CHEMOTHERAPY Prepared By Dr Khalid Ahmad Shadid Chemistry

CHEMOTHERAPY Prepared By Dr. Khalid Ahmad Shadid Chemistry Department Islamic university in madinah 1
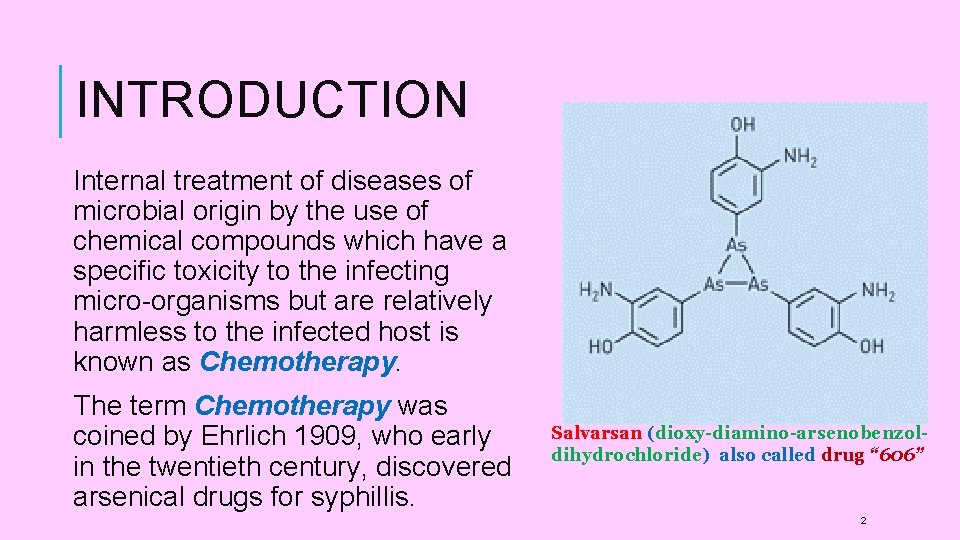
INTRODUCTION Internal treatment of diseases of microbial origin by the use of chemical compounds which have a specific toxicity to the infecting micro-organisms but are relatively harmless to the infected host is known as Chemotherapy. The term Chemotherapy was coined by Ehrlich 1909, who early in the twentieth century, discovered arsenical drugs for syphillis. Salvarsan (dioxy-diamino-arsenobenzoldihydrochloride) also called drug “ 606” 2

INTRODUCTION A therapeutic agent should have following properties : (1) it should be harmful to the invading micro-organism but almost harmless or non-toxic to the host. (2) its action should be localized at the site of the disease. (3) as far as possible it should be free from side effects. 3

INTRODUCTION The success of Chemotherpy depends upon the nature of the interaction of the drug with the invading organism and the mode by which the latter responds to its altered environment. A drug is said to be ‘bacteriostatic’ when it stops further growth and multiplication of bacteria and it is said to be ‘bactericidial’ when stops outrightly kills them. The discovery of antibacterial activity of sulphanilamide in 1934 and of Penicillin in 1940 marked the opening of a new era in the treatment of systemic microbial infections. Before these, means of combating with microorganisms were very limited. 4

