CHLORAMPHENICOL Introduction Chloramphenicol is a broadspectrum antibiotic Chloramphenicol

CHLORAMPHENICOL

Introduction • Chloramphenicol is a broad-spectrum antibiotic. • Chloramphenicol is a bacteriostatic antimicrobial • With the drug's wide use, it became evident that chloramphenicol could cause serious and fatal blood dyscrasias. For this reason, chloramphenicol is now reserved for treatment of life-threatening infections (e. g. , meningitis, rickettsial infections) in patients who cannot take safer alternatives because of resistance or allergies

Source • Originally derived from the bacterium called Streptomyces Venezuelae, isolated by David Gottlieb, in 1947. • But nowa days it is produced synthetically.

Description: Chloramphenicol is a white crystalline compound that is soluble in alcohal but slightly soluble in water and it is bitter in taste. So sometimes we need to increase its solubility when it is intended to be administered by IV route and sometimes we need to mask its taste when it administered in the form of suspension. Thus chloramphenicol palmitate is Tasteless but insoluble in water and is used in suspension while chloramphenicol succinate is soluble in water but bitter in taste and is used for injections.
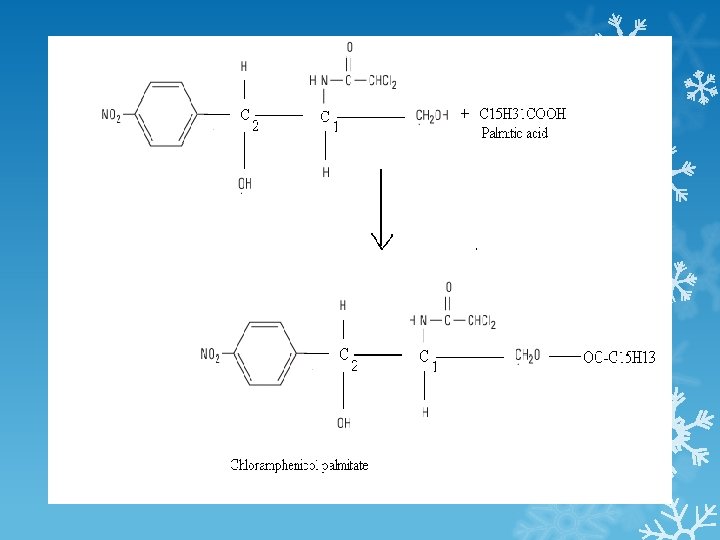

PK The usual dosage of chloramphenicol is 50 -100 mg/kg/d. Chloramphenicol palmitate is a prodrug that is hydrolyzed in the intestine to yield free chloramphenicol. The parenteral formulation is a prodrug, chloramphenicol succinate, which hydrolyzes to yield free chloramphenicol, giving blood levels somewhat lower than those achieved with orally administered drug. Chloramphenicol is widely distributed to virtually all tissues and body fluids, including the central nervous system and cerebrospinal fluid, such that the concentration of chloramphenicol in brain tissue may be equal to that in serum.

PARAMETER Route of administration Metabolism Excretion Half Life Protein binding capacity VALUE Oral, IV In Liver by conjugation Kidney, breast milk 1. 5 – 3. 5 hours 60%

The drug penetrates cell membranes readily. Most of the drug is inactivated either by conjugation with glucuronic acid (principally in the liver) or by reduction to inactive aryl amines. Active chloramphenicol (about 10% of the total dose administered) and its inactive degradation products (about 90% of the total) are eliminated in the urine. A small amount of active drug is excreted into bile and feces. The systemic dosage of chloramphenicol need not be altered in renal insufficiency, but it must be reduced markedly in hepatic failure. Newborns less than a week old and premature infants also clear

chloramphenicol less well, and the dosage should be reduced to 25 mg/kg/d. N) Parenteral administration of Chloramphenicol is generally reserved for situations in which oral therapy is contraindicated, as in the treatment of meningitis and septicemia or when vomiting prohibits oral administration.

