Chicago 2008 Post ASCO Analysis Metastatic Breast Cancer
Chicago 2008: Post - ASCO Analysis: Metastatic Breast Cancer CONTENT • Take Home Message . . . 2 • Chemotherapy. . . . 3 • Navelbine. . . • Targeted therapy . . . 19 • Navelbine. . . 28 ASCO 08 MBC . 9 1

ASCO 08 Metastatic Breast : Take Home Message • No major trials presented during the meeting • Main chemotherapy options remain the same • Many interesting communications on Navelbine in MBC • More and more trial results of targeted therapies • Role of Avastin in MBC: improvement of PFS with no specific targeting of pts, is this enough? • Herceptin more than ever the standard for HER 2+ patients, even after relapse ASCO 08 MBC 2

ASCO 2008: Chemotherapy ASCO 08 MBC 3

Chemotherapy duration in MBC Meta-analysis (Gennari, #1067 p) • Meta-analysis including 8 randomized studies evaluating longer vs shorter CT duration (1942 pts) • Overall, in terms of PFS, longer CT duration was associated with a 35% reduction in the risk of relapse (p=s) • Longer CT duration was associated with an 8% reduction in the risk of death (p=ns) Confirmed benefits of a longer chemotherapy duration ASCO 08 MBC 4
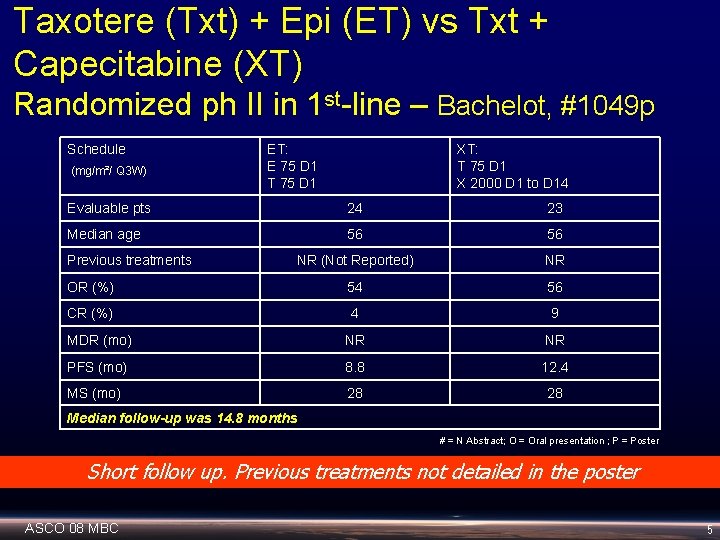
Taxotere (Txt) + Epi (ET) vs Txt + Capecitabine (XT) Randomized ph II in 1 st-line – Bachelot, #1049 p Schedule (mg/m²/ Q 3 W) ET: E 75 D 1 T 75 D 1 XT: T 75 D 1 X 2000 D 1 to D 14 Evaluable pts 24 23 Median age 56 56 NR (Not Reported) NR OR (%) 54 56 CR (%) 4 9 MDR (mo) NR NR PFS (mo) 8. 