CHEMSHEETS ELECTROCHEMISTRY www chemsheets co uk A 2

















- Slides: 46

CHEMSHEETS ELECTROCHEMISTRY © www. chemsheets. co. uk A 2 1077 2 -July-

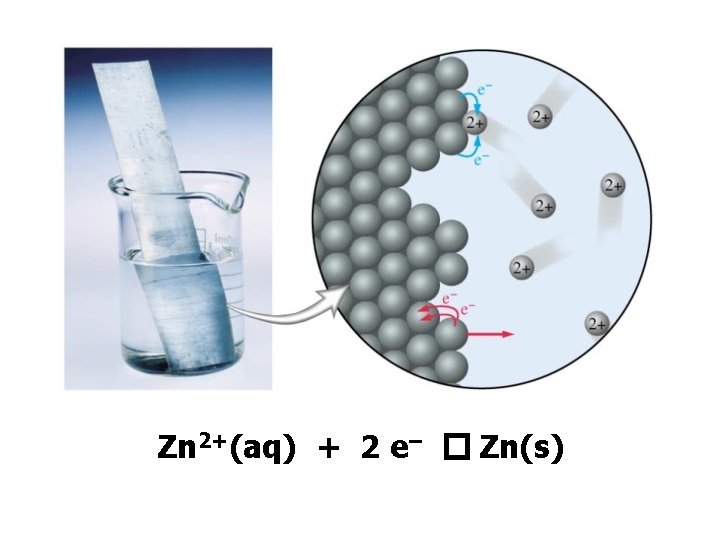
Zn 2+(aq) + 2 e– � Zn(s)

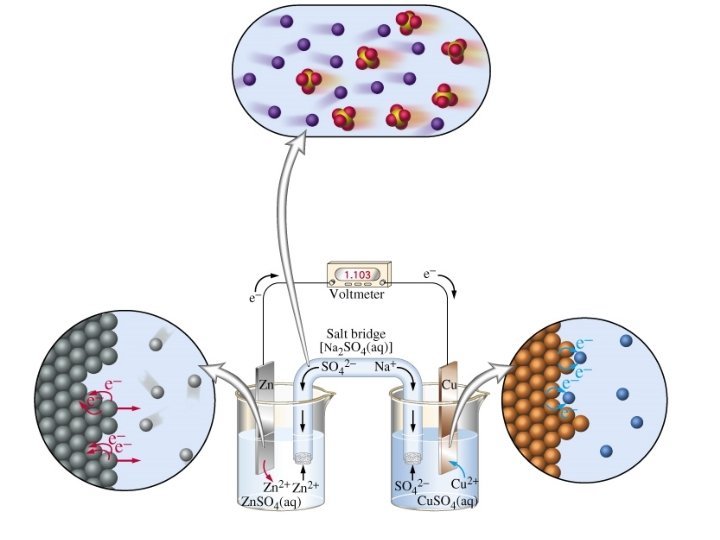
One of the two half cells [a piece of zinc in a solution of zinc nitrate(aq)] Salt bridge [strip of filter paper soaked in saturated KNO 3(aq)]




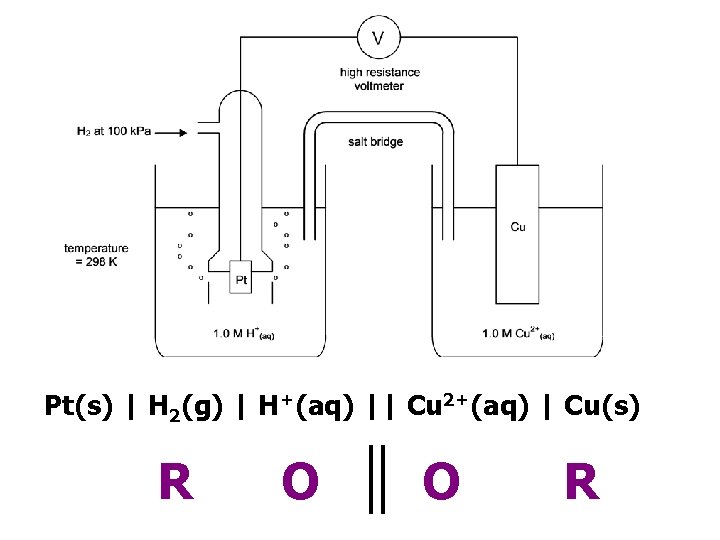
Standard Conditions Concentration 1. 0 mol dm-3 (ions involved in ½ equation) Temperature 298 K Pressure 100 k. Pa (if gases involved in ½ equation) Current Zero (use high resistance voltmeter) © www. chemsheets. co. uk A 2 1077 2 -July-

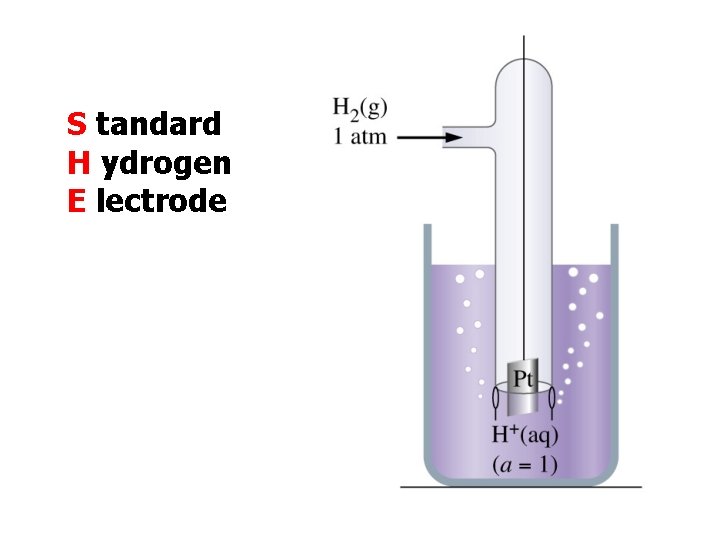
S tandard H ydrogen E lectrode

Pt(s) | H 2(g) | H+(aq) || Cu 2+(aq) | Cu(s) R O O R

R O O R Pt(s) | H 2(g) | H+(aq) || Cu 2+(aq) | Cu(s)

