Chiesis commitment to limbal stem cell deficiency LSCD

Chiesi’s commitment to limbal stem cell deficiency (LSCD) and future directions

Chiesi: our values RELIABLE COMPANY FOCUS ON PEOPLE · Ethics · Transparent · Listening · Development FOCUS ON CUSTOMERS TEAM SPIRIT · Excellence · Devotion · Cohesion · Collaboration VALUE GENERATION · Innovation · Entrepreneurship 2 EVERY ONE OF US IS CHIESI · Sense of belonging · Responsibility
Our vision Focus High level research and development investment Respiratory Knowhow and expertise in product development in the respiratory area is key for growth Neonatology Chiesi aims to offer concrete and effective solutions for perinatal diseases and prematurity syndrome Rare diseases Unmet medical needs and social impact of rare diseases are at the core of our work in this area LSCD is among the areas of interest 3
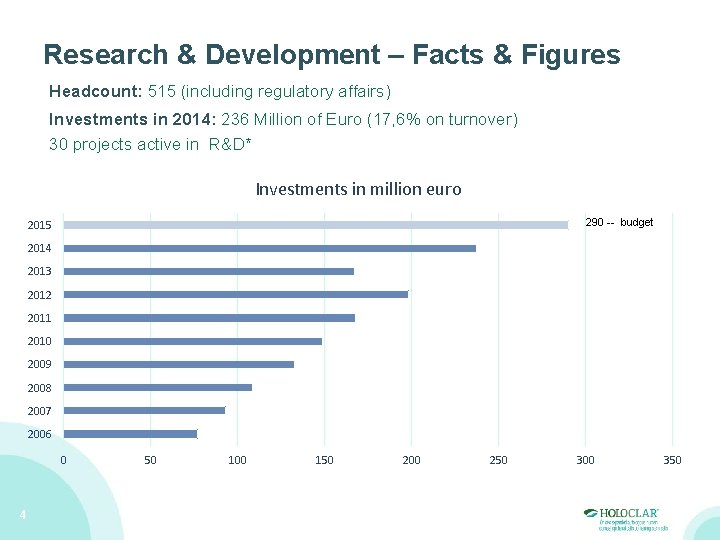
Research & Development – Facts & Figures Headcount: 515 (including regulatory affairs) Investments in 2014: 236 Million of Euro (17, 6% on turnover) 30 projects active in R&D* Investments in million euro 290 -- budget 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 0 4 50 100 150 200 250 300 350
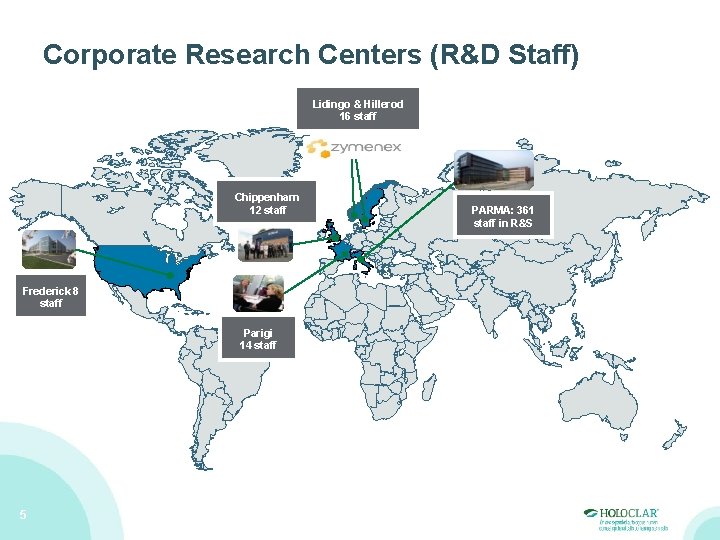
Corporate Research Centers (R&D Staff) Lidingo & Hillerod 16 staff Chippenham 12 staff Frederick 8 staff Parigi 14 staff 5 PARMA: 361 staff in R&S

Chiesi–Holostem collaboration 1 World class technology 2 6 The University’s Centre for Regenerative Medicine 3 New European regulation: Pharma rules, incl. GMP, apply 4 - Regulatory and industrial knowhow - Commercial infrastructure in Europe GMP, good manufacturing practices 1394/2007 CE REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on advanced therapy medicinal products and amending Directive 2001/83/EC and Regulation (EC) No 726/2004