8 12. 4 MS (mo) 28 28 Previous treatments Median follow-up was 14. 8 months # = N Abstract; O = Oral presentation ; P = Poster Short follow up. Previous treatments not detailed in the poster ASCO 08 MBC 5
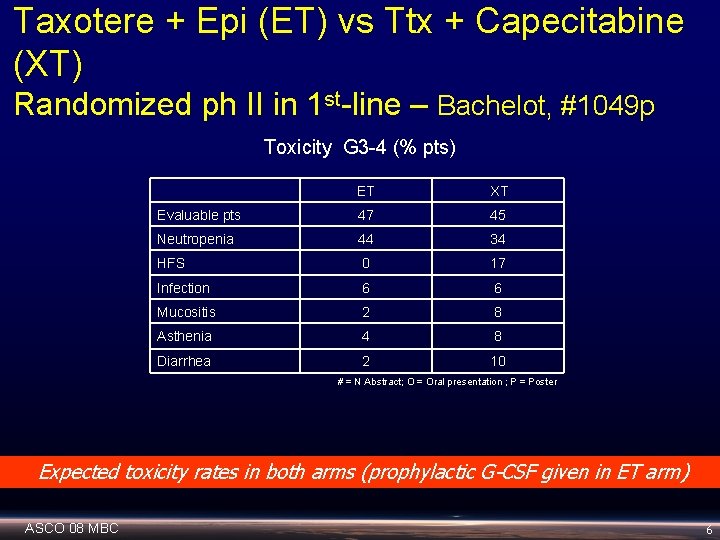
Taxotere + Epi (ET) vs Ttx + Capecitabine (XT) Randomized ph II in 1 st-line – Bachelot, #1049 p Toxicity G 3 -4 (% pts) ET XT Evaluable pts 47 45 Neutropenia 44 34 HFS 0 17 Infection 6 6 Mucositis 2 8 Asthenia 4 8 Diarrhea 2 10 # = N Abstract; O = Oral presentation ; P = Poster Expected toxicity rates in both arms (prophylactic G-CSF given in ET arm) ASCO 08 MBC 6
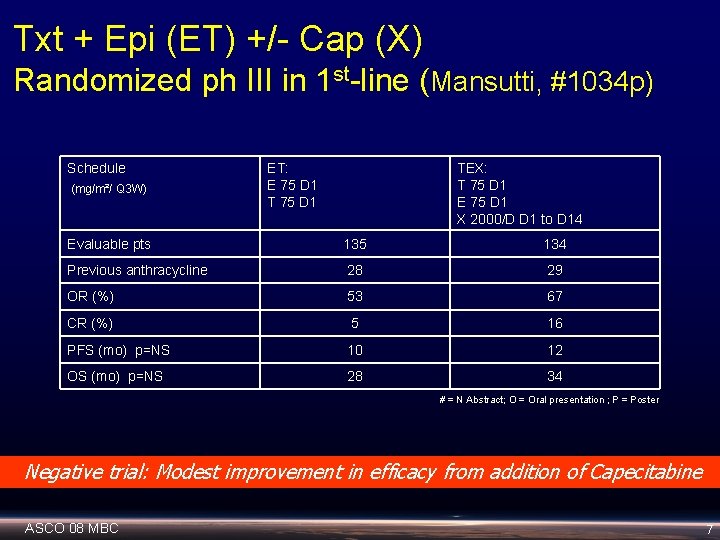
Txt + Epi (ET) +/- Cap (X) Randomized ph III in 1 st-line (Mansutti, #1034 p) Schedule (mg/m²/ Q 3 W) ET: E 75 D 1 TEX: T 75 D 1 E 75 D 1 X 2000/D D 1 to D 14 Evaluable pts 135 134 Previous anthracycline 28 29 OR (%) 53 67 CR (%) 5 16 PFS (mo) p=NS 10 12 OS (mo) p=NS 28 34 # = N Abstract; O = Oral presentation ; P = Poster Negative trial: Modest improvement in efficacy from addition of Capecitabine ASCO 08 MBC 7
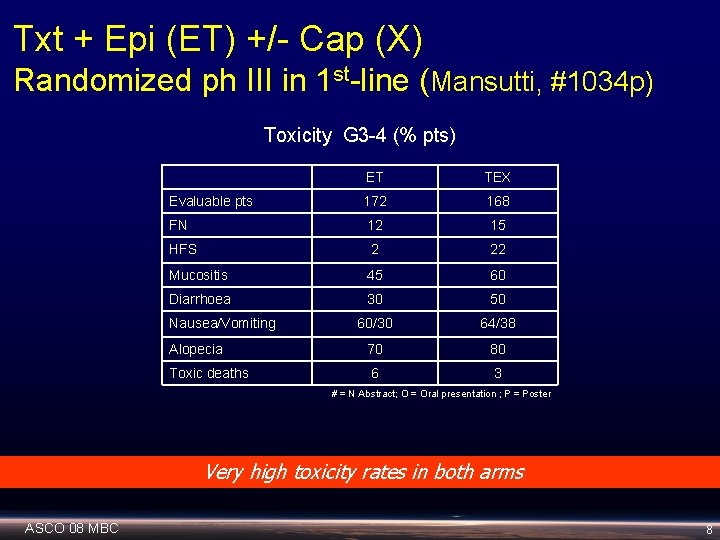
Txt + Epi (ET) +/- Cap (X) Randomized ph III in 1 st-line (Mansutti, #1034 p) Toxicity G 3 -4 (% pts) ET TEX Evaluable pts 172 168 FN 12 15 HFS 2 22 Mucositis 45 60 Diarrhoea 30 50 60/30 64/38 Alopecia 70 80 Toxic deaths 6 3 Nausea/Vomiting # = N Abstract; O = Oral presentation ; P = Poster Very high toxicity rates in both arms ASCO 08 MBC 8