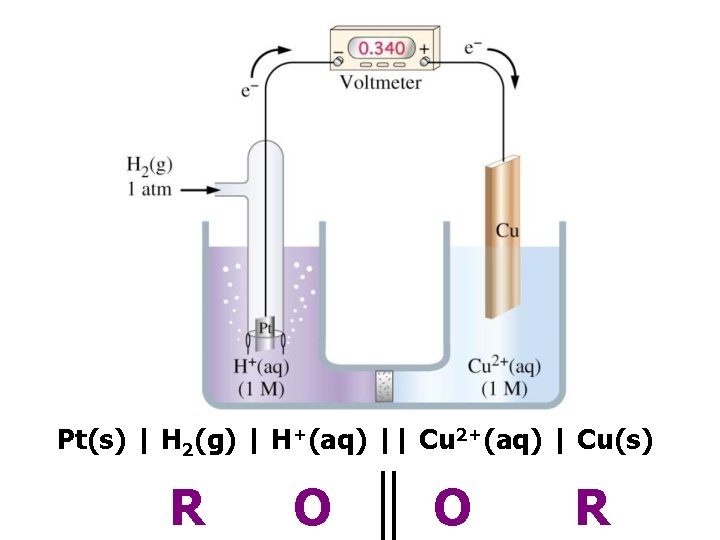
Pt(s) | H 2(g) | H+(aq) || Cu 2+(aq) | Cu(s) R O O R

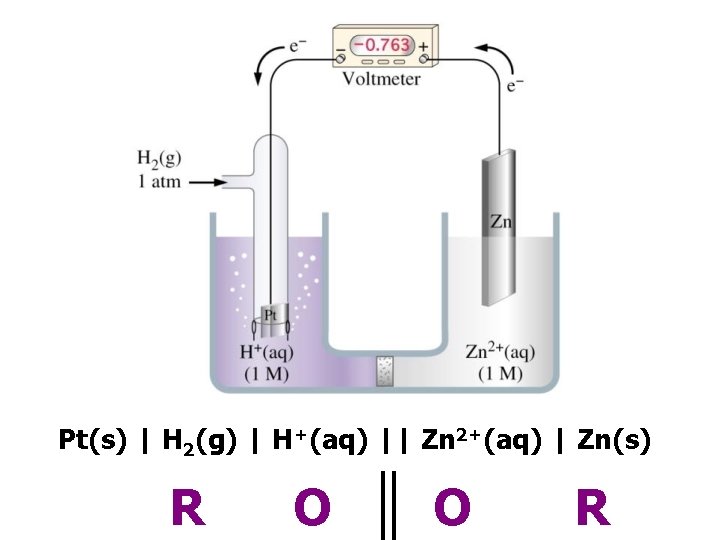
Pt(s) | H 2(g) | H+(aq) || Zn 2+(aq) | Zn(s) R O O R

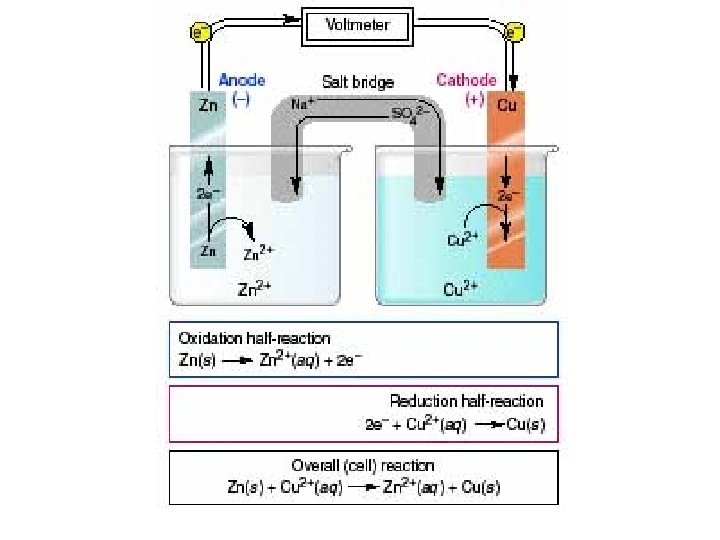
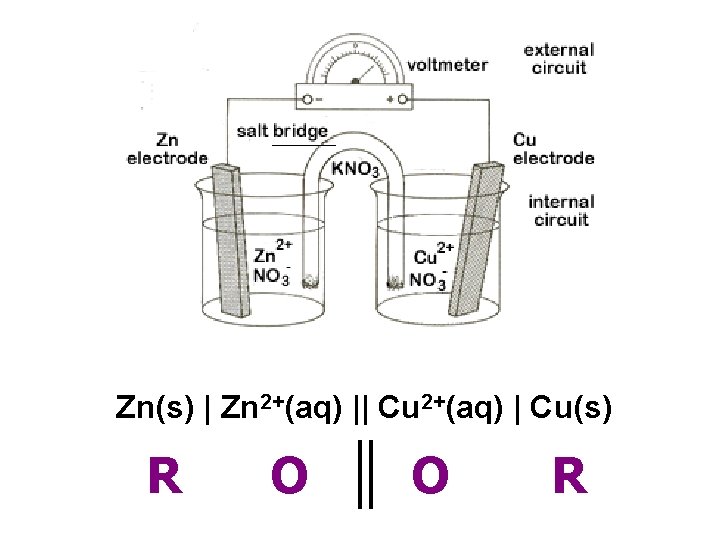
Zn(s) | Zn 2+(aq) || Cu 2+(aq) | Cu(s) R O O R