SULPHONAMIDES sulphanilamide Domagk 1934 discovered that Prontocil, a dye, was remarkably effective against streptococcal and staphyllococcal infections in humans. It was shown that the antibacterial action of Prontosil was due to its transformation in the body into sulphanilamide This led to the synthesis of an enormous number of sulphonamide drugs, several of which had broader antibacterial activity and lower toxicity than the parent compound sulphanilamide. Before the arrival of antibiotics, they were the main drugs in the treatment of bacterial infections. Prontocil 5
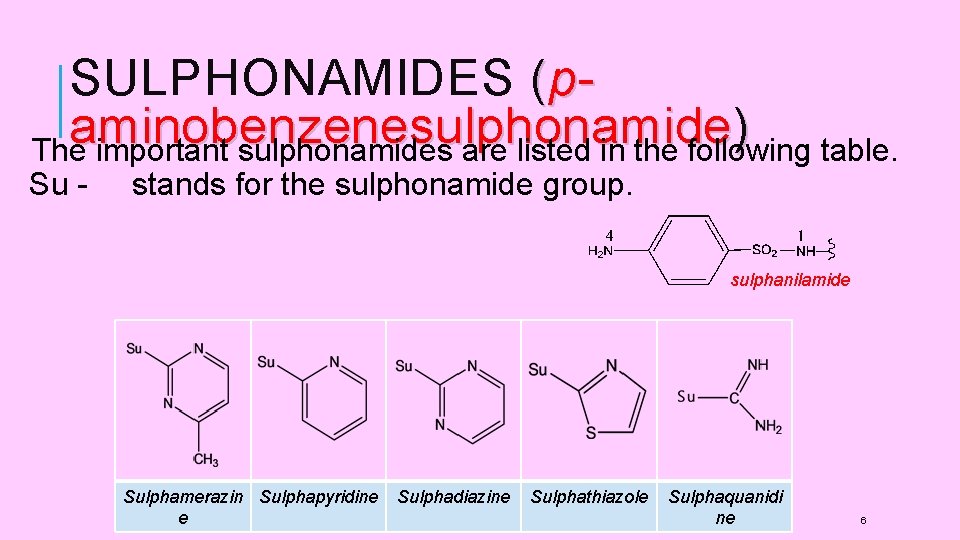
SULPHONAMIDES (paminobenzenesulphonamide ) The important sulphonamides are listed in the following table. Su - stands for the sulphonamide group. sulphanilamide Sulphamerazin Sulphapyridine e Sulphadiazine Sulphathiazole Sulphaquanidi ne 6

SULPHANILAMIDE (p- aminobenzenesulphonamide) Sulphanilamide maybe prepared from acetanilide and Chlorosulfuric acid : P-chloroacetamidobenzenesulphonic acid 7

SULPHONAMIDES Most of the sulpha drugs, at their concentrations used in chemotherapy, are only bacteriostatic in their action. They prevent multiplication of the infecting organism and thus allow the normal defense mechanism of the host to eradicate the infection. The advent of antibiotics a few years later marked an even greater advance in chemotherapy. 8

