Choose a category You will be given the

Choose a category. You will be given the answer. You must give the correct question. Click to begin.

Click here for Final Jeopardy
What’s the matter? No one understands the cloud! The name’s bond. Chemical Bond Junk Drawer Chemistry + lump of stuff from = love Unit 1 Junk Drawer 10 Point 10 Point 20 Points 20 Points 30 Points 30 Points 40 Points 40 Points 50 Points 50 Points

You recorded the length of an object to be 1. 93 cm. How many sig figs does the # have and what is the uncertain digit?

1. 93 cm has 3 sig figs and the ‘ 3’ is the uncertain digit.

A sample has a mass of 3. 5 g and a volume of 0. 075 L. What is the density in g/m. L ?

What is 0. 0467 g/m. L?

Which of the following are isotopes? Atom 1 2 3 Protons 12 12 13 Neutrons 12 13 12 Electrons 12 12 13 4 14 13 14
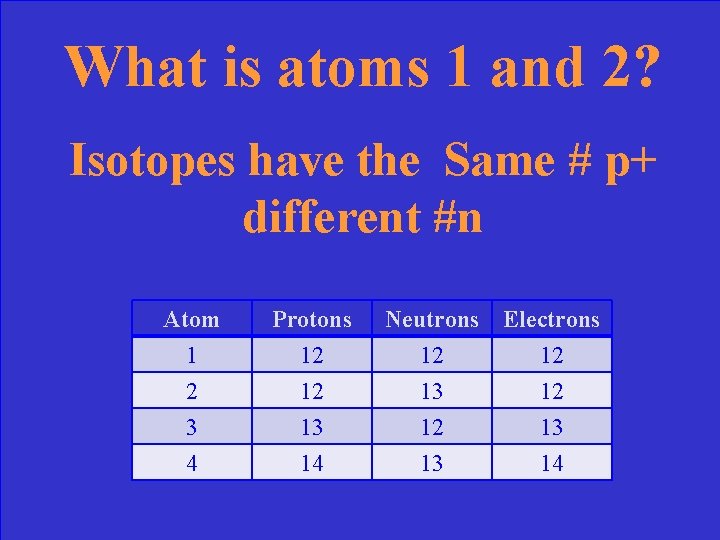
What is atoms 1 and 2? Isotopes have the Same # p+ different #n Atom 1 2 3 Protons 12 12 13 Neutrons 12 13 12 Electrons 12 12 13 4 14 13 14

Hydrogen has three isotopes. . Hydrogen-1, hydrogen-2, and hydrogen-3. Which is most abundant in nature?

What is hydrogen-1, because its mass is closest to hydrogen’s average atomic mass of 1. 01 amu ?

Complete the following nuclear reaction: ?

What is…

What is the subatomic particle with a charge of +1, a mass of 1 amu, and is located inside the nucleus?

What are protons?

How many protons 3+ and electrons does Al have?

What is 13 protons and 10 electrons? It has 3 more p+ than electrons to have a charge of 3+

Why does Ca and Mg have similar chemical properties?

What is they have the same number of valence electrons ? (they are both in 2 A)

How many valence electrons does this element have?
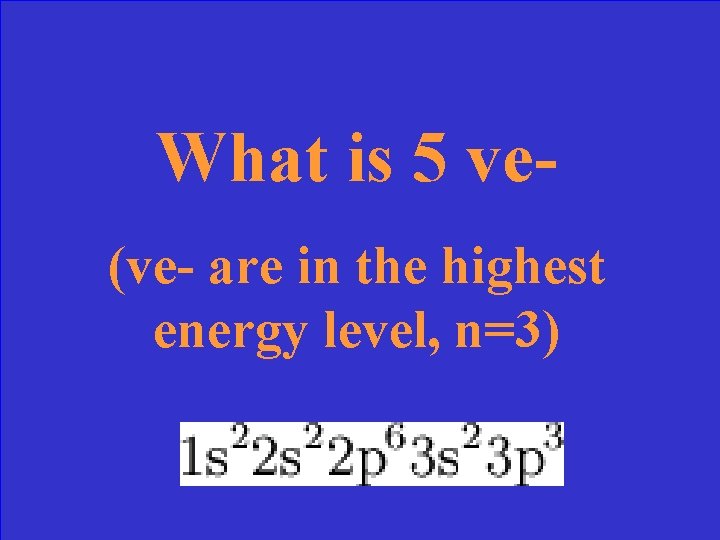
What is 5 ve(ve- are in the highest energy level, n=3)

What is the charge on the ion this element would most likely form?