Antimicrobial Spectrum: Chloramphenicol is a bacteriostatic broad-spectrum antibiotic that is active against both aerobic and anaerobic gram-positive and gram-negative organisms. It is active also against rickettsiae but not chlamydiae. H influenzae, S. Typhi, N meningitidis, and some strains of bacteroides are highly susceptible. It is not active against Pseudomonas aeruginosa or Enterobacter species.

Antibacterial activity H. Influenzae S. typhi N. Meningitidis E. coli S. Pneumoniae V. cholera Ricketsiae Anaerobes- clostridium &

Therapeutic Uses § Therapy with chloramphenicol must be limited to infections for which the benefits of the drug outweigh the risks of the potential toxicities. When other antimicrobial drugs are available that are equally effective and potentially less toxic than chloramphenicol, they should be used

Chloramphenicol has a wide range activity that includes gram+, gram-, aerobic and anaerobic bacteria Typhoid Fever Bacterial Meningitis Anaerobic Infections Rickettsial Diseases Brucellosis

Chloramphenicol Clinical uses Limited because of potential toxicities (a plastic anaemia &) 1. Typhoid fever- s. typhi ( quinolones are preffered) 2. Meningitis – H. influenzae, N. meningitidis, S. pneumoniae ( Ceftriaxone is preffered ) 3. Anaerobic infections- B. fragilis (Metronidazole is the drug of choice) 4. Rickettsial infections – Doxycycline is preffered 5. Bacterial conjunctivitis ( topical )
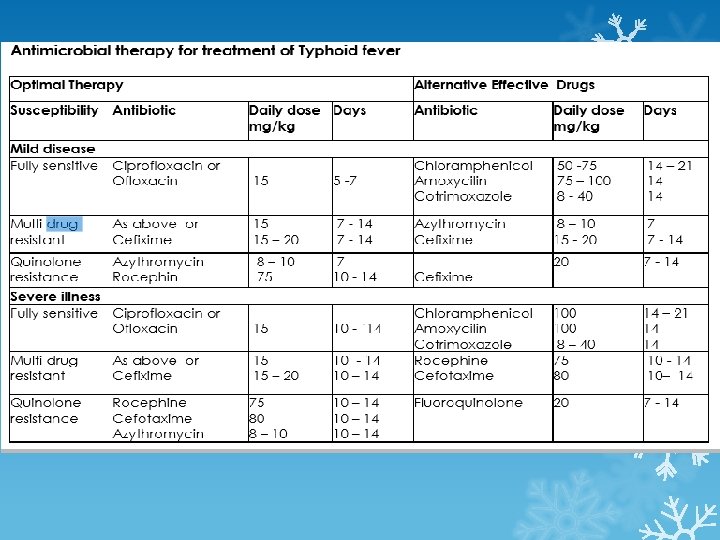

Exp Cloramphenicol is not DOC b/c of its adverse effects, bacterial resistance and availability of better agents. 1. Chloramphenicol is very effective against H. influenzae meningitis. It is an alternative to a β lactam antibiotic for treatment of meningococcal meningitis occurring in patients who have major hypersensitivity reactions to penicillin or bacterial meningitis caused by penicillin resistant strains of pneumococci. The dosage is 50 -100 mg/kg/d in four divided doses. 2. Severe rickettsial infections

3. As drops or ointment, it is used for eye or ear infections. 4. Chloramphenicol was the DOC for enteric fever but now it is on reserve list due to availability of safer drugs (Ciprofloxacin). 5. Since effective CSF levels are obtained, it used to be a choice for treatment of specific bacterial causes of meningitis: Haemophilus influenzae, Neisseria meningitidis, and Strepcoccus pneumoniae.

Mechanism of Action § Chloramphenicol inhibits protein synthesis in bacteria and, to a lesser extent, in eukaryotic cells. The drug readily penetrates bacterial cells, probably by facilitated diffusion.