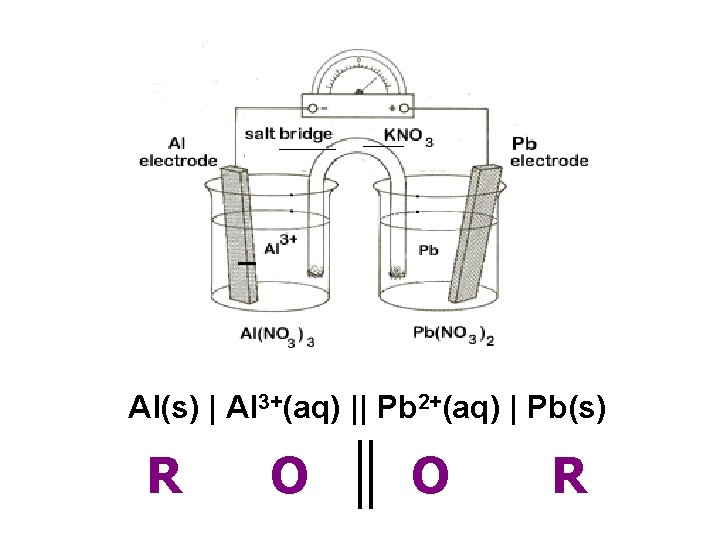
Al(s) | Al 3+(aq) || Pb 2+(aq) | Pb(s) R O O R

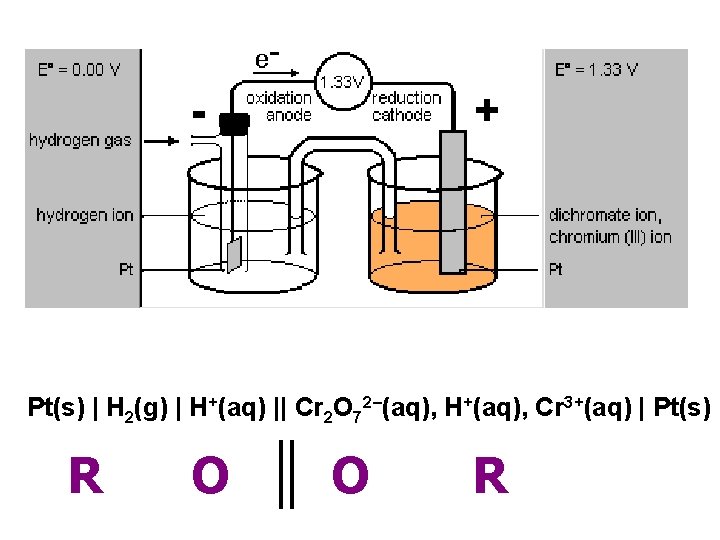
Pt(s) | H 2(g) | H+(aq) || Cr 2 O 72–(aq), H+(aq), Cr 3+(aq) | Pt(s) R O O R

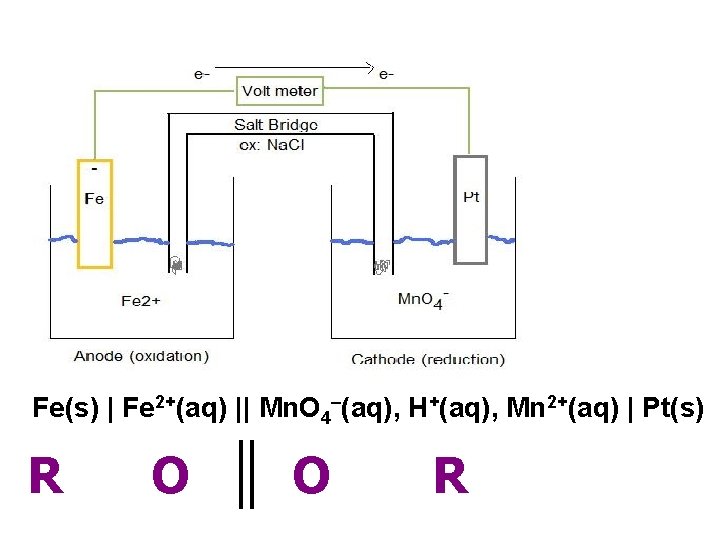
Fe(s) | Fe 2+(aq) || Mn. O 4–(aq), H+(aq), Mn 2+(aq) | Pt(s) R O O R

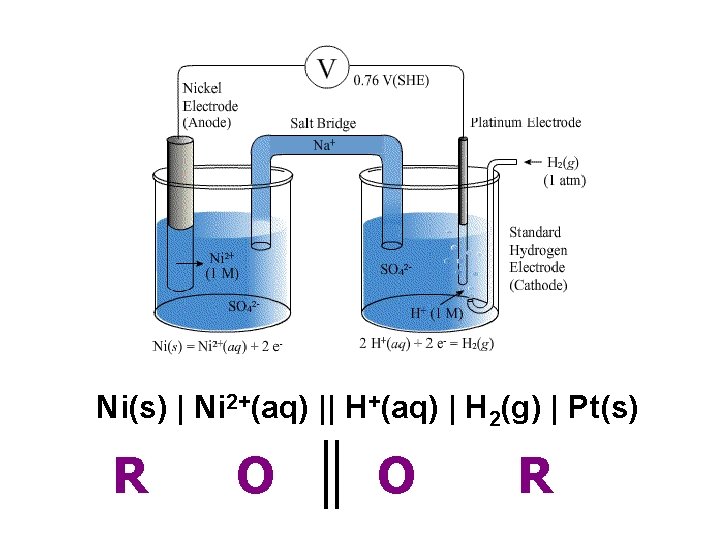
Ni(s) | Ni 2+(aq) || H+(aq) | H 2(g) | Pt(s) R O O R