ASCO 2008: Navelbine and Chemotherapy • • 21 abstracts overall 15 accepted for presentation during the meeting 14 in combination with CT 7 in combination with a targeted therapy ASCO 08 MBC 9

Navelbine in ABC at ASCO 2008 : Chemotherapy Combinations • With Capecitabine – 7 Trials • With Platinum salts – 3 Trials • With Gemcitabine – 2 Trials • With Anthracyclines – 1 Trial • With 5 FU – 1 Trial ASCO 08 MBC 10
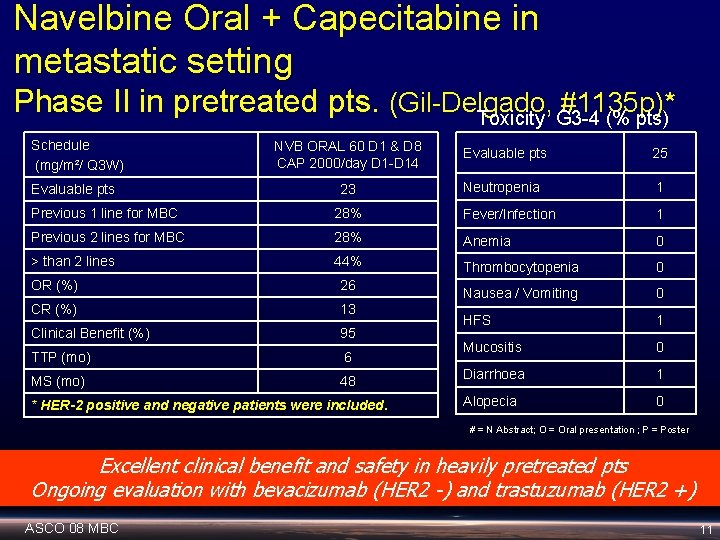
Navelbine Oral + Capecitabine in metastatic setting Phase II in pretreated pts. (Gil-Delgado, #1135 p)* Toxicity G 3 -4 (% pts) Schedule (mg/m²/ Q 3 W) Evaluable pts NVB ORAL 60 D 1 & D 8 CAP 2000/day D 1 -D 14 Evaluable pts 25 23 Neutropenia 1 Previous 1 line for MBC 28% Fever/Infection 1 Previous 2 lines for MBC 28% Anemia 0 > than 2 lines 44% Thrombocytopenia 0 Nausea / Vomiting 0 HFS 1 Mucositis 0 Diarrhoea 1 Alopecia 0 OR (%) 26 CR (%) 13 Clinical Benefit (%) 95 TTP (mo) 6 MS (mo) 48 * HER-2 positive and negative patients were included. # = N Abstract; O = Oral presentation ; P = Poster Excellent clinical benefit and safety in heavily pretreated pts Ongoing evaluation with bevacizumab (HER 2 -) and trastuzumab (HER 2 +) ASCO 08 MBC 11
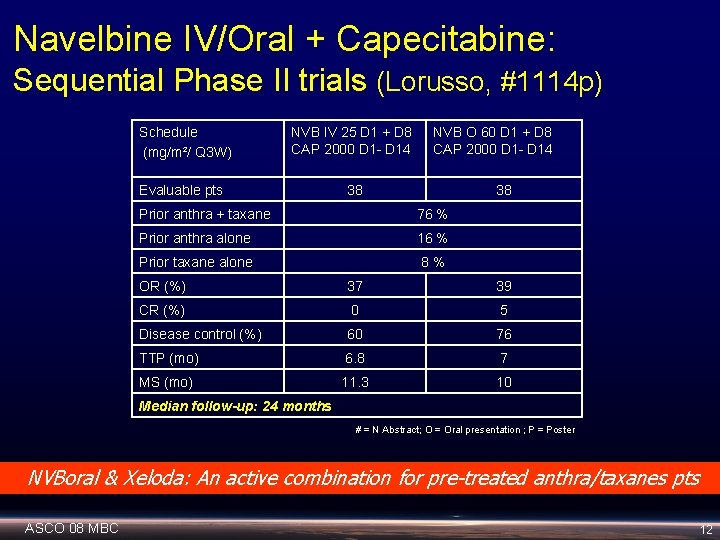
Navelbine IV/Oral + Capecitabine: Sequential Phase II trials (Lorusso, #1114 p) Schedule (mg/m²/ Q 3 W) NVB IV 25 D 1 + D 8 CAP 2000 D 1 - D 14 Evaluable pts NVB O 60 D 1 + D 8 CAP 2000 D 1 - D 14 38 38 Prior anthra + taxane 76 % Prior anthra alone 16 % Prior taxane alone 8% OR (%) 37 39 CR (%) 0 5 Disease control (%) 60 76 TTP (mo) 6. 8 7 MS (mo) 11. 3 10 Median follow-up: 24 months # = N Abstract; O = Oral presentation ; P = Poster NVBoral & Xeloda: An active combination for pre-treated anthra/taxanes pts ASCO 08 MBC 12

Navelbine IV/Oral + Capecitabine: Sequential Phase II trials (Lorusso, #1114 p) Toxicity G 3 -4 (% pts) NVB IV + CAP NVB ORAL + CAP Evaluable pts 38 38 Neutropenia 10 21 Anemia (grade 2 -3) 0 13 Thrombocytopenia 0 3 HFS (grade 2 -3) 5 5 Diarrhea (grade 2) 8 8 # = N Abstract; O = Oral presentation ; P = Poster Well-tolerated regimen ASCO 08 MBC 13