ANTIBIOTICS Antibiotics are substances produced ( metabolites ) by microorganisms which are harmful to other micro-organisms growing in the same medium and inhibit the growth or activity of certain bacteria ( bacterio static effect ) or kill the bacteria ( bactericidal ). Thus, one microbe is put against another microbe. Alexander Fleming, in 1929, observed that a mould, Penicillium notatum, produced a substance that possessed potent 9

ANTIBIOTICS Penicillin had a very high activity against Gram positive organisms including Staphylococci, Streptococci, Pneumococci, Gonococci etc. It was first used clinically in 1941 and the results were so astonishing that world-wide interest was roused immediately. This stimulated search for other antibiotics, and since 1940, a large number of antibiotics have been isolated from fermentation mold of bacteria and actinomycetes. In 1944, Waksman isolated Streptomycin from a strain of actinomycetes, streptomyces griseus. Antibiotics like Chloromycetin, Chlorotetracyclin, Oxytetracyclinare used to combat a large number of pathogens from the larger viruses to protozoa, 10
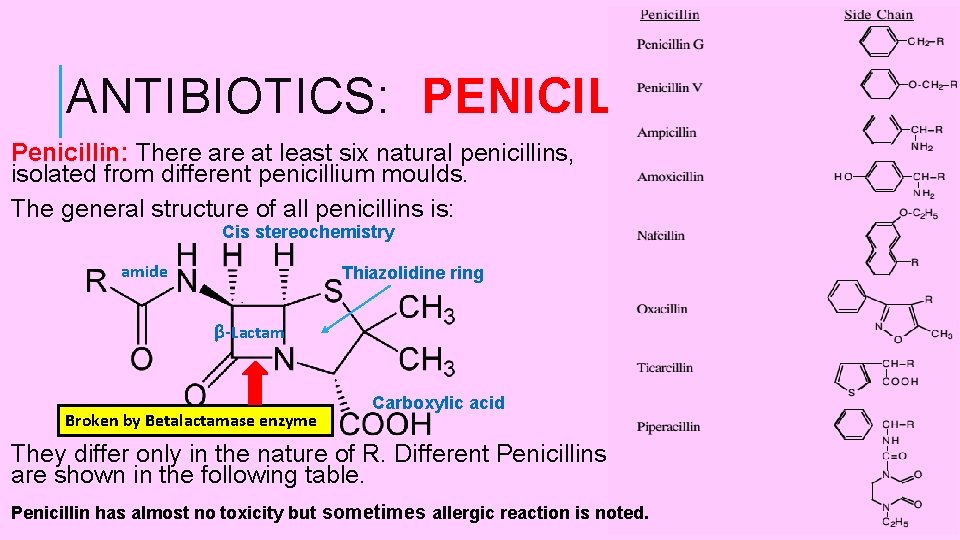
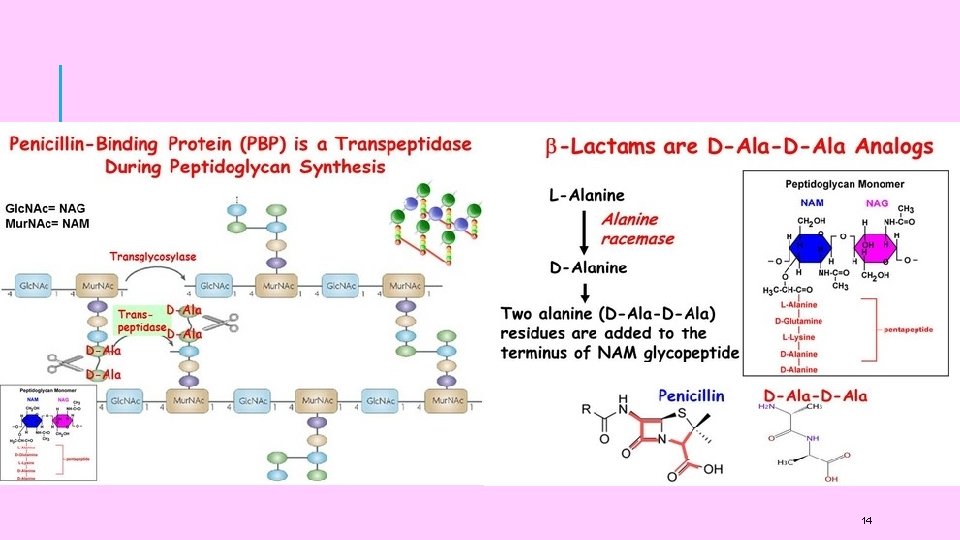
