CHEMSHEETS BOND POLARITY www chemsheets co uk AS

CHEMSHEETS BOND POLARITY © www. chemsheets. co. uk AS 1026 07 -Jun-2016
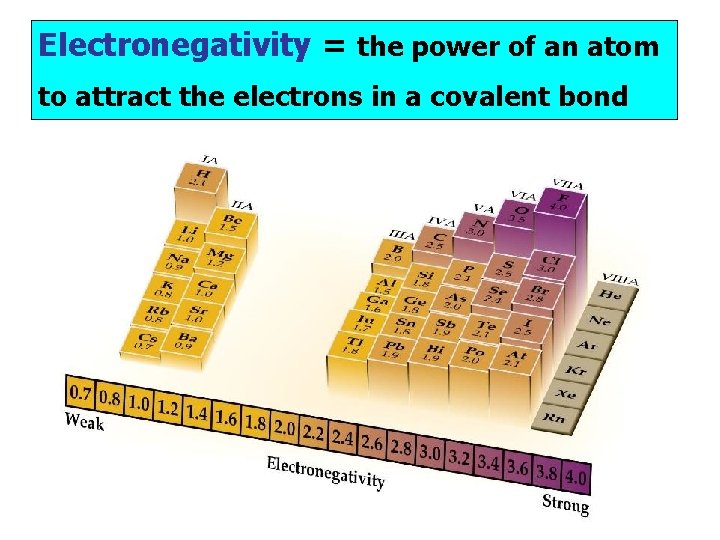
Electronegativity = the power of an atom to attract the electrons in a covalent bond
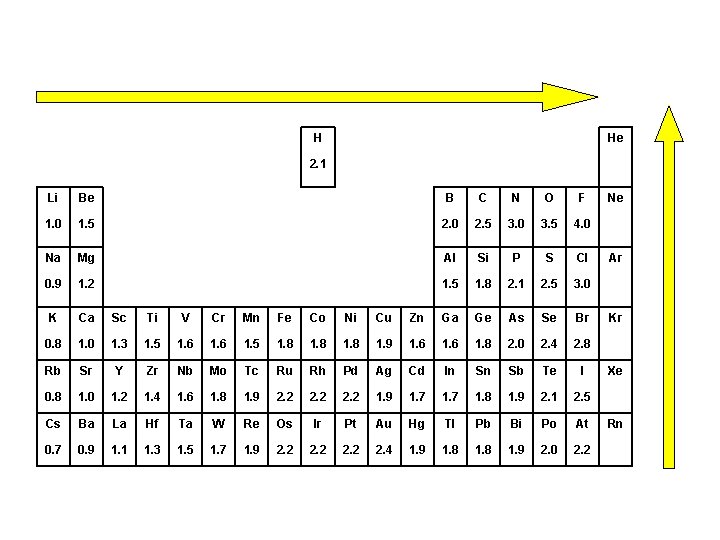
H He 2. 1 Li Be B C N O F 1. 0 1. 5 2. 0 2. 5 3. 0 3. 5 4. 0 Na Mg Al Si P S Cl 0. 9 1. 2 1. 5 1. 8 2. 1 2. 5 3. 0 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br 0. 8 1. 0 1. 3 1. 5 1. 6 1. 5 1. 8 1. 9 1. 6 1. 8 2. 0 2. 4 2. 8 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I 0. 8 1. 0 1. 2 1. 4 1. 6 1. 8 1. 9 2. 2 1. 9 1. 7 1. 8 1. 9 2. 1 2. 5 Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At 0. 7 0. 9 1. 1 1. 3 1. 5 1. 7 1. 9 2. 2 2. 4 1. 9 1. 8 1. 9 2. 0 2. 2 Ne Ar Kr Xe Rn
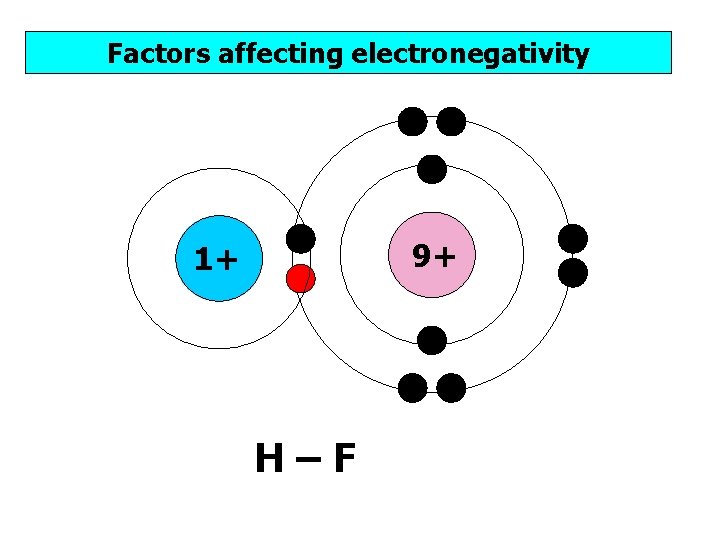
Factors affecting electronegativity 9+ 1+ H–F

Factors affecting electronegativity 1) Nuclear charge – the more protons, the stronger the attraction from the nucleus to the bonding pair of electrons. 2) Atomic radius – the closer the bonding electrons to the nucleus, the stronger the attraction from the nucleus to the bonding pair of electrons. 3) Shielding – the less shells of electrons shielding (repelling) the bonding electrons, the stronger the attraction from the nucleus to the bonding pair of electrons.

