Chirality chiral Greek for hands chiral nonsuperimposable mirror

Chirality “chiral” Greek for hands chiral = non-superimposable mirror images chiral = no plane of symmetry C* C with 4 different substituents stereocenter chiral C
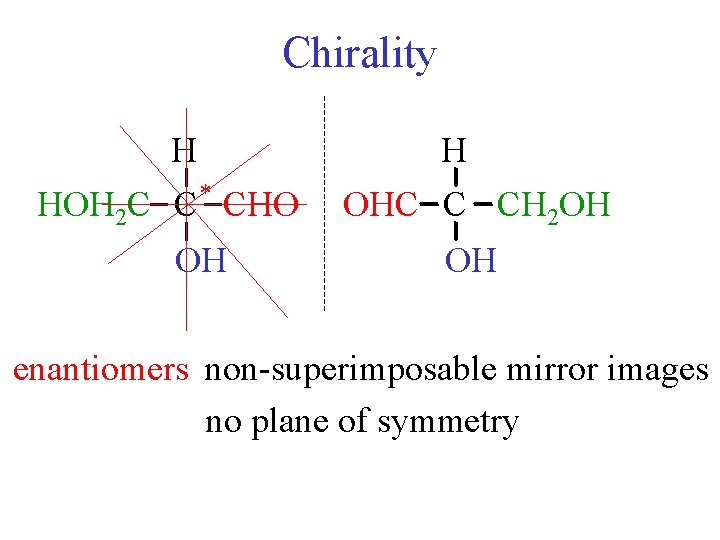
Chirality H HOH 2 C C * CHO OH H OHC C CH 2 OH OH enantiomers non-superimposable mirror images no plane of symmetry
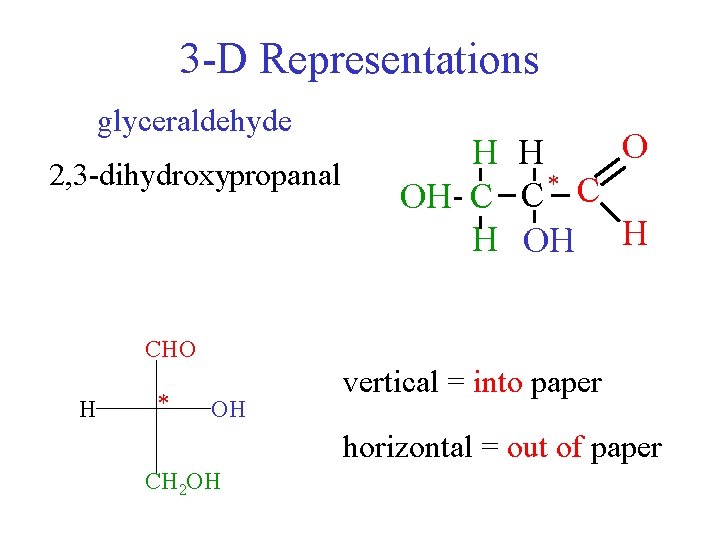
3 -D Representations glyceraldehyde 2, 3 -dihydroxypropanal O H H C HOH C C * CHO OH 2 C H OH H CHO H * OH vertical = into paper horizontal = out of paper CH 2 OH
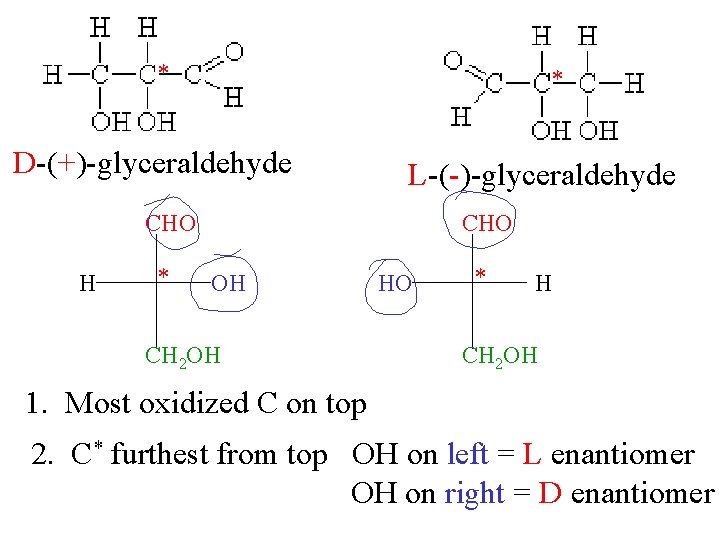
* * D-(+)-glyceraldehyde L-(-)-glyceraldehyde CHO H * CHO OH CH 2 OH HO * H CH 2 OH 1. Most oxidized C on top 2. C* furthest from top OH on left = L enantiomer OH on right = D enantiomer

