Cholesterol Synthesis Transport Excretion BIOMEDICAL IMPORTANCE Cholesterol is

Cholesterol Synthesis, Transport, & Excretion BIOMEDICAL IMPORTANCE Cholesterol is present in tissues and in plasma either as free cholesterol or combined with a long-chain fatty acid as cholesteryl ester, the storage form. In plasma, both forms are transported in lipoproteins. Cholesterol is an amphipathic lipid and as such is an essential structural component of membranes and of the outer layer of plasma lipoproteins.

It is synthesized in many tissues from acetyl. Co. A and is the precursor of all other steroids in the body, including corticosteroids, sex hormones, bile acids, and vitamin D. As a typical product of animal metabolism, cholesterol occurs in foods of animal origin such as egg yolk, meat, liver, and brain. Plasma low-density lipoprotein (LDL) is the vehicle of uptake of cholesterol and cholesteryl ester into many tissues.

Free cholesterol is removed from tissues by plasma high-density lipoprotein (HDL) and transported to the liver, where it is eliminated from the body either unchanged or after conversion to bile acids in the process known as reverse cholesterol transport. Cholesterol is a major constituent of gallstones. However, its chief role in pathologic processes is as a factor in the genesis of atherosclerosis of vital arteries, causing cerebrovascular, coronary, and peripheral vascular disease.

CHOLESTEROL IS DERIVED ABOUT EQUALLY FROM THE DIET & FROM BIOSYNTHESIS A little more than half the cholesterol of the body arises by synthesis (about 700 mg/d), and the remainder is provided by the average diet. The liver and intestine account for approximately 10% each of total synthesis in humans. Virtually all tissues containing nucleated cells are capable of cholesterol synthesis, which occurs in the endoplasmic reticulum and the cytosol.

Acetyl-Co. A Is the Source of All Carbon Atoms in Cholesterol The biosynthesis of cholesterol may be divided into five steps: (1) Synthesis of mevalonate from acetyl-Co. A (Figure 26– 1). (2) Formation of isoprenoid units from mevalonate by loss of CO 2 (Figure 26– 2). (3) Condensation of six isoprenoid units form squalene (Figure 26– 2). (4) Cyclization of squalene give rise to the parent steroid, lanosterol. (5) Formation of cholesterol from lanosterol (Figure 26– 3).
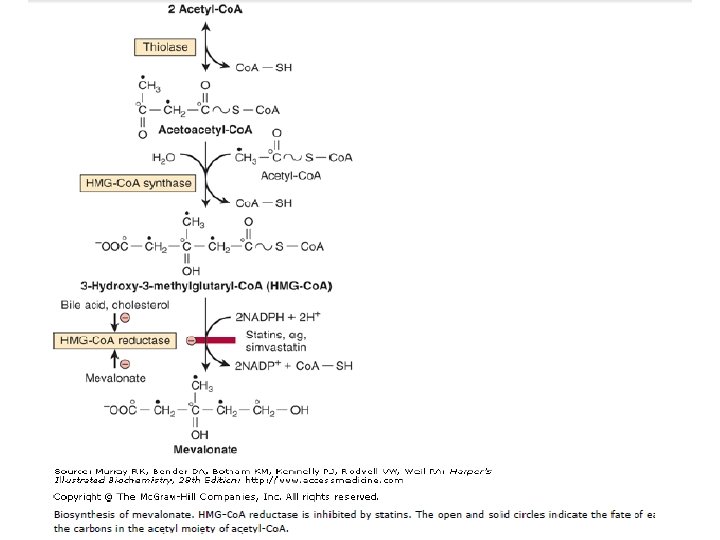

Step 1—Biosynthesis of Mevalonate: HMGCo. A (3 -hydroxy-3 -methylglutaryl-Co. A) is formed by the reactions used in mitochondria to synthesize ketone bodies (Figure 22– 7). However, since cholesterol synthesis is extramitochondrial, the two pathways are distinct. Initially, two molecules of acetyl-Co. A condense to form acetoacetyl-Co. A catalyzed by cytosolic thiolase.

Acetoacetyl-Co. A condenses with a further molecule of acetyl-Co. A catalyzed by HMGCo. A synthase to form HMG-Co. A, which is reduced to mevalonate by NADPH catalyzed by HMG-Co. A reductase. This is the principal regulatory step in the pathway of cholesterol synthesis and is the site of action of the most effective class of cholesterol-lowering drugs, the statins, which are HMG-Co. A reductase inhibitors (Figure 26– 1).

CHOLESTEROL SYNTHESIS IS CONTROLLED BY REGULATION OF HMGCo. A REDUCTASE Regulation of cholesterol synthesis is exerted near the beginning of the pathway, at the HMG-Co. A reductase step. The reduced synthesis of cholesterol in starving animals is accompanied by a decrease in the activity of the enzyme.

• However, it is only hepatic synthesis that is inhibited by dietary cholesterol. HMG-Co. A reductase in liver is inhibited by mevalonate, the immediate product of the pathway, and by cholesterol, the main product. Cholesterol and metabolites repress transcription of the HMGCo. A reductase via activation of a sterol regulatory elementbinding protein (SREBP) transcription factor. SREBPs are a family of proteins that regulate the transcription of a range of genes involved in the cellular uptake and metabolism of cholesterol and other lipids.

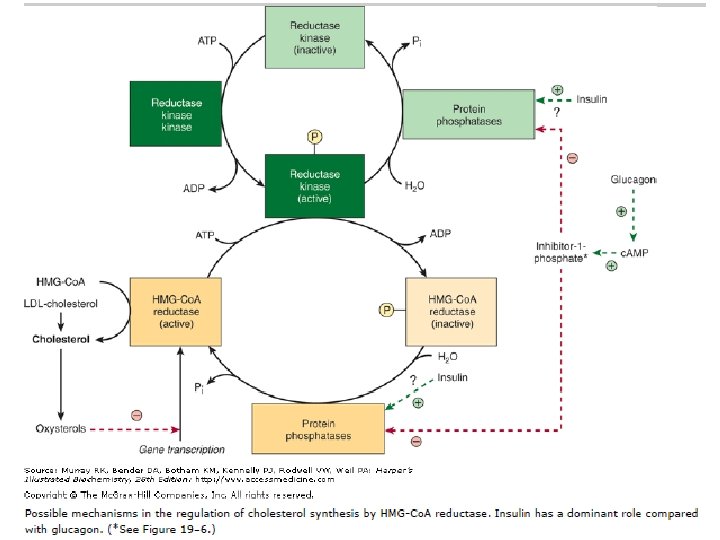
A diurnal variation occurs in both cholesterol synthesis and reductase activity. In addition to these mechanisms regulating the rate of protein synthesis, the enzyme activity is also modulated more rapidly by post-translational modification (Figure 26– 4). Insulin or thyroid hormone increases HMG-Co. A reductase activity, whereas glucagon or glucocorticoids decrease it.

Activity is reversibly modified by phosphorylation-dephosphorylation mechanisms, some of which may be c. AMPdependent and therefore immediately responsive to glucagon. Attempts to lower plasma cholesterol in humans by reducing the amount of cholesterol in the diet produce variable results. Generally, a decrease of 100 mg in dietary cholesterol causes a decrease of approximately 0. 13 mmol/L of serum
- Slides: 13