Navelbine Oral + Capecitabine: Feasibility study for dose optimisation (Anton, #1105 p) Schedule (mg/m²/ Q 3 W) Number of pts Prior CT (% pts) OR (%) Recommended dose (level IV) NVB ORAL D 1 & D 8, different dose levels CAP D 1 -D 14, different dose levels 18 in adjuvant setting: 55% In metastatic setting: 22% Both: 5% 33% for dose levels III and IV NVB ORAL 80 D 1 + D 8 CAP 2000/day D 1 to D 14, both q 3 w No further dose escalation performed in this study Both agents safely used at recommended single agent dose # = N Abstract; O = Oral presentation ; P = Poster Feasible combination with a dose of NVBO of 80 mg/m 2 D 1 and D 8 q 3 w ASCO 08 MBC 14
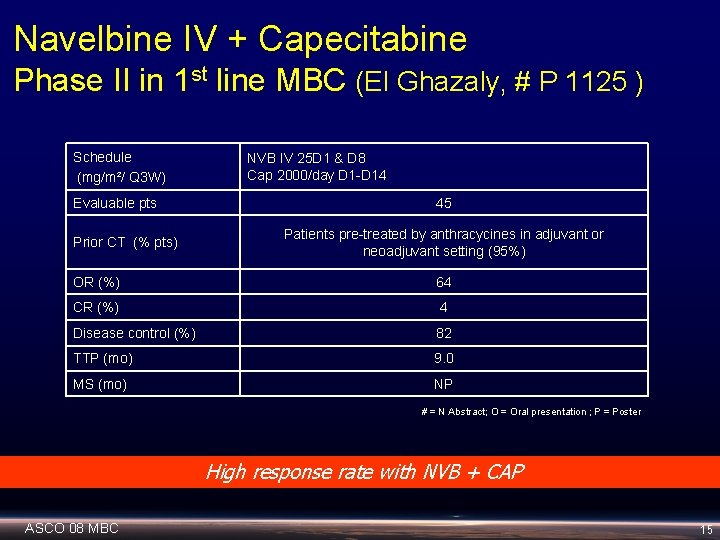
Navelbine IV + Capecitabine Phase II in 1 st line MBC (El Ghazaly, # P 1125 ) Schedule (mg/m²/ Q 3 W) Evaluable pts Prior CT (% pts) NVB IV 25 D 1 & D 8 Cap 2000/day D 1 -D 14 45 Patients pre-treated by anthracycines in adjuvant or neoadjuvant setting (95%) OR (%) 64 CR (%) 4 Disease control (%) 82 TTP (mo) 9. 0 MS (mo) NP # = N Abstract; O = Oral presentation ; P = Poster High response rate with NVB + CAP ASCO 08 MBC 15

Navelbine IV + Capecitabine Phase II in 1 st line MBC (El Ghazaly, # P 1125 ) Toxicity G 3 -4 (% pts) Evaluable pts 45 Neutropenia 13 Febrile Neutropenia 2 Anemia 4 Thrombocytopenia 4 Nausae/Vomiting 16 HFS 7 Mucositis 11 Diarrhoea 9 Local Phlebitis 7 # = N Abstract; O = Oral presentation ; P = Poster 7% of peripheral phlebitis could be avoided with the ALL ORAL combination ASCO 08 MBC 16
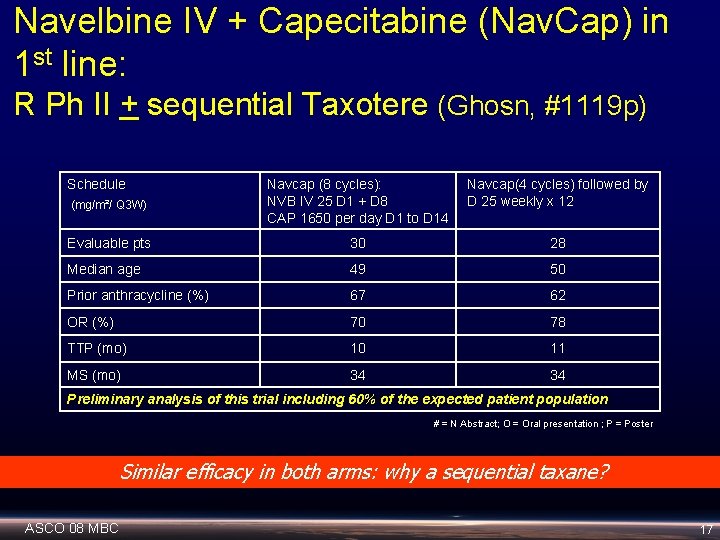
Navelbine IV + Capecitabine (Nav. Cap) in 1 st line: R Ph II + sequential Taxotere (Ghosn, #1119 p) Schedule (mg/m²/ Q 3 W) Navcap (8 cycles): NVB IV 25 D 1 + D 8 CAP 1650 per day D 1 to D 14 Navcap(4 cycles) followed by D 25 weekly x 12 Evaluable pts 30 28 Median age 49 50 Prior anthracycline (%) 67 62 OR (%) 70 78 TTP (mo) 10 11 MS (mo) 34 34 Preliminary analysis of this trial including 60% of the expected patient population # = N Abstract; O = Oral presentation ; P = Poster Similar efficacy in both arms: why a sequential taxane? ASCO 08 MBC 17
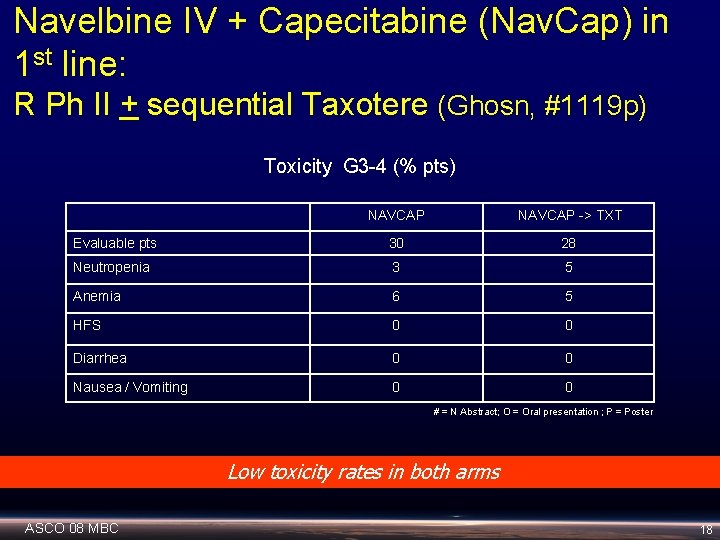
Navelbine IV + Capecitabine (Nav. Cap) in 1 st line: R Ph II + sequential Taxotere (Ghosn, #1119 p) Toxicity G 3 -4 (% pts) NAVCAP -> TXT Evaluable pts 30 28 Neutropenia 3 5 Anemia 6 5 HFS 0 0 Diarrhea 0 0 Nausea / Vomiting 0 0 # = N Abstract; O = Oral presentation ; P = Poster Low toxicity rates in both arms ASCO 08 MBC 18