Enantiomers identical in most properties differ in: 1. interaction with polarized light 2. interaction with chiral environments Light interacts with molecules when it passes through them [interaction of electrical fields] When light encounters mirror image of molecule, interaction is reversed

Polarized light One enantiomer - rotate light to the left (-) Other enantiomer - rotate light to the right (+) in 50/50 mix - no net rotation racemic mixture one enantiomer - polarized light will be rotated optical activity

Chirality 2. Enantiomers in chiral environments hands chiral mittens achiral - plane of symmetry achiral environments don’t distinguish between enantiomers gloves chiral - no plane of symmetry chiral environments do distinguish between enantiomers
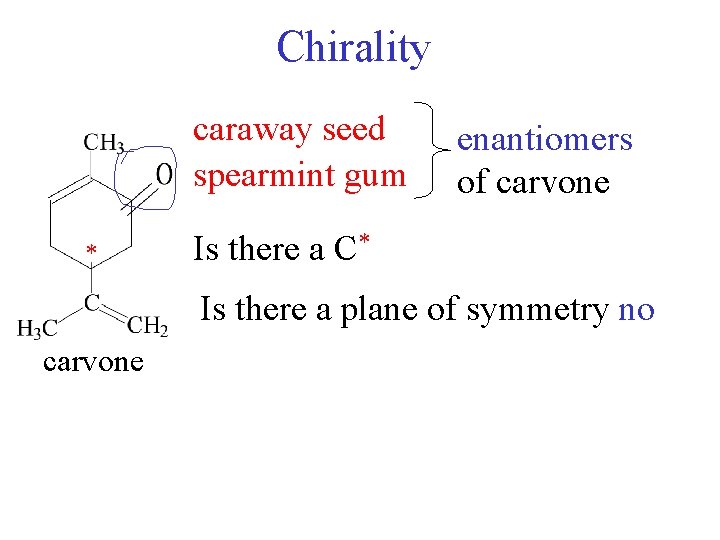