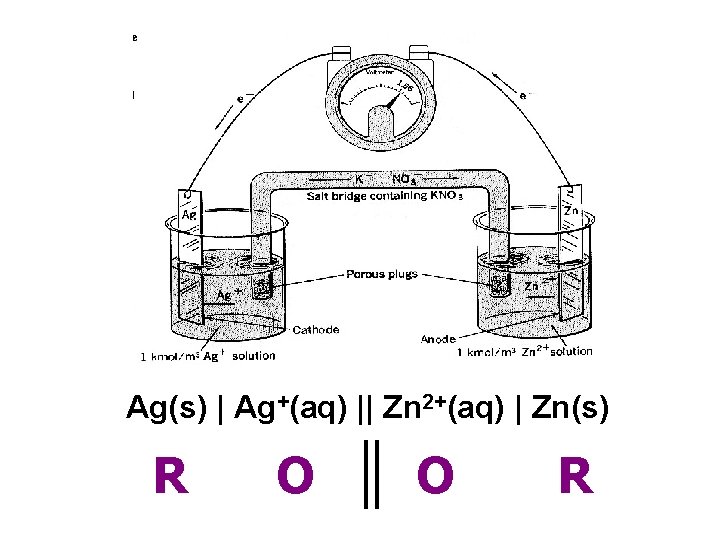
Ag(s) | Ag+(aq) || Zn 2+(aq) | Zn(s) R O O R

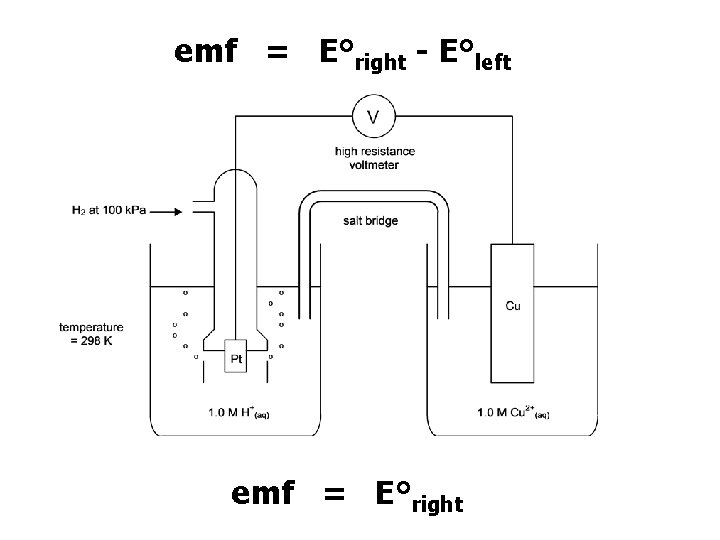
emf = E°right - E°left emf = E°right

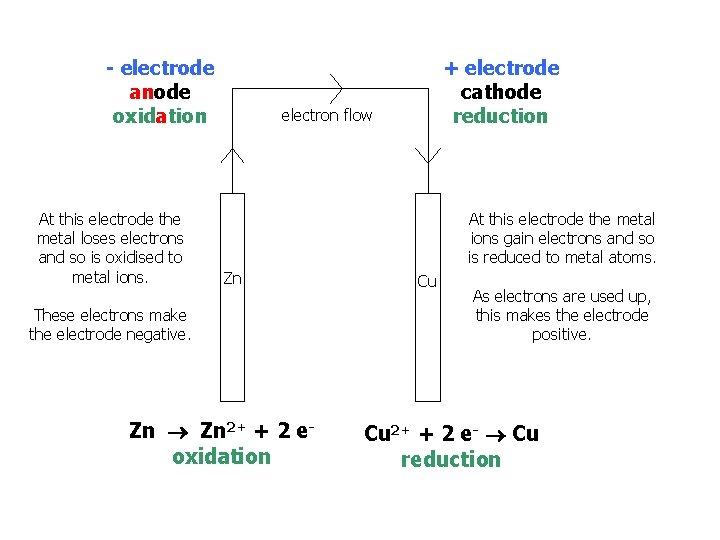
- electrode anode oxidation At this electrode the metal loses electrons and so is oxidised to metal ions. + electrode cathode reduction electron flow At this electrode the metal ions gain electrons and so is reduced to metal atoms. Zn These electrons make the electrode negative. Zn 2+ + 2 eoxidation Cu As electrons are used up, this makes the electrode positive. Cu 2+ + 2 e- Cu reduction

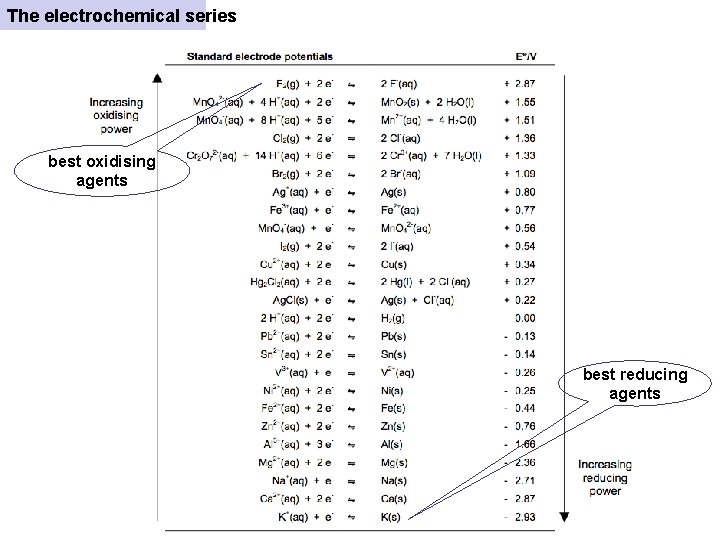
The electrochemical series best oxidising agents best reducing agents