ASCO 2008: Targeted Therapy ASCO 08 MBC 19
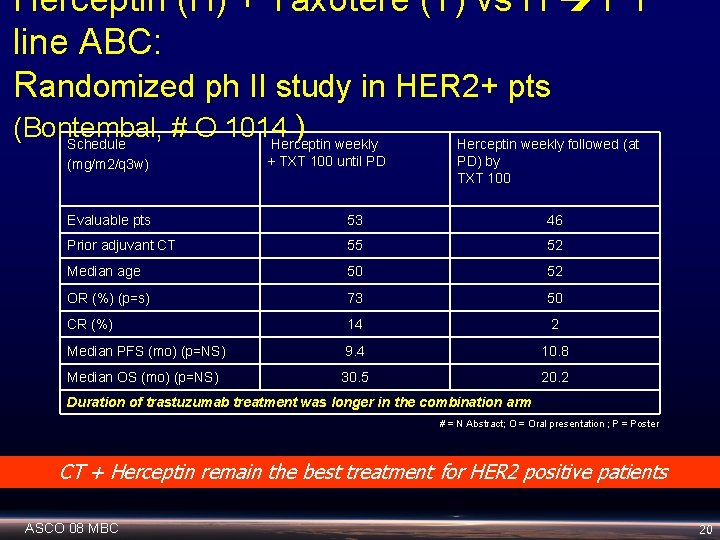
Herceptin (H) + Taxotere (T) vs H T 1 st line ABC: Randomized ph II study in HER 2+ pts (Bontembal, # O 1014 ) Schedule Herceptin weekly (mg/m 2/q 3 w) + TXT 100 until PD Herceptin weekly followed (at PD) by TXT 100 Evaluable pts 53 46 Prior adjuvant CT 55 52 Median age 50 52 OR (%) (p=s) 73 50 CR (%) 14 2 Median PFS (mo) (p=NS) 9. 4 10. 8 Median OS (mo) (p=NS) 30. 5 20. 2 Duration of trastuzumab treatment was longer in the combination arm # = N Abstract; O = Oral presentation ; P = Poster CT + Herceptin remain the best treatment for HER 2 positive patients ASCO 08 MBC 20
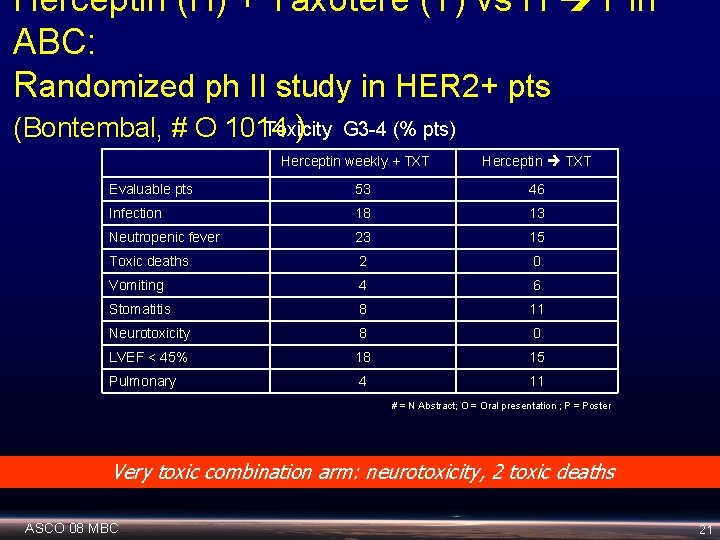
Herceptin (H) + Taxotere (T) vs H T in ABC: Randomized ph II study in HER 2+ pts Toxicity (Bontembal, # O 1014 ) G 3 -4 (% pts) Herceptin weekly + TXT Herceptin TXT Evaluable pts 53 46 Infection 18 13 Neutropenic fever 23 15 Toxic deaths 2 0 Vomiting 4 6 Stomatitis 8 11 Neurotoxicity 8 0 LVEF < 45% 18 15 Pulmonary 4 11 # = N Abstract; O = Oral presentation ; P = Poster Very toxic combination arm: neurotoxicity, 2 toxic deaths ASCO 08 MBC 21