The first and only EMA-approved stem cell-based medicinal product 7
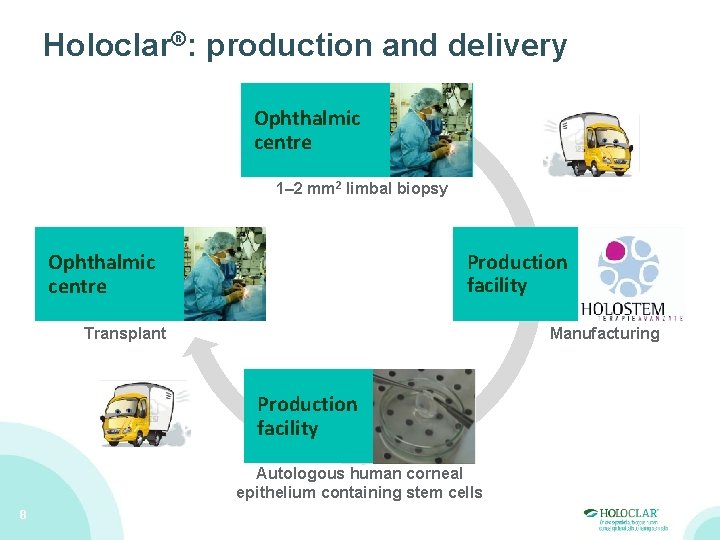
Holoclar®: production and delivery Ophthalmic centre 1– 2 mm 2 limbal biopsy Production facility Ophthalmic centre Transplant Manufacturing Production facility Autologous human corneal epithelium containing stem cells 8
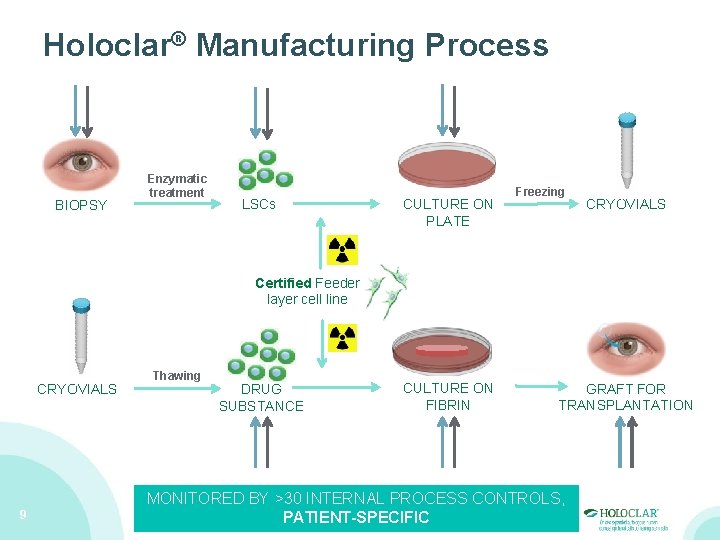
Holoclar® Manufacturing Process BIOPSY Enzymatic treatment LSCs CULTURE ON PLATE Freezing CRYOVIALS Certified Feeder layer cell line CRYOVIALS 9 Thawing DRUG SUBSTANCE CULTURE ON FIBRIN GRAFT FOR TRANSPLANTATION MONITORED BY >30 INTERNAL PROCESS CONTROLS, PATIENT-SPECIFIC
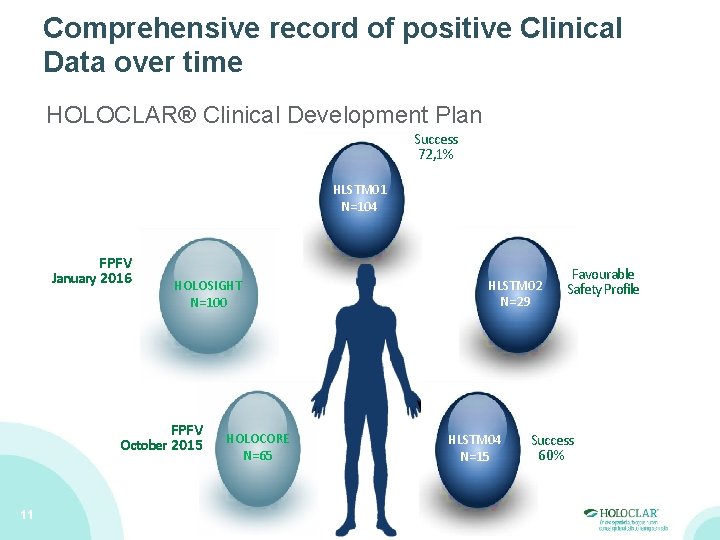
Comprehensive record of positive Clinical Data over time HOLOCLAR® Clinical Development Plan Success 72, 1% HLSTM 01 N=104 FPFV January 2016 HOLOSIGHT N=100 FPFV October 2015 11 HOLOCORE N=65 HLSTM 02 N=29 HLSTM 04 N=15 Favourable Safety Profile Success 60%
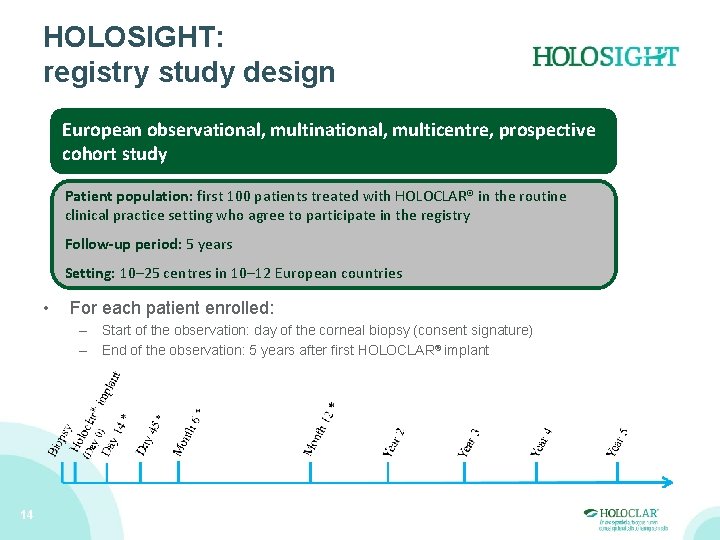
HOLOSIGHT: registry study design European observational, multinational, multicentre, prospective cohort study Patient population: first 100 patients treated with HOLOCLAR® in the routine clinical practice setting who agree to participate in the registry Follow-up period: 5 years Setting: 10– 25 centres in 10– 12 European countries • For each patient enrolled: – Start of the observation: day of the corneal biopsy (consent signature) – End of the observation: 5 years after first HOLOCLAR® implant 14