ANTIBIOTICS: PENICILLIN Penicillin: There at least six natural penicillins, isolated from different penicillium moulds. The general structure of all penicillins is: Cis stereochemistry amide Thiazolidine ring β-Lactam Broken by Betalactamase enzyme Carboxylic acid They differ only in the nature of R. Different Penicillins are shown in the following table. Penicillin has almost no toxicity but sometimes allergic reaction is noted. 11
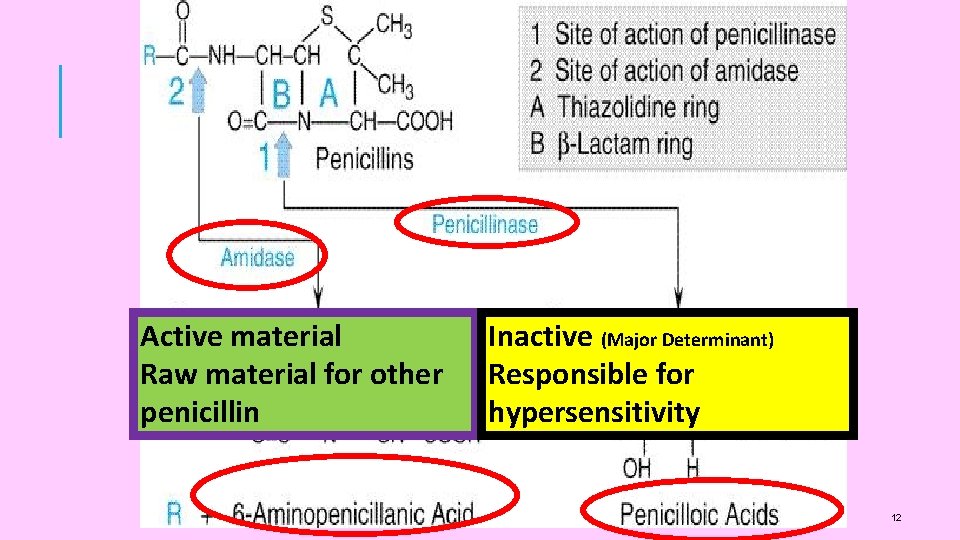
Active material Raw material for other penicillin Inactive (Major Determinant) Responsible for hypersensitivity 12
13
14

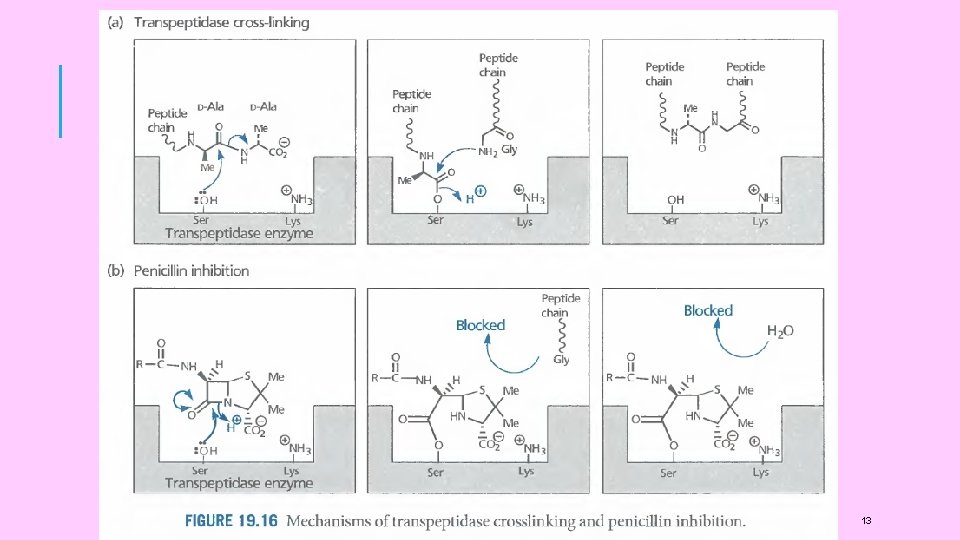
DRUG ACTION OF ANTIBACTERIALS The present concept of the mechanism of action of antibacterials is that they act by ‘selective interference’ with certain phases of microbial metabolism. This ‘selective toxic action’ can occur in either of the two ways: (i) indirectly, by inhibiting the synthesis of necessary nutrient which is essential for pathogenic microbes. Consequently, a number of subsequent metabolic processes will be blocked for want of this essential nutrient. or, (ii) directly, by interfering with the structure or metabolism of bacterial cells. A drug may be similar to the metabolite and although it will not inhibit the synthesis of the nutrient, it will interfere with the utilization of the metabolite in the conversion of more complex materials needed in the life of the 15
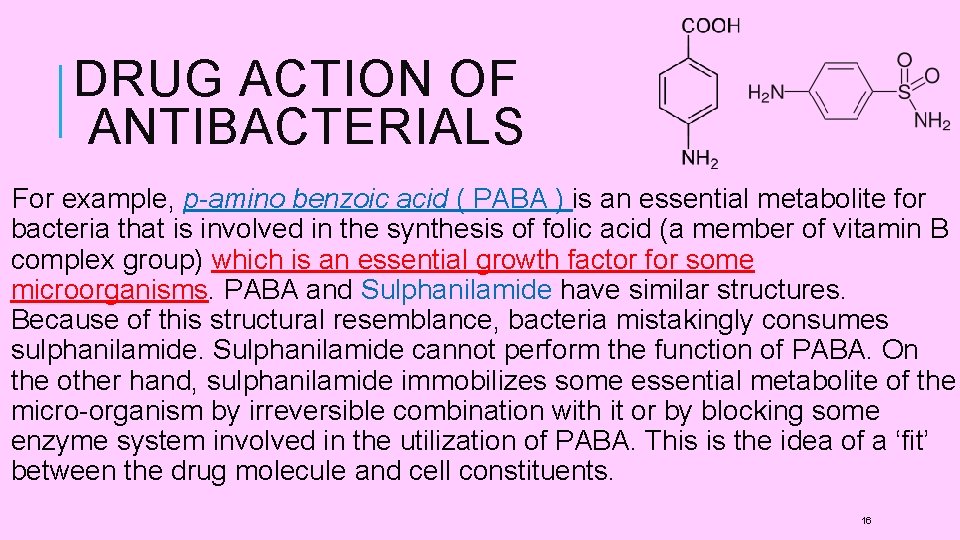