Capecitabine (CAP) vs CAP + Herceptin (H) in ABC: Phase III in HER 2+ pts in PD with H (Von Minckwitz, 1025 p) Schedule (mg/m²/ Q 3 W) CAP 2500 per day, D 1 to D 14 + Herceptin q 3 w Evaluable pts 78 78 Median age 59 52 OR (%) 27 48 CR (%) 3 8 Clinical Benefit (%) 54 75 TTP (mo) p = 0. 034 5. 6 8. 2 MS (mo) p = NS 20. 4 25. 5 Trial stopped with 65% of the planned number of pts because of poor accrual # = N Abstract; O = Oral presentation ; P = Poster Continuation of treatment with Herceptin beyond progression is an active option ASCO 08 MBC 22
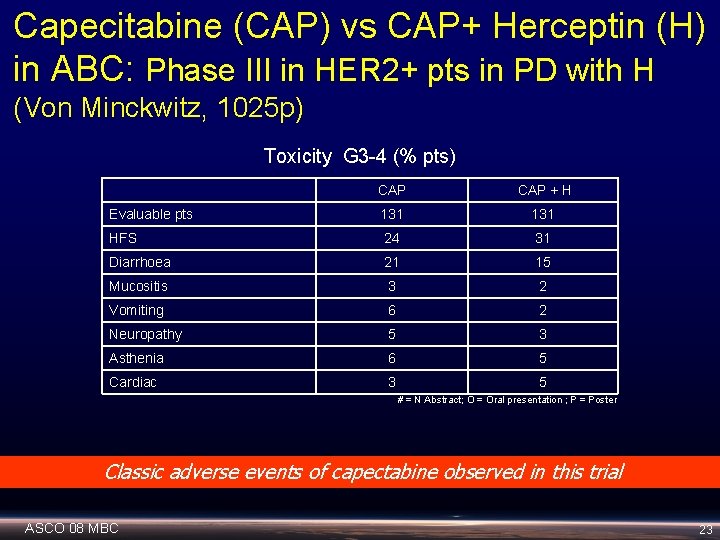
Capecitabine (CAP) vs CAP+ Herceptin (H) in ABC: Phase III in HER 2+ pts in PD with H (Von Minckwitz, 1025 p) Toxicity G 3 -4 (% pts) CAP + H Evaluable pts 131 HFS 24 31 Diarrhoea 21 15 Mucositis 3 2 Vomiting 6 2 Neuropathy 5 3 Asthenia 6 5 Cardiac 3 5 # = N Abstract; O = Oral presentation ; P = Poster Classic adverse events of capectabine observed in this trial ASCO 08 MBC 23
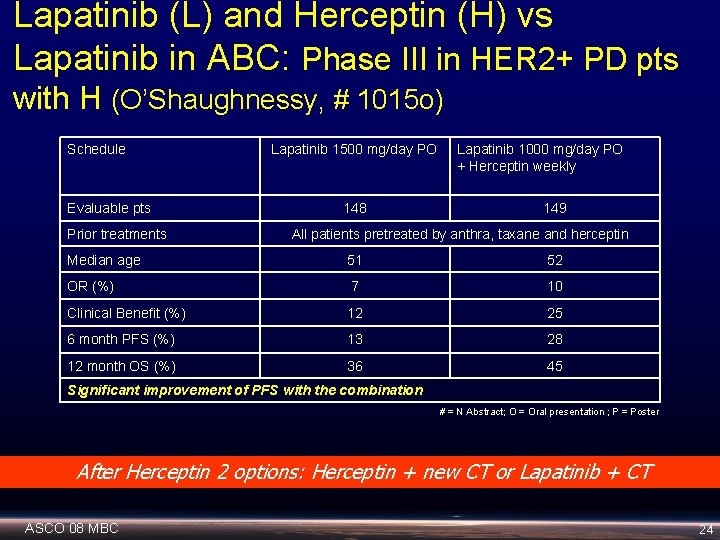
Lapatinib (L) and Herceptin (H) vs Lapatinib in ABC: Phase III in HER 2+ PD pts with H (O’Shaughnessy, # 1015 o) Schedule Evaluable pts Prior treatments Lapatinib 1500 mg/day PO 148 Lapatinib 1000 mg/day PO + Herceptin weekly 149 All patients pretreated by anthra, taxane and herceptin Median age 51 52 OR (%) 7 10 Clinical Benefit (%) 12 25 6 month PFS (%) 13 28 12 month OS (%) 36 45 Significant improvement of PFS with the combination # = N Abstract; O = Oral presentation ; P = Poster After Herceptin 2 options: Herceptin + new CT or Lapatinib + CT ASCO 08 MBC 24
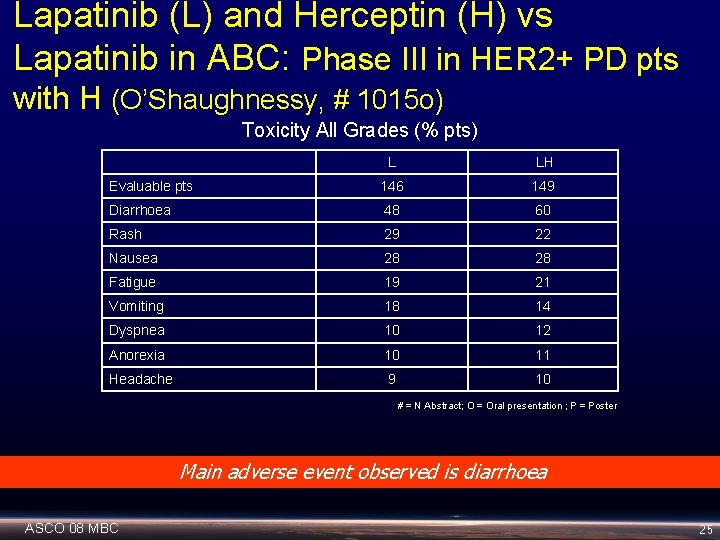