It would gain 3 veto have a charge of 3 -

What is the name of… Ca. F 2 P 3 O 5 Cr 2(SO 4)

Ca. F 2 = calcium fluoride P 3 O 5 = triphosphorus pentoxide Cr 2(SO 4) = chromium (I) sulfate

What are the formulas for. . Copper (II) bromide Carbon tetrahydride

Copper (II) bromide = Cu. Br 2 Carbon tetrahydride = CH 4

What is the shape AND polarity of NF 3 ?

What is trigonal pyramidal and polar because the F pull the ve- more

What are the diatomic elements? AND What is their molecular polarity?

What is H 2 N 2 O 2 F 2 Cl 2 Br 2 I 2 ? They are all nonpolar due to an equal ‘pull’ of ve-.

What type of covalent bond is the weakest (single triple or double? )

Single bonds are the weakest.

How many atoms are in 400 grams of Ca? 6. 02 x 1023 representative particles = 1 mole

24 6. 01 x 10 What is Ca atoms?
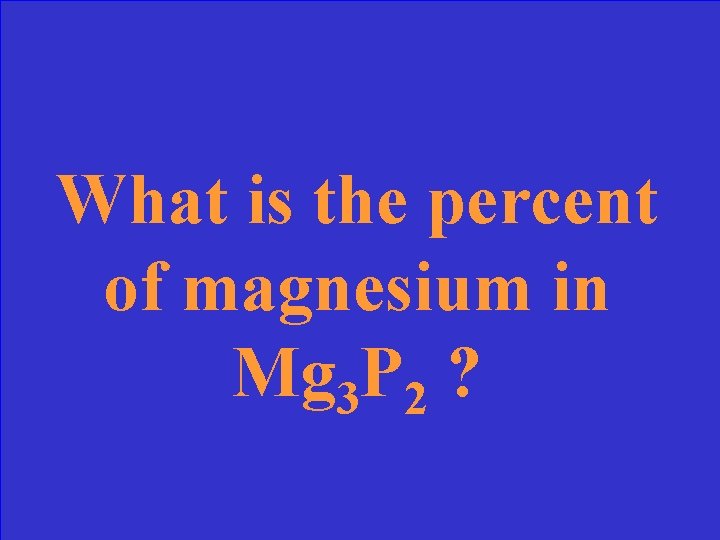
What is the percent of magnesium in Mg 3 P 2 ?

What is 54. 1% Mg ? Mass of Mg / total mass of compound x 100 = 72. 93 / 134. 87 x 100

A compound has an empirical formula of CH 2 O. The molar mass of the compound is 180 g/mole. What is the molecular formula of this compound?

What is C 6 H 12 O 6 because 180 g/mol is 6 times as big as 30 g/mol so the molecular formula has 6 x the number of moles of each element in CH 2 O

What is the concentration when 60 g of Mg. Cl 2 are dissolved in 1. 5 L of solution?

What is the concentration when 60 g of Mg. Cl 2 are dissolved in 1. 5 L of solution?

How many moles of Na. OH are in 400 m. L of a 2 M solution?
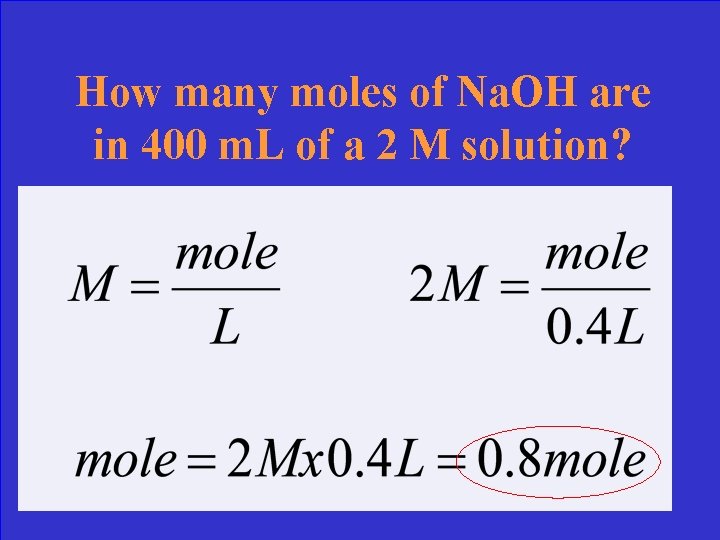
How many moles of Na. OH are in 400 m. L of a 2 M solution?