DRUG ACTION OF ANTIBACTERIALS For example, p-amino benzoic acid ( PABA ) is an essential metabolite for bacteria that is involved in the synthesis of folic acid (a member of vitamin B complex group) which is an essential growth factor for some microorganisms. PABA and Sulphanilamide have similar structures. Because of this structural resemblance, bacteria mistakingly consumes sulphanilamide. Sulphanilamide cannot perform the function of PABA. On the other hand, sulphanilamide immobilizes some essential metabolite of the micro-organism by irreversible combination with it or by blocking some enzyme system involved in the utilization of PABA. This is the idea of a ‘fit’ between the drug molecule and cell constituents. 16
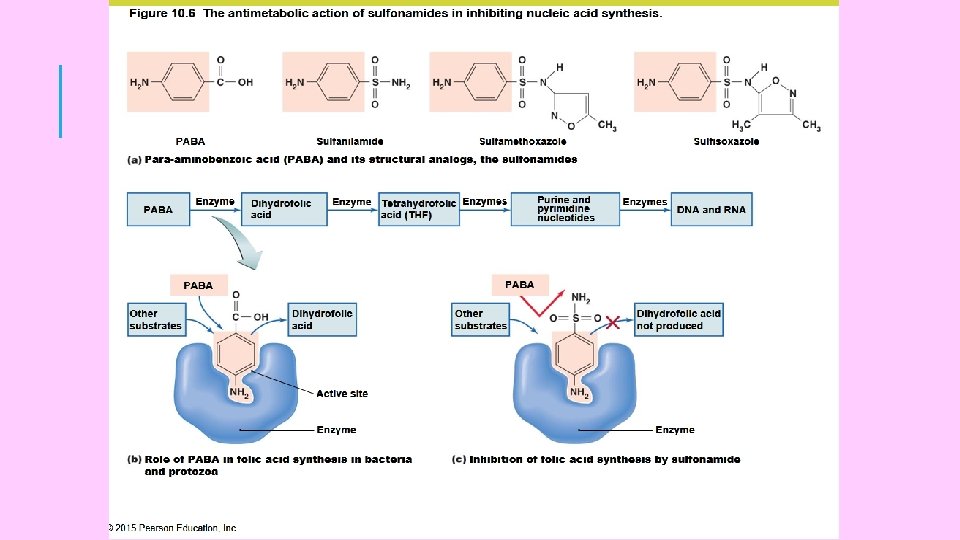

DRUG ACTION OF ANTIBACTERIALS Most of the antibiotics exert their toxic action on bacteria by impairing either: (i) structure and/or synthesis of the bacterial cell wall. or (ii) the function and/or synthesis of the cytoplasmic membrane or (iii) the intracellular synthesis of proteins and/or nucleic acid Protein synthesis is a fundamental process in all living cells and those antibiotics that interfere with the synthesis of proteins within bacterial cells are effective against both Gram positive and Gram negative bacteria Penicillin G inhibits the synthesis of bacterial cell wall. Chloromycetin and tetracyclines inhibit intracellular protein 18
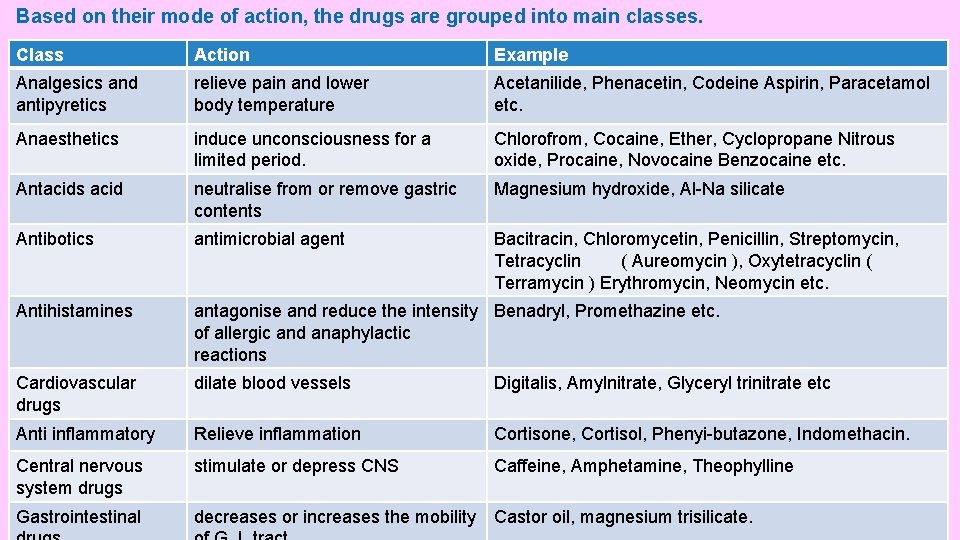