Lapatinib (L) and Herceptin (H) vs Lapatinib in ABC: Phase III in HER 2+ PD pts with H (O’Shaughnessy, # 1015 o) Toxicity All Grades (% pts) L LH Evaluable pts 146 149 Diarrhoea 48 60 Rash 29 22 Nausea 28 28 Fatigue 19 21 Vomiting 18 14 Dyspnea 10 12 Anorexia 10 11 Headache 9 10 # = N Abstract; O = Oral presentation ; P = Poster Main adverse event observed is diarrhoea ASCO 08 MBC 25
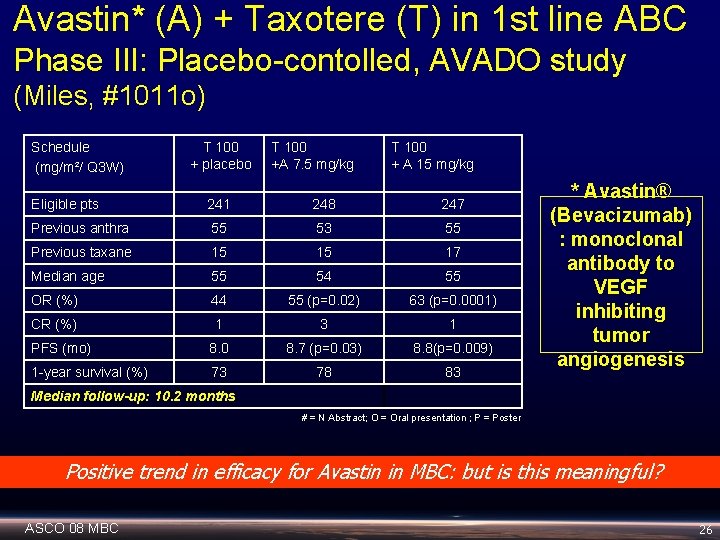
Avastin* (A) + Taxotere (T) in 1 st line ABC Phase III: Placebo-contolled, AVADO study (Miles, #1011 o) Schedule (mg/m²/ Q 3 W) T 100 + placebo T 100 +A 7. 5 mg/kg T 100 + A 15 mg/kg Eligible pts 241 248 247 Previous anthra 55 53 55 Previous taxane 15 15 17 Median age 55 54 55 OR (%) 44 55 (p=0. 02) 63 (p=0. 0001) CR (%) 1 3 1 PFS (mo) 8. 0 8. 7 (p=0. 03) 8. 8(p=0. 009) 1 -year survival (%) 73 78 83 * Avastin® (Bevacizumab) : monoclonal antibody to VEGF inhibiting tumor angiogenesis Median follow-up: 10. 2 months # = N Abstract; O = Oral presentation ; P = Poster Positive trend in efficacy for Avastin in MBC: but is this meaningful? ASCO 08 MBC 26
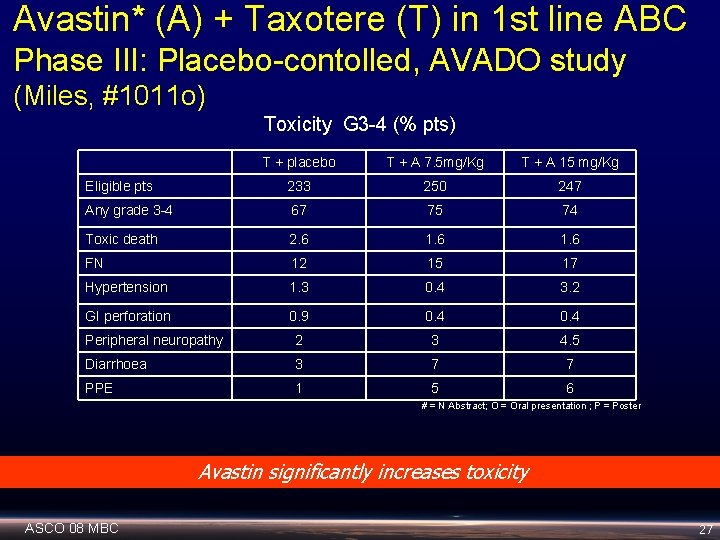
Avastin* (A) + Taxotere (T) in 1 st line ABC Phase III: Placebo-contolled, AVADO study (Miles, #1011 o) Toxicity G 3 -4 (% pts) T + placebo T + A 7. 5 mg/Kg T + A 15 mg/Kg Eligible pts 233 250 247 Any grade 3 -4 67 75 74 Toxic death 2. 6 1. 6 FN 12 15 17 Hypertension 1. 3 0. 4 3. 2 GI perforation 0. 9 0. 4 Peripheral neuropathy 2 3 4. 5 Diarrhoea 3 7 7 PPE 1 5 6 # = N Abstract; O = Oral presentation ; P = Poster Avastin significantly increases toxicity ASCO 08 MBC 27

