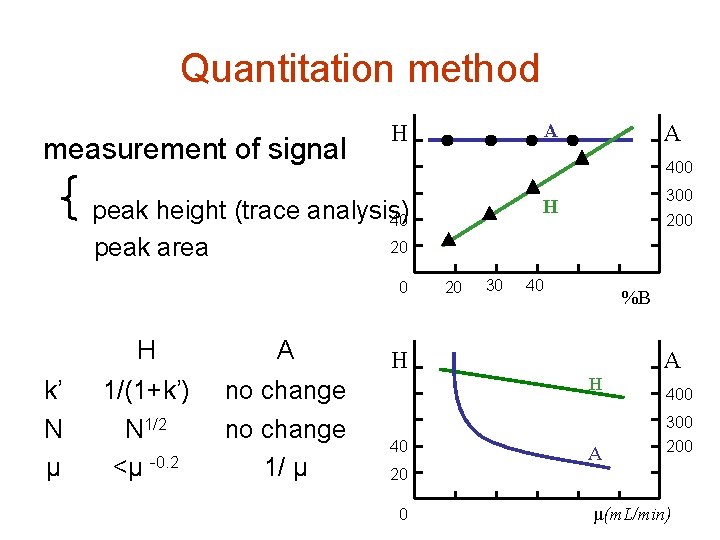
Quantitation method measurement of signal A H 400 peak height (trace analysis) 40 20 peak area 0 k’ N μ H A 1/(1+k’) N 1/2 <μ -0. 2 no change 1/ μ A 300 H 20 30 200 40 %B H A H 400 300 40 A 200 20 0 μ(m. L/min)

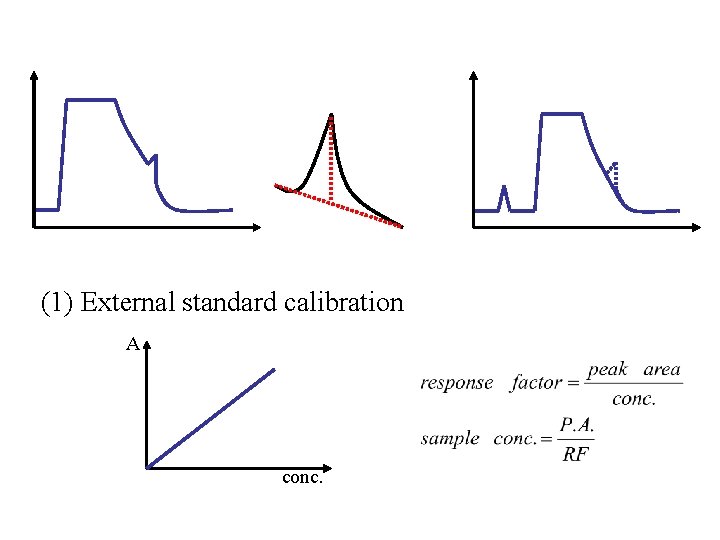
(1) External standard calibration A conc.

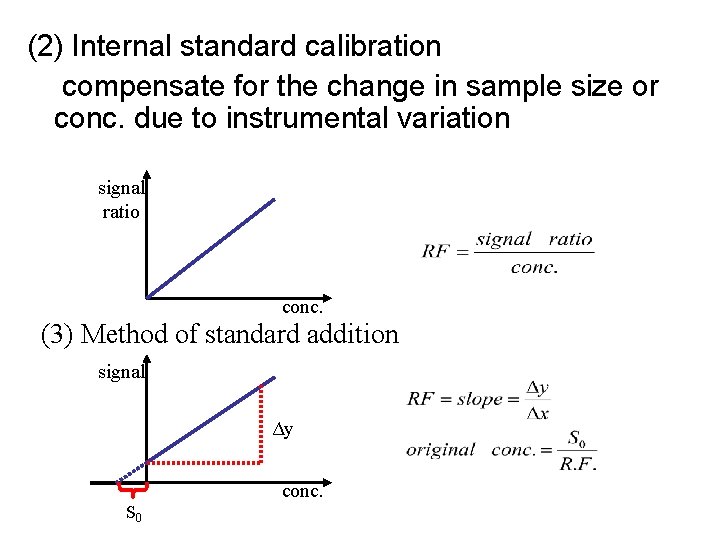
(2) Internal standard calibration compensate for the change in sample size or conc. due to instrumental variation signal ratio conc. (3) Method of standard addition signal Δy conc. S 0

Error (1) constant systematic error (2) proportional systematic error S=a+bc S=b’c (1) (2) S=bc

Recovery (sample preparation) (1) pure reference standard (2) pure reference standard + sample preparation xp:mass or conc. of ref. analyte after sample preparation (3) xp → constant systematic error → proportional systematic error (4) recovery rate x 0