Chirality caraway seed spearmint gum * enantiomers of carvone Is there a C* Is there a plane of symmetry no carvone
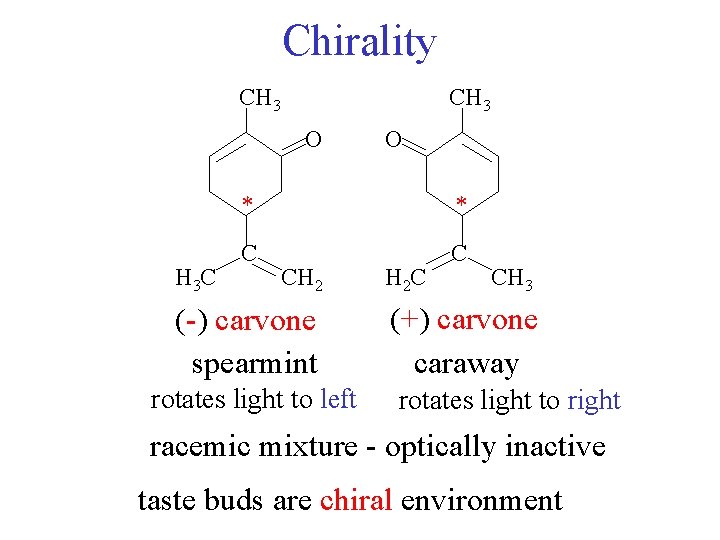
Chirality CH 3 O H 3 C O * * C C CH 2 (-) carvone spearmint rotates light to left H 2 C CH 3 (+) carvone caraway rotates light to right racemic mixture - optically inactive taste buds are chiral environment
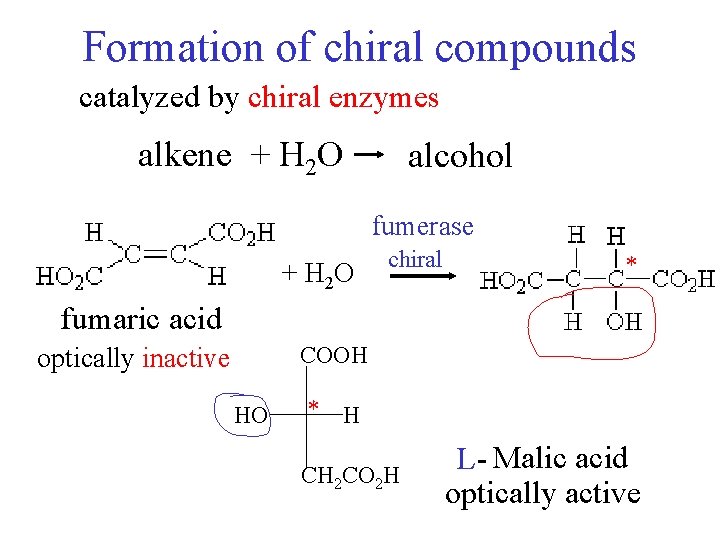
Formation of chiral compounds catalyzed by chiral enzymes alkene + H 2 O alcohol fumerase + H 2 O chiral * fumaric acid COOH optically inactive HO * H CH 2 CO 2 H L- Malic acid optically active
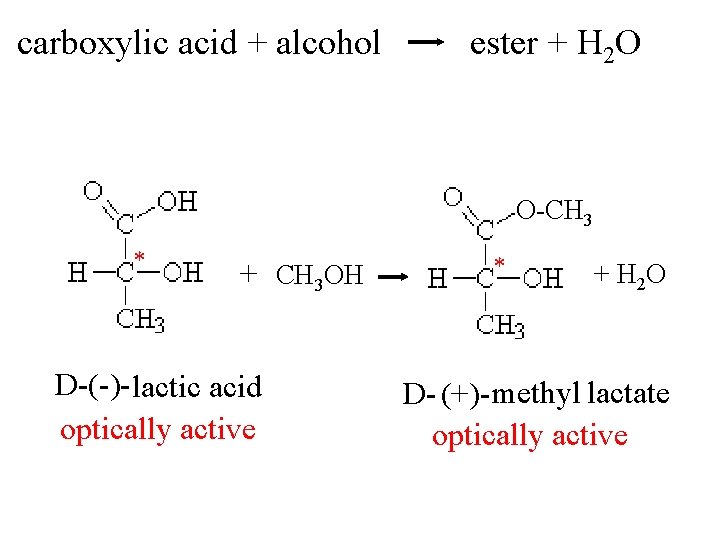
carboxylic acid + alcohol ester + H 2 O O-CH 3 * + CH 3 OH D-(-)- lactic acid optically active * + H 2 O D- (+)- methyl lactate optically active
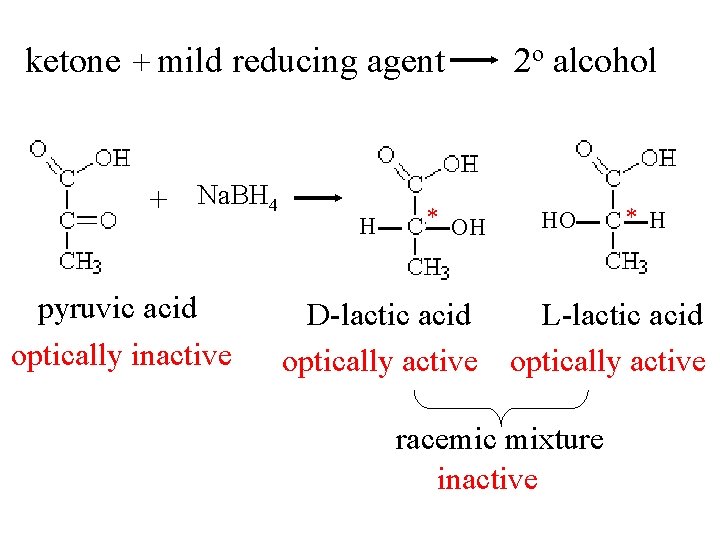