Chloramphenicol is a potent inhibitor of microbial protein synthesis. It binds reversibly to the 50 S subunit of the bacterial ribosome and inhibits the peptidyl transferase thus inhibiting the peptide bond formation b/w peptide chain at P site and amino acid at site A (protein synthesis)

Fig. Steps in bacterial protein synthesis and targets of several antibiotics. Amino acids are shown as numbered circles. The 70 S ribosomal m. RNA complex is shown with its 50 S and 30 S subunits. In step 1, the charged t. RNA unit carrying amino acid 8 binds to the acceptor site A on the 70 S ribosome. The peptidyl t. RNA at the donor site, with amino acids 1 through 7, then binds the growing amino acid chain to amino acid 8 (transpeptidation, step 2). The uncharged t. RNA left at the donor site is released (step 3), and the new

8 -amino acid chain with its t. RNA shifts to the peptidyl site (translocation, step 4). The antibiotic binding sites are shown schematically as triangles. Chloramphenicol (C) and macrolides (M) bind to the 50 S subunit and block transpeptidation (step 2). The tetracyclines (T) bind to the 30 S subunit and prevent binding of the incoming charged t. RNA unit (step 1). Note Chloramphenicol and the macrolide class of antibiotics both interact with the 50 S ribosomal subunit, chloramphenicol is not a macrolide. Furthermore, their mechanisms are slightly different. While chloramphenicol directly interferes with substrate binding, macrolides sterically block the progression of the growing peptide

MOA (S) (I) It acts by inhibiting protein synthesis in bacteria by binding reversibly to 50 S subunit and preventing the binding of amino aceyl t. RNA to the acceptor site on the 50 S subunit. It interferes with the interaction b/w peptidyl transferase and amino acid, thereby preventing peptide bond formation.

MOA (L) (i) Chloramphenicol (Chloromycetin) is a nitrobenzene derivative that affects protein synthesis by binding to the 50 S ribosomal subunit and preventing peptide Bond formation. It prevents the attachment of the amino acid end of aminoacyl-t. RNA to the A site, hence the association of peptidyltransferase with the amino acid substrate.
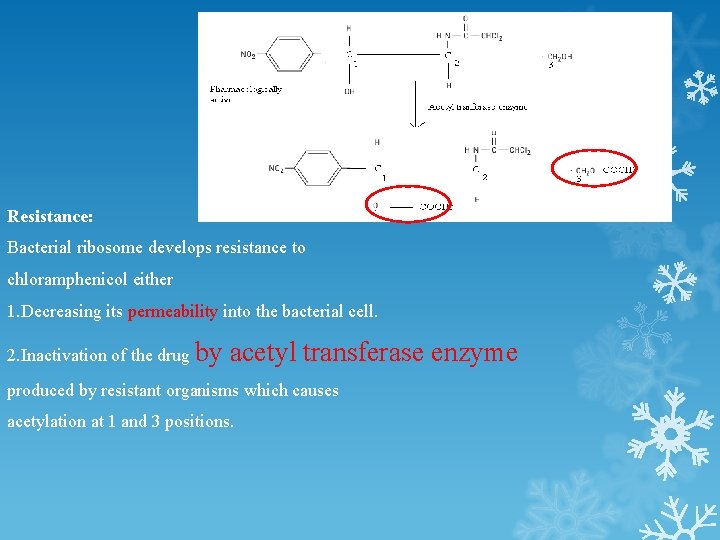
Resistance: Bacterial ribosome develops resistance to chloramphenicol either 1. Decreasing its permeability into the bacterial cell. 2. Inactivation of the drug by acetyl transferase enzyme produced by resistant organisms which causes acetylation at 1 and 3 positions.

Interaction with other drugs Chloramphenicol inhibits hepatic microsomal enzymes that metabolize several drugs. Half-lives are prolonged, and the serum concentrations of phenytoin, tolbutamide, chlorpropamide, Cyclophosphamide and warfarin are increased. Some drugs like chronic administration of phenobarbitone or acute administration of rifampicin may increase the elimination of chloramphenicol by enzyme induction. Like other bacteriostatic inhibitors of microbial protein synthesis, chloramphenicol can antagonize bactericidal drugs such as penicillins or aminoglycosides.