Factors affecting electronegativity 1) Nuclear charge – more protons, stronger attraction between nucleus and bonding pair of electrons. 2) Atomic radius – closer to the nucleus, stronger attraction between nucleus and bonding pair of electrons. 3) Shielding – less shells of electrons between the nucleus and the electrons, less shielding (less repulsion), stronger attraction between nucleus and bonding pair of electrons.

Trend down a group Electronegativity decreases • Atomic radius increases • More shielding • Less attraction between nucleus and bonding pair of electrons
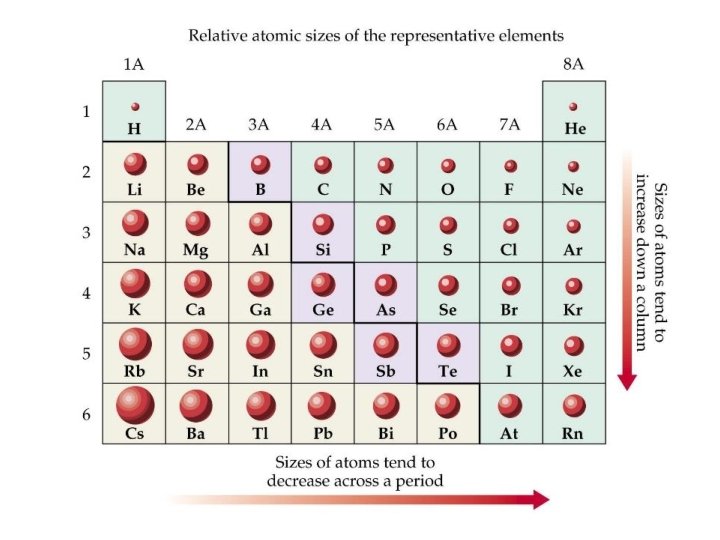

Trend across a period Electronegativity increases • Atomic radius decreases • More nuclear charge • Stronger attraction between nucleus and bonding pair of electrons

Formation of a covalent bond

Non-polar bond Polar bond
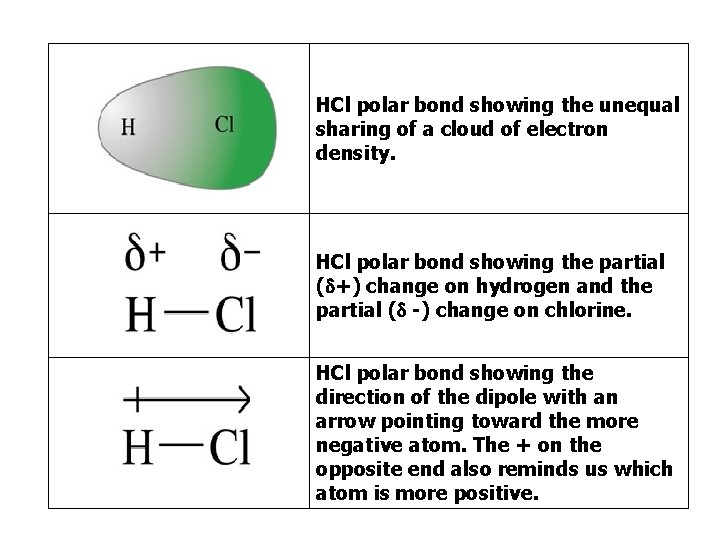
HCl polar bond showing the unequal sharing of a cloud of electron density. HCl polar bond showing the partial ( +) change on hydrogen and the partial ( -) change on chlorine. HCl polar bond showing the direction of the dipole with an arrow pointing toward the more negative atom. The + on the opposite end also reminds us which atom is more positive.