ketone + mild reducing agent + Na. BH 4 pyruvic acid optically inactive H * OH 2 o alcohol HO * H D-lactic acid L-lactic acid optically active racemic mixture inactive
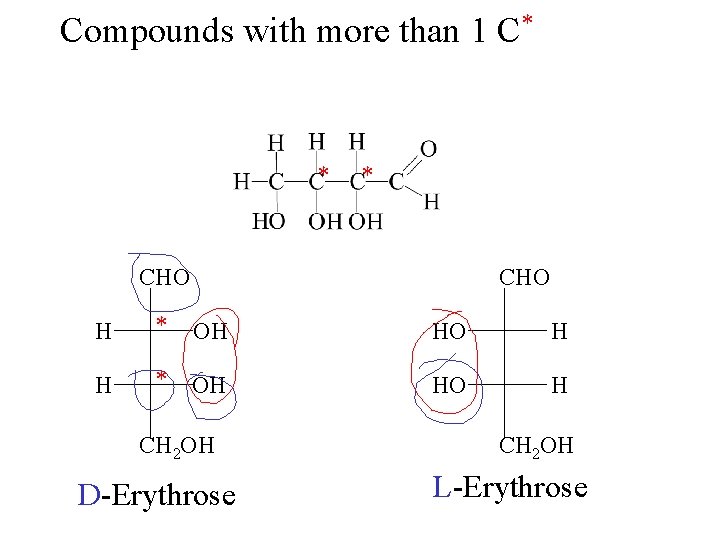
Compounds with more than 1 C* * * CHO H * OH HO H CH 2 OH D-Erythrose CH 2 OH L-Erythrose
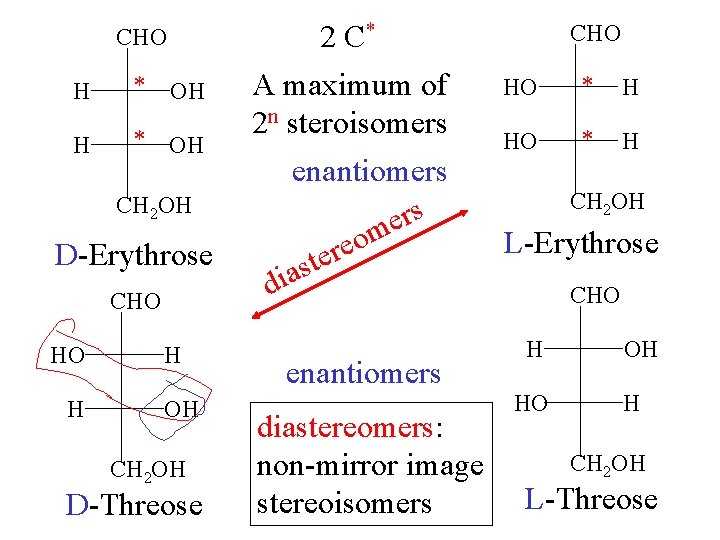
CHO H * OH CH 2 OH D-Erythrose CHO HO H H OH CH 2 OH D-Threose 2 C* A maximum of 2 n steroisomers enantiomers rs e m o e r e t s a di enantiomers diastereomers: non-mirror image stereoisomers CHO HO * H CH 2 OH L-Erythrose CHO H HO OH H CH 2 OH L-Threose
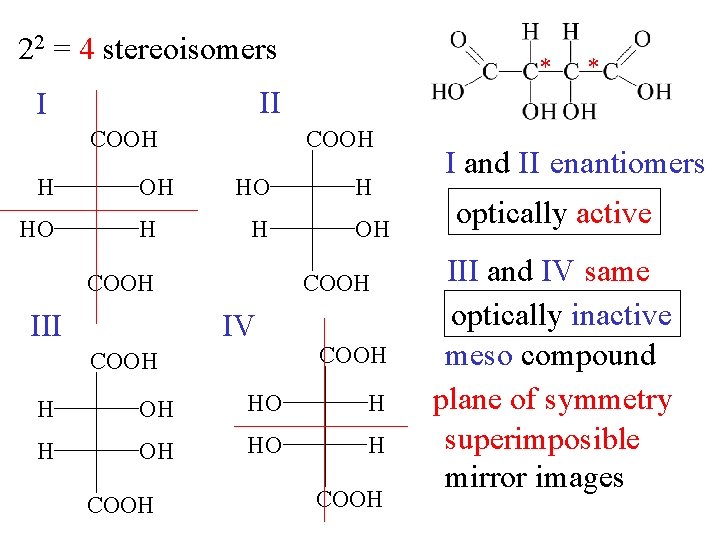
22 = 4 stereoisomers COOH HO * II I H * OH H COOH HO H OH COOH III H IV COOH HO H COOH I and II enantiomers optically active III and IV same optically inactive meso compound plane of symmetry superimposible mirror images
- Slides: 15