CHEMSHEETS CELLS BATTERIES www chemsheets co uk GCSE

CHEMSHEETS CELLS & BATTERIES © www. chemsheets. co. uk GCSE 1161 20 -Feb-17

SIMPLE CELL Two electrodes (two different metals) e. g. Cu + Zn Electrolyte (liquid that allows electricity to flow e. g. juice in lemon, salt solution

Factors affecting the voltage: • Identity of metals (bigger the difference in reactivity → bigger voltage) • Electrolyte (identity and/or concentration) © www. chemsheets. co. uk GCSE 1161 20 -Feb-17

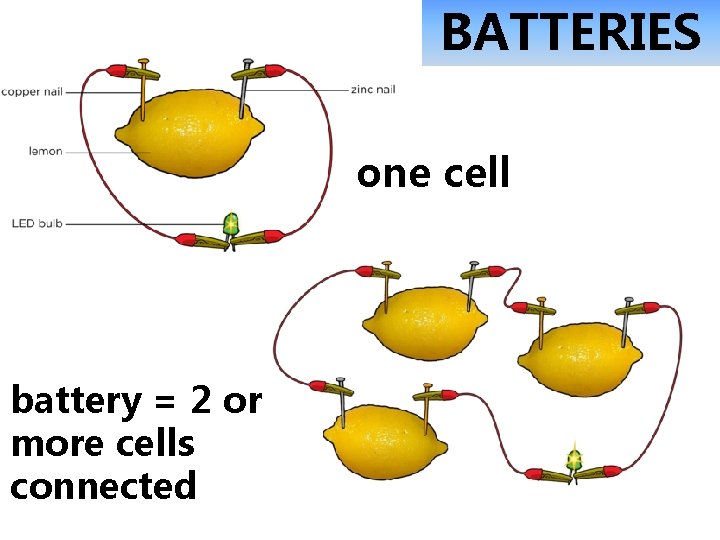
BATTERIES one cell battery = 2 or more cells connected

6 V battery contains four 1. 5 V cells
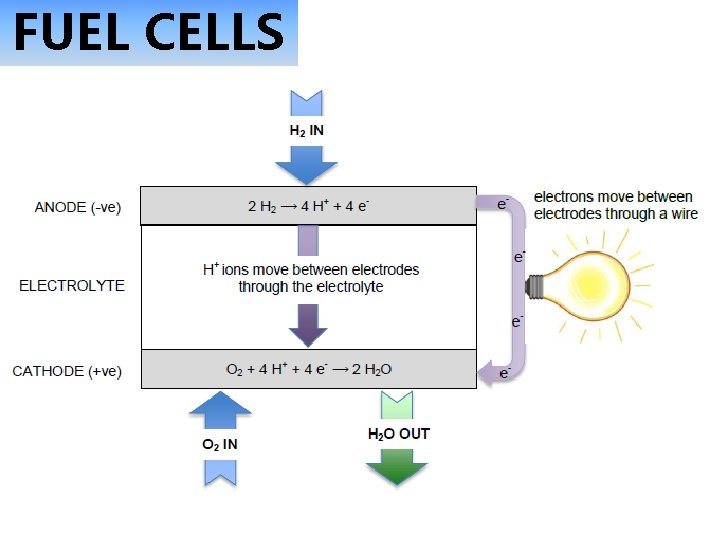
FUEL CELLS


FUEL CELLS Toyota Mira

FUEL CELLS

FUEL CELLS Simple hydrogen

Hydrogen fuel cell stack
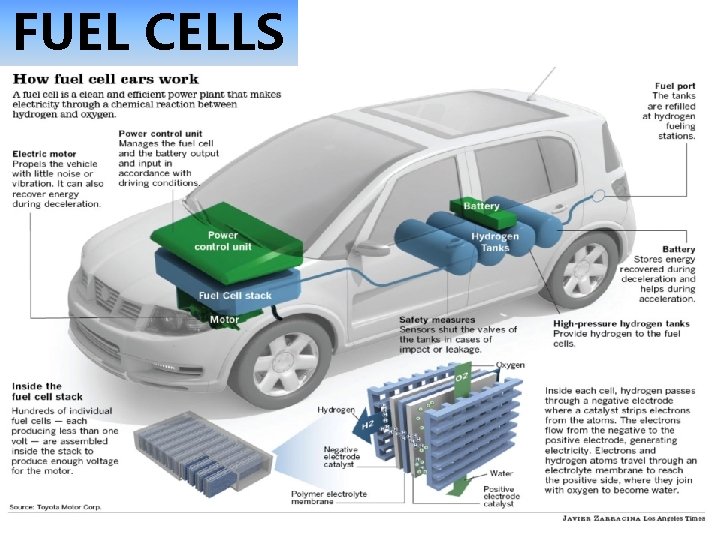
FUEL CELLS



type of cell note non-rechargeable goes flat (the can be recharged voltage drops to (because the zero) when the chemical reactions inside reactions can be stop (because one reversed when of the reactants connected to an has been used external electric up) current) example alkaline cells lithium-ion cells s (in mobile phones) pros cheap can be used many times cheaper than fuel cells cons lots of used cells takes time to fuel cells have a continuous fuel supply so do not go flat or need recharging hydrogen fuel cell only waste product is water continuous supply of electricity (as long as there is a continuous supply of fuel) very expensive
- Slides: 16