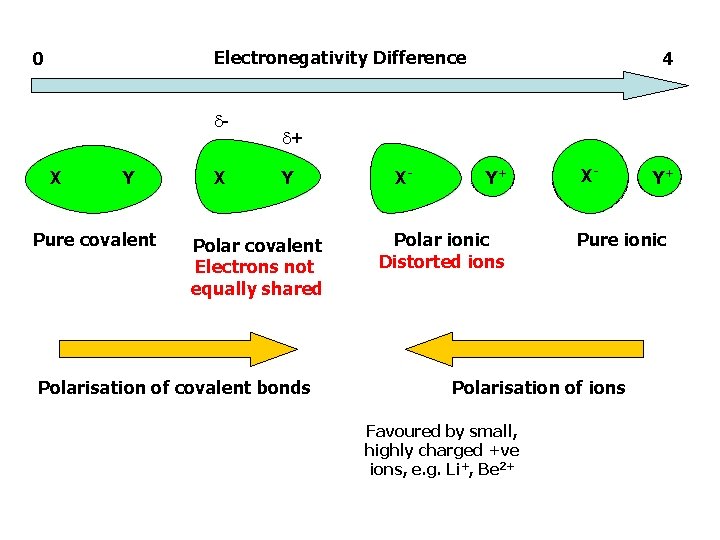
Electronegativity Difference 0 X Y Pure covalent X 4 + Y Polar covalent Electrons not equally shared Polarisation of covalent bonds X- Y+ Polar ionic Distorted ions X- Pure ionic Polarisation of ions Favoured by small, highly charged +ve ions, e. g. Li+, Be 2+ Y+
Covalent bonding Ionic bonding
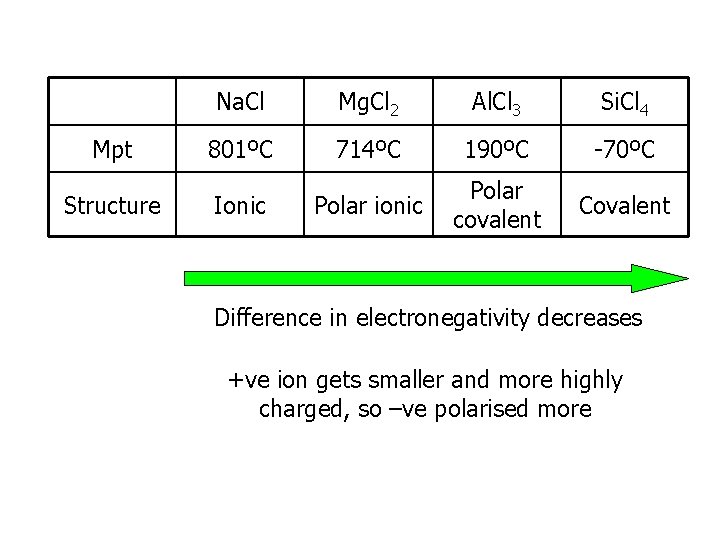