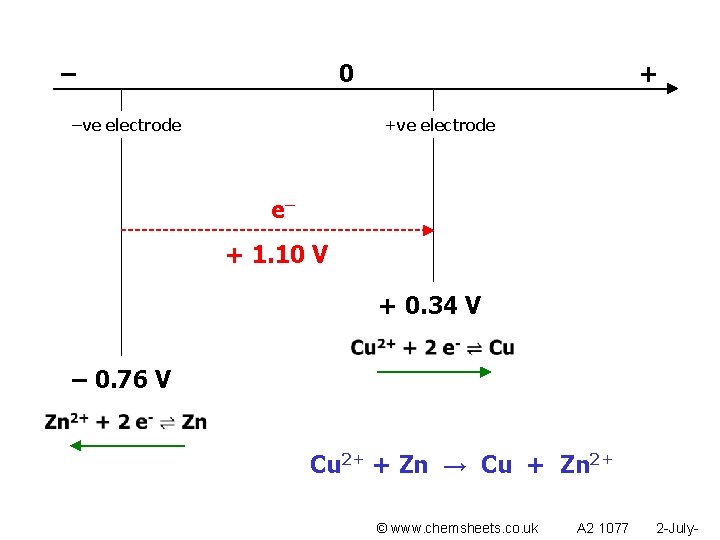
The more +ve electrode gains electrons (+ charge attracts electrons)

– 0 –ve electrode + +ve electrode e– + 1. 10 V + 0. 34 V – 0. 76 V Cu 2+ + Zn → Cu + Zn 2+ © www. chemsheets. co. uk A 2 1077 2 -July-

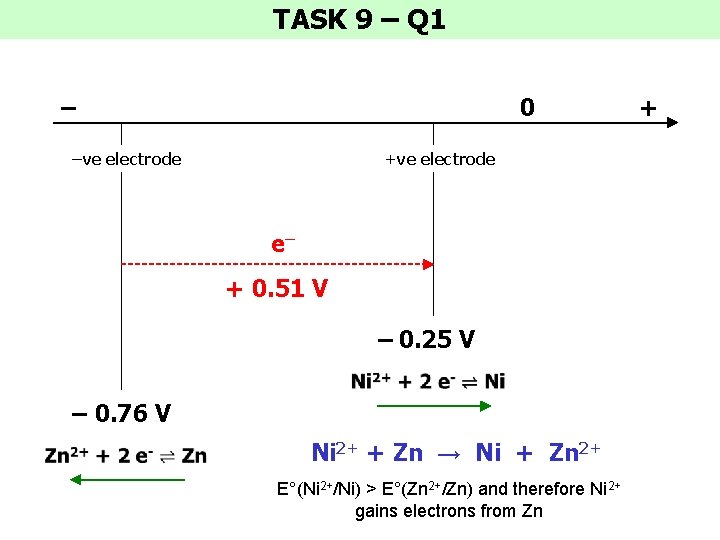
TASK 9 – Q 1 – 0 –ve electrode +ve electrode e– + 0. 51 V – 0. 25 V – 0. 76 V Ni 2+ + Zn → Ni + Zn 2+ E°(Ni 2+/Ni) > E°(Zn 2+/Zn) and therefore Ni 2+ gains electrons from Zn +

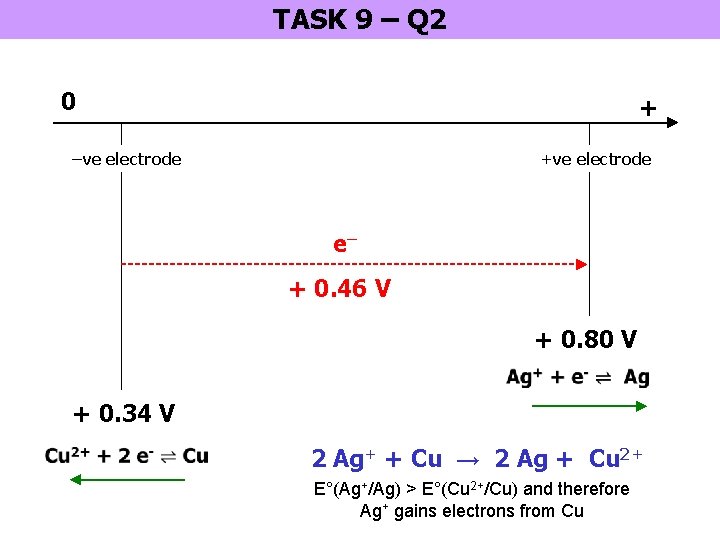
TASK 9 – Q 2 0 + –ve electrode +ve electrode e– + 0. 46 V + 0. 80 V + 0. 34 V 2 Ag+ + Cu → 2 Ag + Cu 2+ E°(Ag+/Ag) > E°(Cu 2+/Cu) and therefore Ag+ gains electrons from Cu

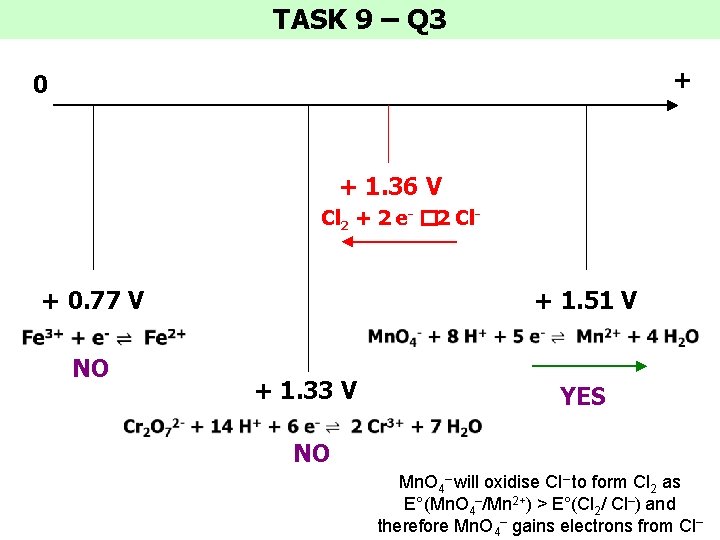
TASK 9 – Q 3 + 0 + 1. 36 V Cl 2 + 2 e- � 2 Cl- + 0. 77 V NO + 1. 51 V + 1. 33 V YES NO Mn. O 4– will oxidise Cl– to form Cl 2 as E°(Mn. O 4–/Mn 2+) > E°(Cl 2/ Cl–) and therefore Mn. O 4– gains electrons from Cl–