Provides Easy and Supported access to treatment • Holoclar will be delivered to patients in Europe thanks to an integrated and “service-based” logistic business model • Chiesi does not only guarantee to deliver the product but the structure will also ensure all the logistic phases of biopsy taking, manufacturing, shipment of cell tissue and implant 15

HOLOCLAR® Key Values 16 1. First EMA-approved product for LSCD due to ocular burns 2. Revolutionary Product: First stem-cell product approved in EU using Autologous Stem Cells 3. Comprehensive record of positive Clinical Data over time 4. Certified High-Quality production standard 5. Highly labour-intensive product by extremely Qualified personnel 6. Provides Easy and Supported access to treatment 7. Proven to be a Cost-Effective alternative to conservative management
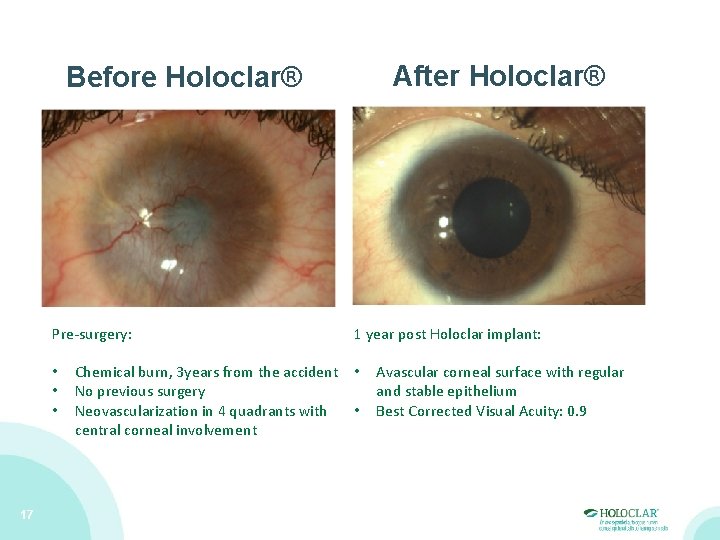
After Holoclar® Before Holoclar® Pre-surgery: • • • 17 1 year post Holoclar implant: Chemical burn, 3 years from the accident • No previous surgery • Neovascularization in 4 quadrants with central corneal involvement Avascular corneal surface with regular and stable epithelium Best Corrected Visual Acuity: 0. 9
- Slides: 14