Why does this emission spectrum show discrete lines instead of continuous streams of light?
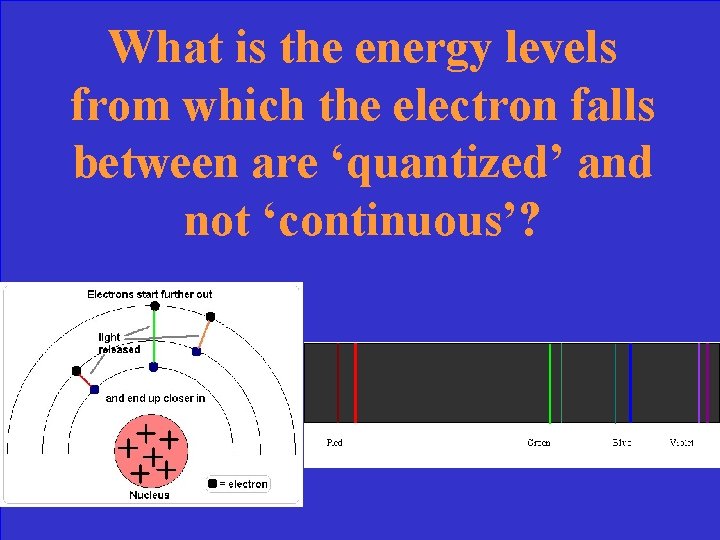
What is the energy levels from which the electron falls between are ‘quantized’ and not ‘continuous’?

_____ is the energy needed to remove an electron and _______ is the attraction an atom has for electrons in a bond.

Ionization energy is the energy needed to remove an electron and electronegativity is the attraction an atom has for electrons in a bond.

How many total atoms are in 3 Cu 3(PO 4)2 ?

3 Cu 3(PO 4)2 has 39 atoms… Cu 3(PO 4)2 is Cu Cu Cu PO 4 3 Cu + 2 P + 8 O = 13 atoms But we have 3 sets of the compound so 3 x 13 = 39 atoms

Which is larger… Mg or P or 2+ Mg 3 P

2+ Mg Mg or because by losing 2 e- to become 2+ it loses an energy level 3 P P or because by gaining 3 e- there is more e- repulsion

Describe these solutions as saturated, unsaturated, or supersaturated. 1. The solution has more solute dissolved than what is typical at that temperature 2. The solution has the maximum amount of solute dissolved at that temperature.

1. The solution has more solute dissolved than what is typical at that temperature = supersaturated 2. The solution has the maximum amount of solute dissolved at that temperature. = saturated

Make your wager
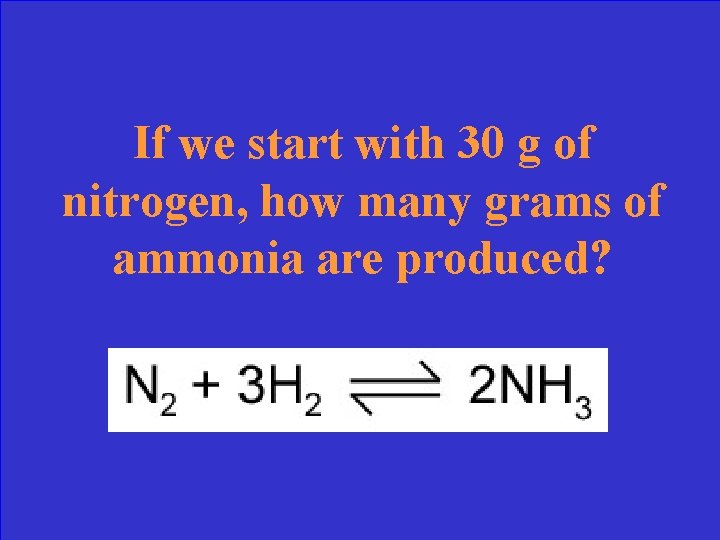
If we start with 30 g of nitrogen, how many grams of ammonia are produced?

What is 36. 4 grams NH 3 ?
- Slides: 56