CHLORINATION DISINFECTION PHYSICAL UV RADIATION HEAT MEMBRANE FILTERS

CHLORINATION

DISINFECTION: PHYSICAL: UV RADIATION, HEAT, MEMBRANE FILTERS CHEMICAL: CHLORINE, OZONE, CHLORINE DIOXIDE, IODINE, OTHER ANTIMICROBIAL CHEMICALS

� to inactivate pathogens so that they are not infectious to humans and animals E. coli, Campylobacter, Shigella � Giardia lamblia, Cryptosporidia � Viruses � � achieved by altering or destroying structures or functions of essential components within the pathogens proteins (structural proteins, enzymes, transport proteins, etc) � nucleic acids (genomic DNA or RNA, m. RNA, t. RNA, etc) � lipids (lipid bi-layer membranes, other lipids) �

MECHANISM OF ACTION OF CHLORINE � Cl enters the cell wall of bacteria � Inactivates the enzymes needed for metabolic processes � Affecting the permeability of bacterial cell membrane resulting into the leakage of cellular components
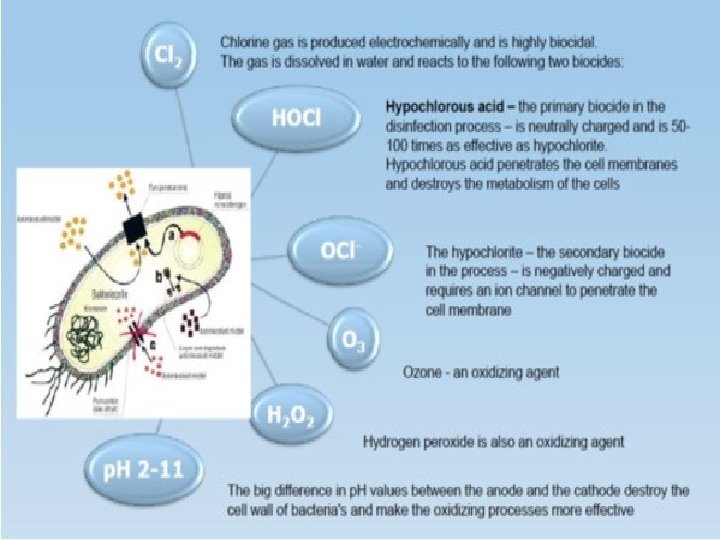
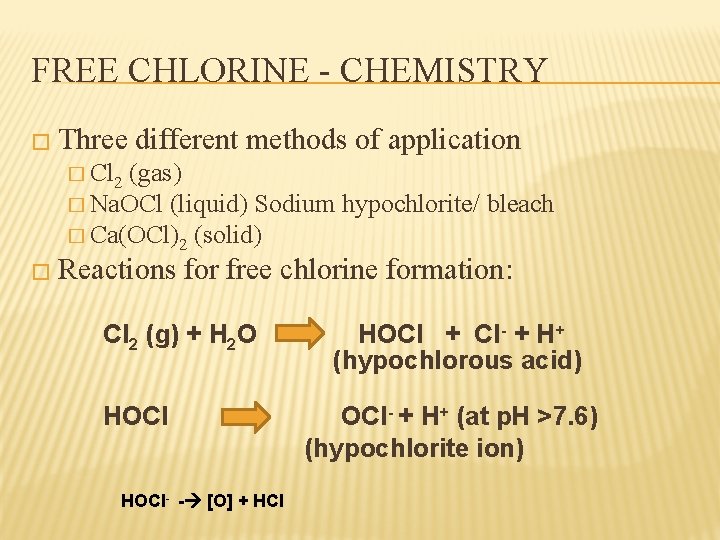
FREE CHLORINE - CHEMISTRY � Three different methods of application � Cl 2 (gas) � Na. OCl (liquid) Sodium hypochlorite/ bleach � Ca(OCl)2 (solid) � Reactions for free chlorine formation: Cl 2 (g) + H 2 O HOCl- - [O] + HCl HOCl + Cl- + H+ (hypochlorous acid) OCl- + H+ (at p. H >7. 6) (hypochlorite ion)

CHLORAMINE FORMATION � HOCl and OCl are called free residual chlorine � Free chlorine compounds+ organic/inorganic compounds chorinated copmounds Eg. Formation of chloramines: � HOCl + NH 3 => NH 2 Cl + H 2 O (mono) � NH 2 Cl + HOCl => NHCl 2 + H 2 O (di) � NHCl 2 + HOCl => NCl 3 + H 2 O (tri)

CHLORINE DEMAND � Free chlorine compounds have highest disinfecting power � The difference in the amount of chlorine added to free residual chlorine is called chlorine demand � Chlorine demand = chlorine added – free chlorine

METHODS OF CHLORINATION � Plain chlorination � Super chlorination � Breakpoint chlorination

PLAIN CHLORINATION � Addition of Cl in excess to Cl demand � Well water/ borewells � Dosage= 0. 5 -1. 0 mg/L � Residual Cl= 0. 1 -0. 2 mg/L

SUPER CHLORINATION � Attempts to kill bacteria fails � Introduction of a chlorinated solution of not less than 1000 ppm in the well water � Excess of Cl can be removed by dechlorinating agents- Sodium thiosulphate/ SO 2

BREAKPOINT CHLORINATION � Breakpoint Chlorination- is when enough chlorine is added to satisfy the chlorine demand to react with all the dissolved ammonia.



CHLORINE (ADVANTAGES AND DISADVANTAGES) � Advantages Effective against all types of microbes � Relatively simple maintenance and operation � Inexpensive � � Disadvantages Corrosive � High toxicity � High chemical hazard � Highly sensitive to inorganic and organic loads � Formation of harmful disinfection by-products (DBP’s) �

CHLORAMINES (ADVANTAGES AND DISADVANTAGES) � Advantages Less corrosive � Less toxicity and chemical hazards � Relatively tolerable to inorganic and organic loads � No known formation of DBP � Relatively long-lasting residuals � � Disadvantages � Not so effective against viruses, protozoan cysts, and bacterial spores
- Slides: 16