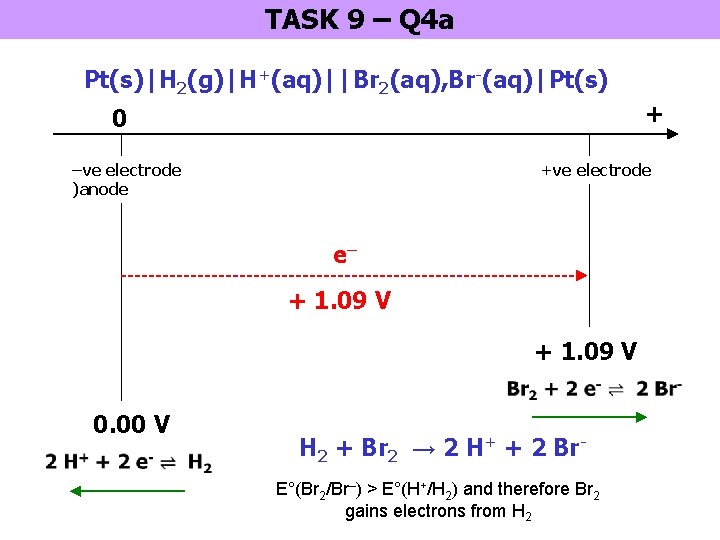
TASK 9 – Q 4 a Pt(s)|H 2(g)|H+(aq)||Br 2(aq), Br-(aq)|Pt(s) + 0 –ve electrode )anode +ve electrode e– + 1. 09 V 0. 00 V H 2 + Br 2 → 2 H+ + 2 Br. E°(Br 2/Br–) > E°(H+/H 2) and therefore Br 2 gains electrons from H 2

TASK 9 – Q 4 b Cu(s)|Cu 2+(aq)||Fe 3+(aq), Fe 2+(aq)|Pt(s) 0 –ve electrode +ve electrode e– + 0. 43 V + 0. 77 V + 0. 34 V 2 Fe 3+ + Cu → 2 Fe 2+ + Cu 2+ E°(Fe 3+/Fe 2+) > E°(Cu 2+/Cu) and therefore Fe 3+ gains electrons from Cu +

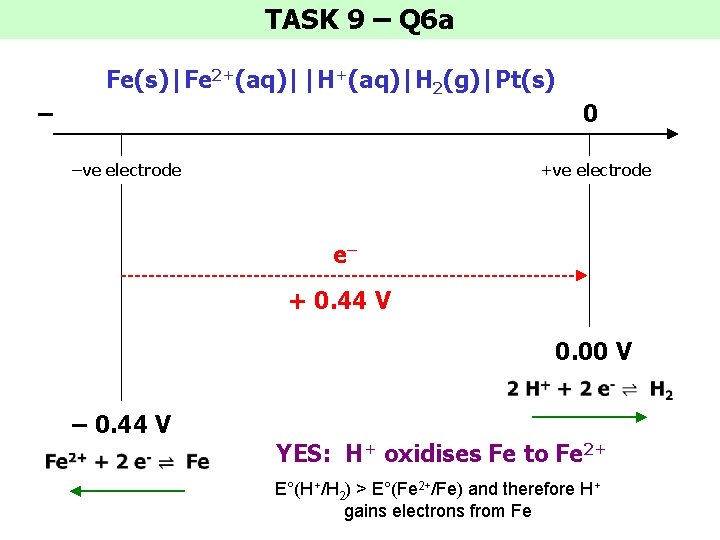
TASK 9 – Q 6 a Fe(s)|Fe 2+(aq)||H+(aq)|H 2(g)|Pt(s) – 0 –ve electrode +ve electrode e– + 0. 44 V 0. 00 V – 0. 44 V YES: H+ oxidises Fe to Fe 2+ E°(H+/H 2) > E°(Fe 2+/Fe) and therefore H+ gains electrons from Fe

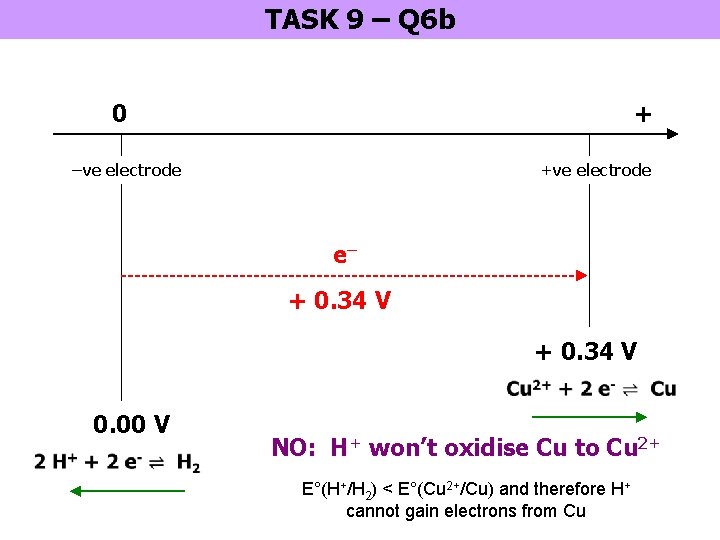
TASK 9 – Q 6 b 0 + –ve electrode +ve electrode e– + 0. 34 V 0. 00 V NO: H+ won’t oxidise Cu to Cu 2+ E°(H+/H 2) < E°(Cu 2+/Cu) and therefore H+ cannot gain electrons from Cu