Adverse Reactions

Adverse Reactions-----------------1. Gastrointestinal disturbances These include Nausea, vomiting, and diarrhea. The chances of intestinal superinfection (unlike tetracycline) are rare b/c it is completely absorbed from GIT. 2. Bone marrow depression (Aplastic anemia) when used for 1 -2 weeks or more. 3. Toxicity for newborn infants (Neonates) Newborn infants lack an effective glucuronic acid conjugation mechanism for the degradation and

detoxification of chloramphenicol. Consequently, when infants are given dosages above 50 mg/kg/d, the drug may accumulate, resulting in the gray baby syndrome, with vomiting, flaccidity, hypothermia, gray color, shock, and collapse. To avoid this toxic effect, chloramphenicol should be used with caution in infants and the dosage limited to 50 mg/kg/d or less (during the first week of life) in full-term infants more than 1 week old and 25 mg/kg/d in premature infants.

Toxicity in neonates; If neonates, specially premature babies, are exposed to 75 mg/kg/day or more of chloramphenicol, chloramphenicol toxicity, commonly called gray baby syndrome may develop wihin two to three days of administration of the drug. It starts with vomiting, refusal to suck, irregular and rapid respiration, abdominal distension, periods of cynosis, with loose green stool. Within another twenty four hours they become flaccid, develop hypothermia and turns ashes gray. Metabolic acidosis may appear and death occurs in 40% of the patients.

The mechanisms responsible for he gray syndrome are 1. Failure of the drug to be conjugated with glucuronic acid due to in adequate activity of glucuronyl transferase in the liver in the neonate period 2. Inadequate renal excretion of unconjugated drug in neonate.

Preparations available: 1. Chloromax by Pharmedic Inj. 1 gm chloramphenicol (As sodium succinate) 2. Chloromycetin by parke-Davis/Pfizer Cap- 250 mg Chloramphenicol base Susp: Per 5 m. L: Chloramphenicol palmitate equiv. to 125 mg chloramphenicol base. 3. Neo-Phenicol by PDH Susp: Per 5 m. L: Chloramphenicol palmitate equiv. to 125 mg chloramphenicol base.

Dosage (I) The usual dose is 50 mg/kg/day in four divided doses: the usual dose in an adult male is therefore around 750 mg four times daily; this dose is doubled in severe illness. Half the dose is used in premature babies or neonates, because they do not metabolise the drug as effectively. chloramphenicol is sold as chloramphenicol palmitate ester. Chloramphenicol palmitate ester is inactive, and is hydrolysed to active chloramphenicol in the small intestine. There is no difference in bioavailability between chloramphenicol and chloramphenicol

palmitate. The intravenous (IV) preparation of chloramphenicol is the succinate ester, because pure chloramphenicol does not dissolve in water. This creates a problem: chloramphenicol succinate ester is an inactive prodrug and must first be hydrolysed to chloramphenicol; the hydrolysis process is incomplete and 30% of the dose is lost unchanged in the urine, therefore serum concentrations of chloramphenicol are only 70% of those achieved when chloramphenicol is given orally. For this reason, the chloramphenicol dose needs to be increased to 75 mg/kg/day when administered IV in

order to achieve levels equivalent to the oral dose. The oral route is therefore preferred to the intravenous route. Chloramphenicol and the liver Chloramphenicol is metabolised by the liver to chloramphenicol glucuronate (which is inactive). In liver impairment, the dose of chloramphenicol must therefore be reduced. There is no standard dose reduction for chloramphenicol in liver impairment, and the dose should be adjusted according to measured plasma concentrations. Chloramphenicol is also noted for its cause of "Gray Baby Syndrome" because of infants lack of the enzyme glucoronyl transferase which is the main pathway.

Clinical features Toxic levels of chloramphenicol after 2– 9 days result in: 1. Vomiting 2. Ashen gray color of the skin 3. Limp body tone 4. Hypotension (low blood pressure) 5. Cyanosis blue discolouration of lips and skin. 5. Hypothermia 6. Cardiovascular collapse

Treatment Chloramphenicol therapy is discontinued immediately; exchange transfusion may be required to remove the drug. Prevention The condition can be prevented by using chloramphenicol at the recommended doses and monitoring blood levels, or alternatively, third generation cephalosporins can be effectively substituted for the drug, without the associated toxicity

Cynosis: Bluish coloration of the skin due to low level of oxygen in blood. Hemolytic anemia: It is characterized by rupture of RBC’s. Agranulocytosis: Decrease in no. of granulocytes. Pancytopenia The decrease in the number of RBC’s , WBCs and Platelets.
- Slides: 38