Mpt Structure Na. Cl Mg. Cl 2 Al. Cl 3 Si. Cl 4 801ºC 714ºC 190ºC -70ºC Polar ionic Polar covalent Covalent Ionic Difference in electronegativity decreases +ve ion gets smaller and more highly charged, so –ve polarised more
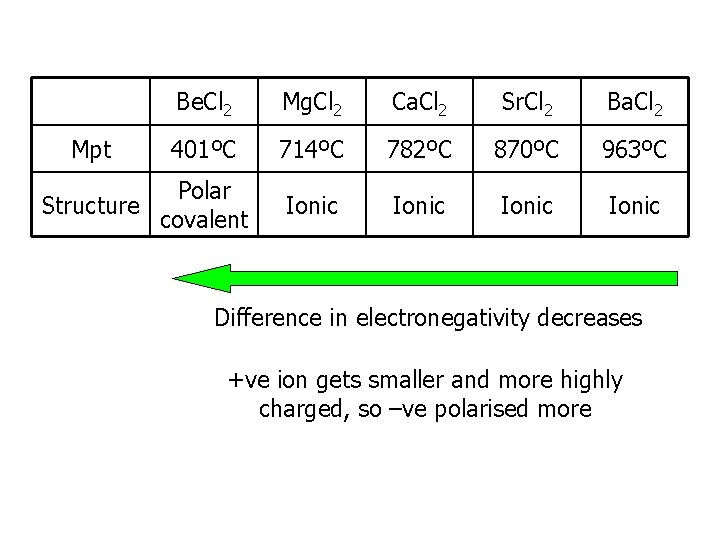
Mpt Be. Cl 2 Mg. Cl 2 Ca. Cl 2 Sr. Cl 2 Ba. Cl 2 401ºC 714ºC 782ºC 870ºC 963ºC Ionic Polar Structure covalent Difference in electronegativity decreases +ve ion gets smaller and more highly charged, so –ve polarised more
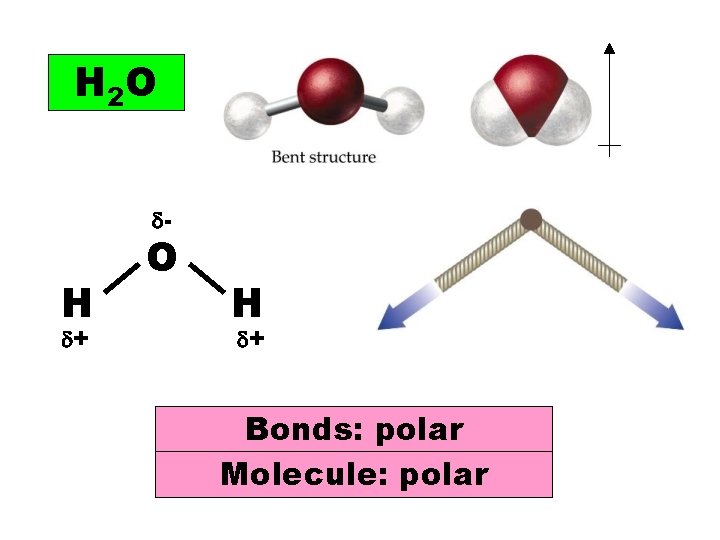