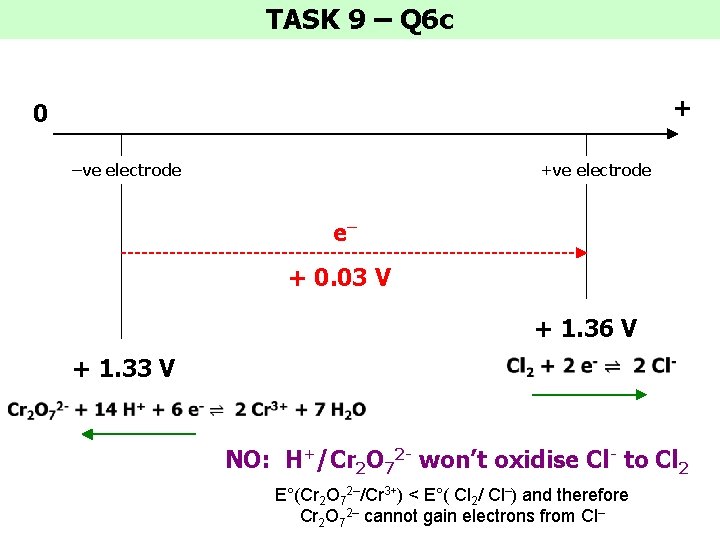
TASK 9 – Q 6 c + 0 –ve electrode +ve electrode e– + 0. 03 V + 1. 36 V + 1. 33 V NO: H+/Cr 2 O 72 - won’t oxidise Cl- to Cl 2 E°(Cr 2 O 72–/Cr 3+) < E°( Cl 2/ Cl–) and therefore Cr 2 O 72– cannot gain electrons from Cl–

TASK 9 – Q 6 d Pt(s)|Cl-(aq)|Cl 2(g)||Mn. O 4 - (aq), H+(aq), Mn 2+(aq)|Pt(s) + 0 –ve electrode +ve electrode e– + 0. 03 V + 1. 51 V + 1. 36 V YES: H+/Mn. O 4 - oxidises Cl- to Cl 2 E°(Mn. O 4–/Mn 2+) > E°(Cl 2/ Cl–) and therefore Mn. O 4– gains electrons from Cl–

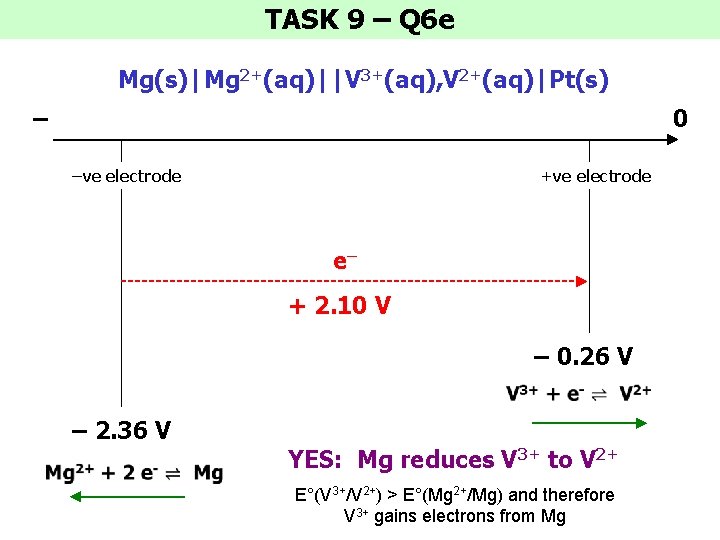
TASK 9 – Q 6 e Mg(s)|Mg 2+(aq)||V 3+(aq), V 2+(aq)|Pt(s) – 0 –ve electrode +ve electrode e– + 2. 10 V – 0. 26 V – 2. 36 V YES: Mg reduces V 3+ to V 2+ E°(V 3+/V 2+) > E°(Mg 2+/Mg) and therefore V 3+ gains electrons from Mg

Commercial Cells rechargeable Non

Non-rechargeable cells – Zinc-carbon • Standard cell • Short life – 0. 80 V Zn(NH 3)22+ + 2 e- Zn + 2 NH 3 +0. 70 V 2 Mn. O 2 + 2 H+ + 2 e– Mn 2 O 3 + H 2 O 2 Mn. O 2 + 2 H+ + Zn + 2 NH 3 Mn 2 O 3 + H 2 O + Zn(NH 3)22+ Emf = +1. 50 V

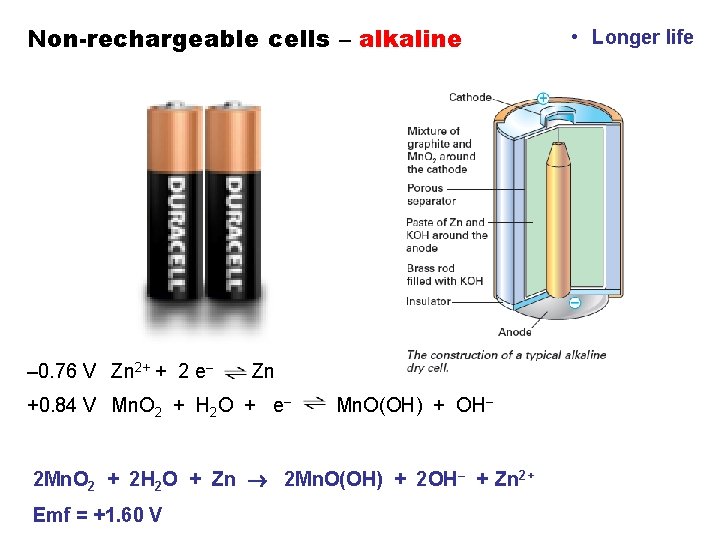
Non-rechargeable cells – alkaline – 0. 76 V Zn 2+ + 2 e– Zn +0. 84 V Mn. O 2 + H 2 O + e– Mn. O(OH) + OH– 2 Mn. O 2 + 2 H 2 O + Zn 2 Mn. O(OH) + 2 OH– + Zn 2+ Emf = +1. 60 V • Longer life