Based on their mode of action, the drugs are grouped into main classes. Class Action Example Analgesics and antipyretics relieve pain and lower body temperature Acetanilide, Phenacetin, Codeine Aspirin, Paracetamol etc. Anaesthetics induce unconsciousness for a limited period. Chlorofrom, Cocaine, Ether, Cyclopropane Nitrous oxide, Procaine, Novocaine Benzocaine etc. Antacids acid neutralise from or remove gastric contents Magnesium hydroxide, Al-Na silicate Antibotics antimicrobial agent Bacitracin, Chloromycetin, Penicillin, Streptomycin, Tetracyclin ( Aureomycin ), Oxytetracyclin ( Terramycin ) Erythromycin, Neomycin etc. Antihistamines antagonise and reduce the intensity Benadryl, Promethazine etc. of allergic and anaphylactic reactions Cardiovascular drugs dilate blood vessels Digitalis, Amylnitrate, Glyceryl trinitrate etc Anti inflammatory Relieve inflammation Cortisone, Cortisol, Phenyi-butazone, Indomethacin. Central nervous system drugs stimulate or depress CNS Caffeine, Amphetamine, Theophylline Gastrointestinal decreases or increases the mobility Castor oil, magnesium trisilicate.
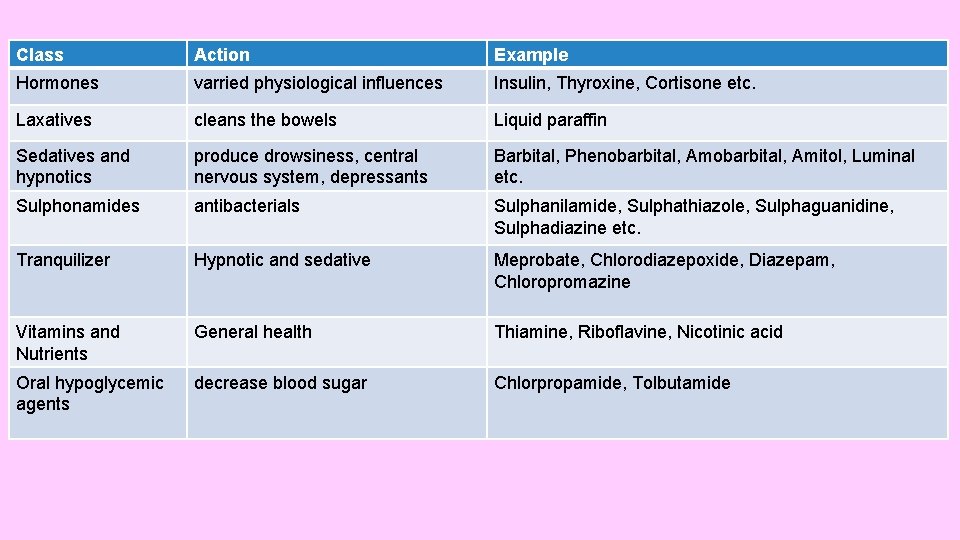
Class Action Example Hormones varried physiological influences Insulin, Thyroxine, Cortisone etc. Laxatives cleans the bowels Liquid paraffin Sedatives and hypnotics produce drowsiness, central nervous system, depressants Barbital, Phenobarbital, Amitol, Luminal etc. Sulphonamides antibacterials