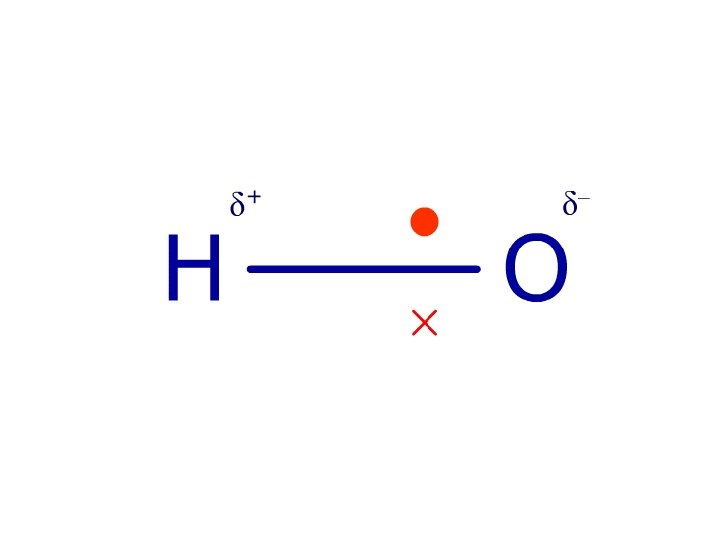
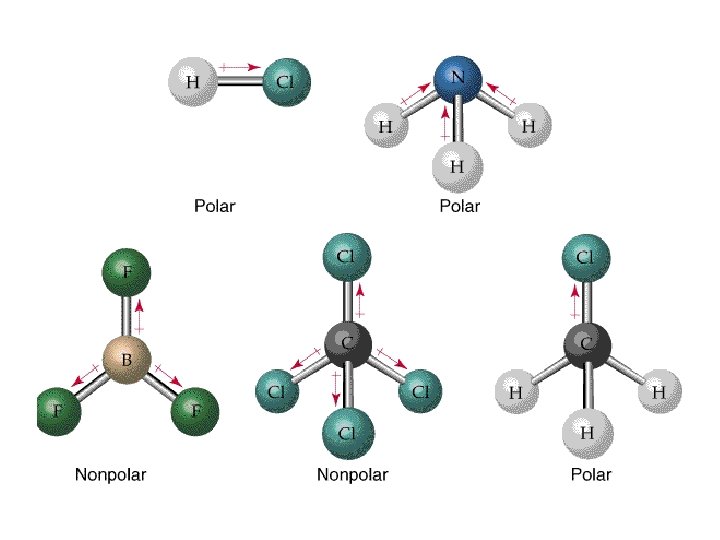
H 2 O - H + O H + Bonds: polar Molecule: polar
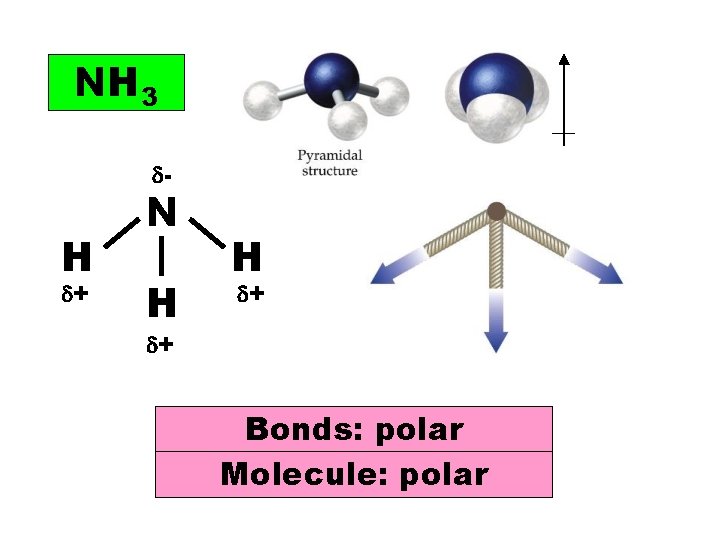
NH 3 - H + N H H + + Bonds: polar Molecule: polar
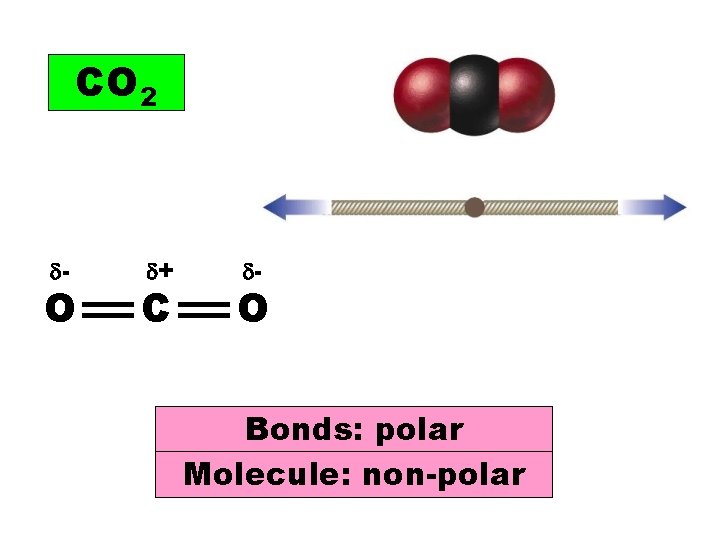
CO 2 - O + C - O Bonds: polar Molecule: non-polar
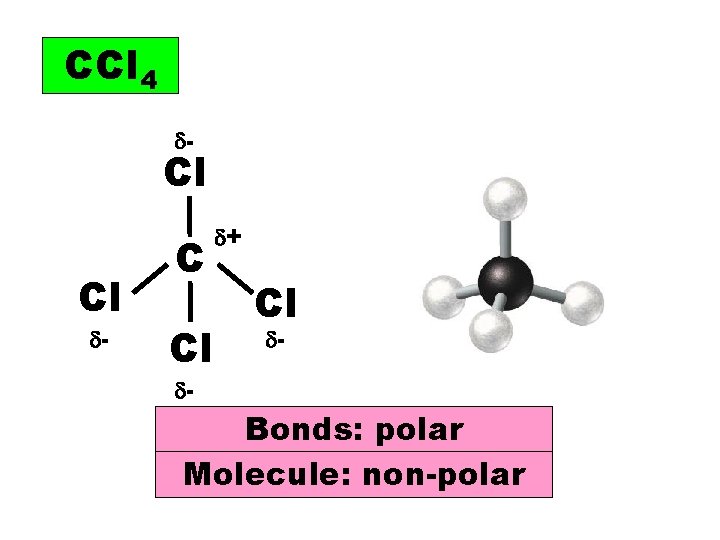
CCl 4 - Cl Cl - C Cl + Cl - - Bonds: polar Molecule: non-polar
- Slides: 22