Commercial Cells Rechargeable

Rechargeable cells – Li ion • Rechargeable • Most common rechargeable cell +0. 60 V Li+ + Co. O 2 + e– – 3. 00 V Li+ + e– Li. Co. O 2 Li In use: Co. O 2 + Li Li. Co. O 2 Charging: Li. Co. O 2 + Li Emf = +3. 60 V

Rechargeable cells – lead-acid +1. 68 V Pb. O 2 + 3 H+ + HSO 4– + 2 e– – 0. 36 V Pb. SO 4 + H+ + 2 e– In use: • Used in sealed car batteries (6 cells giving about 12 V overall) Pb. SO 4 + 2 H 2 O Pb + HSO 4– Pb. O 2 + Pb + 2 H+ + 2 HSO 4– 2 Pb. SO 4 + 2 H 2 O Charging: 2 Pb. SO 4 + 2 H 2 O Pb. O 2 + Pb + 2 H+ + 2 HSO 4– Emf = +2. 04 V

Rechargeable cells – nickel-cadmium +0. 52 V Ni. O(OH) + 2 H 2 O + 2 e– – 0. 88 V Cd(OH)2 + 2 e– In use: Ni(OH)2 + 2 OH– Cd + 2 OH– Ni. O(OH) + 2 H 2 O + Cd Ni(OH)2 + Cd(OH)2 Charging: Ni(OH)2 + Cd(OH)2 Ni. O(OH) + 2 H 2 O + Cd Emf = +1. 40 V

Commercial Cells Fuel


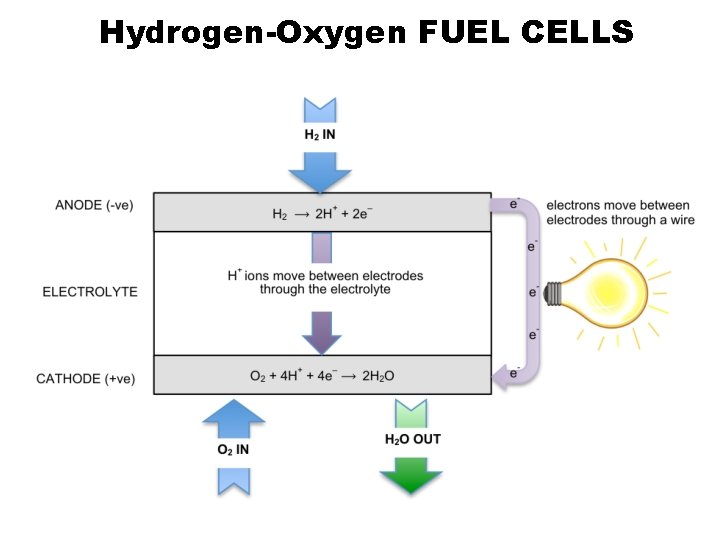
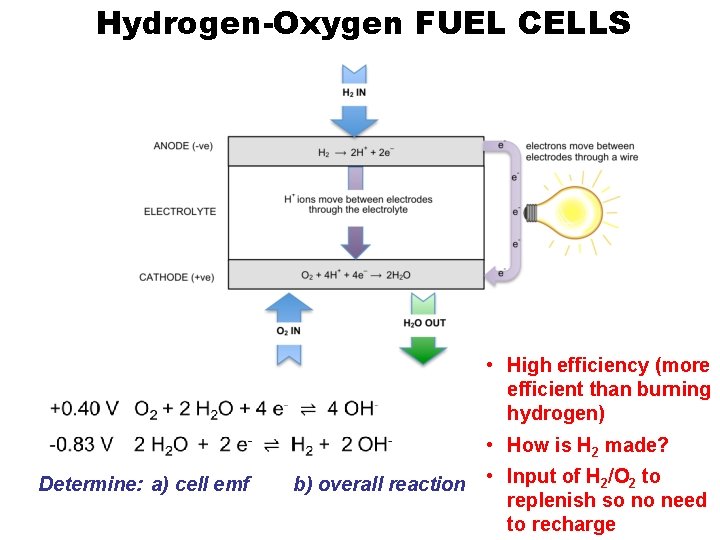
Hydrogen-Oxygen FUEL CELLS

Hydrogen-Oxygen FUEL CELLS • High efficiency (more efficient than burning hydrogen) • How is H 2 made? Determine: a) cell emf b) overall reaction • Input of H 2/O 2 to replenish so no need to recharge

hydrogen fuel cell (alkaline) Negative electrode (anode) H 2 + 2 OH– � 2 H 2 O + 2 e– Positive electrode (cathode) O 2 + 2 H 2 O + 4 e– � 4 OH– E° = – 0. 83 V hydrogen fuel cell (acidic) H 2 � 2 H+ + 2 e– E° = +0. 00 V O 2 + 4 H+ + 4 e– � 2 H 2 O E° = +0. 40 V E° = +1. 23 V Overall equation 2 H 2 + O 2 � 2 H 2 O Cell emf +1. 23 V © www. chemsheets. co. uk A 2 1077 2 -July-

Benefits & risks of using cells Benefits Using cells Using nonrechargeable cells Using rechargeable cells Risks portable source of electrical energy waste issues cheap waste issues less waste cheaper in the long run some waste issues (at end of useful life) lower environmental impact need constant supply of fuels only waste product is water hydrogen is flammable & explosive do not need re-charging hydrogen usually made using fossil fuels high cost of fuel cells Using hydrogen fuel cells very efficient © www. chemsheets. co. uk A 2 1077 2 -July-