ASCO 2008: Navelbine and Targeted Therapy • • 21 abstracts overall 15 accepted for presentation during the meeting 14 in combination with CT 7 in combination with a targeted therapy ASCO 08 MBC 28

Navelbine in ABC at ASCO 2008 : Targeted Therapy Combinations • + Trastuzumab – 4 Trials • + RAD 001 (everolimus) – 2 Trials • + PTK 787/ZK 222584 – 1 Trial ASCO 08 MBC 29
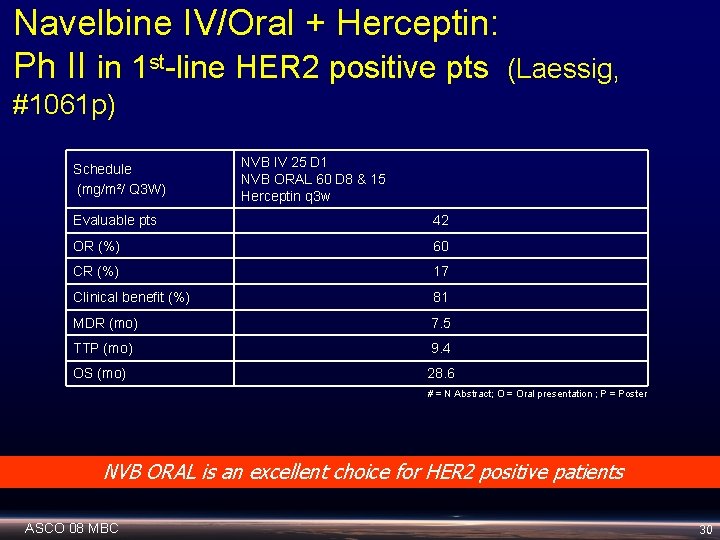
Navelbine IV/Oral + Herceptin: Ph II in 1 st-line HER 2 positive pts (Laessig, #1061 p) Schedule (mg/m²/ Q 3 W) NVB IV 25 D 1 NVB ORAL 60 D 8 & 15 Herceptin q 3 w Evaluable pts 42 OR (%) 60 CR (%) 17 Clinical benefit (%) 81 MDR (mo) 7. 5 TTP (mo) 9. 4 OS (mo) 28. 6 # = N Abstract; O = Oral presentation ; P = Poster NVB ORAL is an excellent choice for HER 2 positive patients ASCO 08 MBC 30

Navelbine IV/Oral + Herceptin: Ph II in 1 st-line HER 2 positive pts (Laessig, #1061 p) Toxicity G 3 -4 (% pts) Evaluable pts 42 Leucopenia 12 Febrile Neutropenia 0 Anemia 0 Thrombocytopenia 0 Nausea 2 Vomiting 0 Neurotoxicity 0 Cardiotoxicity 0 # = N Abstract; O = Oral presentation ; P = Poster Excellent tolerability with few grade III-IV events ASCO 08 MBC 31

Navelbine + Taxotere + Herceptin with GCSF: Ph II in 1 st-line HER 2 positive pts (Kash, #1033 p) Schedule (mg/m²/ Q 3 W) Evaluable pts Prior CT (% pts) TXT 60 D 1 NVB 27. 5 D 8 + D 15 Weekly Herceptin G-CSF D 2 to D 21 76 First-line CT in HER 2 positive patients OR (%) 71 PFS (mo) 19 MS (mo) 39 # = N Abstract; O = Oral presentation ; P = Poster Best survival rate published to date in this population ASCO 08 MBC 32

Navelbine + Taxotere + Herceptin with GCSF: Ph II in 1 st-line HER 2 positive pts (Kash, #1033 p) Toxicity G 3 -4 (% pts) Evaluable pts 76 Grade IV event 21 Grade III event 30 Neutropenia Predominant grade IV event Fatigue 10 Hyperglycemia 7 # = N Abstract; O = Oral presentation ; P = Poster Very aggressive regimen for HER 2 positive patients ASCO 08 MBC 33
- Slides: 33