Sulphanilamide, Sulphathiazole, Sulphaguanidine, Sulphadiazine etc. Tranquilizer Hypnotic and sedative Meprobate, Chlorodiazepoxide, Diazepam, Chloropromazine Vitamins and Nutrients General health Thiamine, Riboflavine, Nicotinic acid Oral hypoglycemic agents decrease blood sugar Chlorpropamide, Tolbutamide

SYNTHESES OF SOME INDIVIDUAL DRAGS 1. Paracetamol : ( p-acetamidophenol or p-acetyl amino phenol). It is a white, crystalline, odorless powder. Use : It is used as an analgesic and antipyretic. It can relieve headache, arthritic and other pains arising from muscles and joints, and It is also known as acetaminophen. It is an intermediate compound in the preparation of Phenacetin, another analgesic and antipyretic. Preparation: Paracetamol is prepared as follows. (i) by reduction of p-nitrophenol with Tin and glacial acetic acid (with or without aceticanhydride) Phenacetin 21 (ii) from p-amino phenol by the action of glacial acetic acid and acetic anhydride. (as shown in second step in (
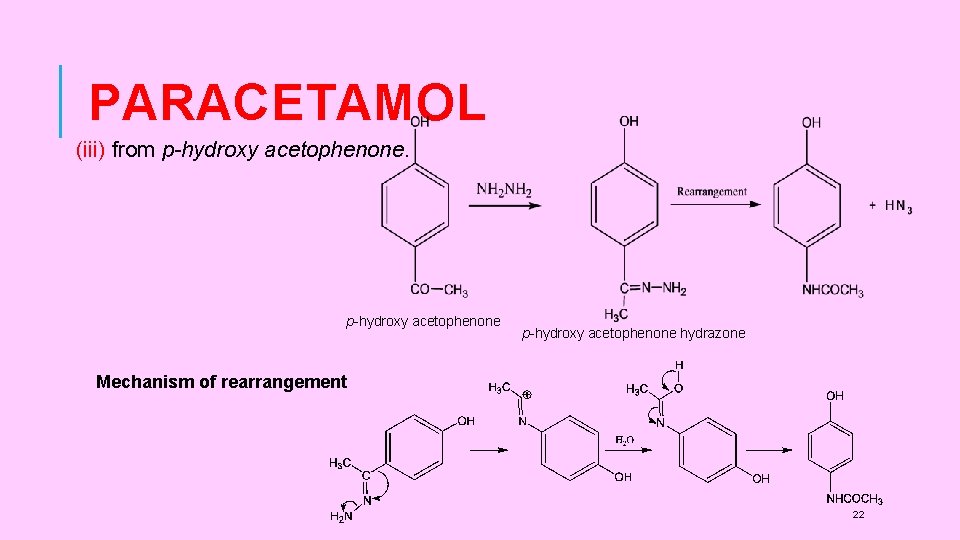
PARACETAMOL (iii) from p-hydroxy acetophenone hydrazone Mechanism of rearrangement 22
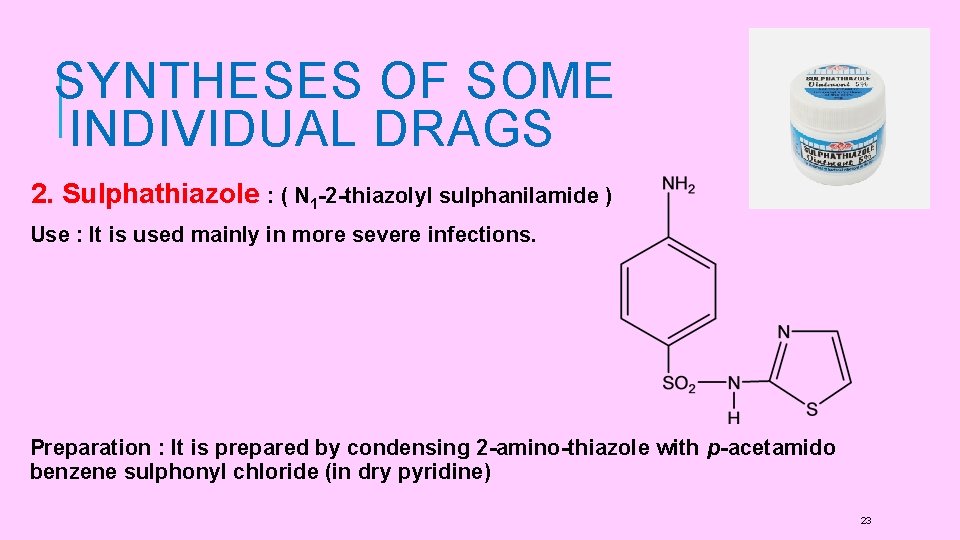
SYNTHESES OF SOME INDIVIDUAL DRAGS 2. Sulphathiazole : ( N 1 -2 -thiazolyl sulphanilamide ) Use : It is used mainly in more severe infections. Preparation : It is prepared by condensing 2 -amino-thiazole with p-acetamido benzene sulphonyl chloride (in dry pyridine) 23
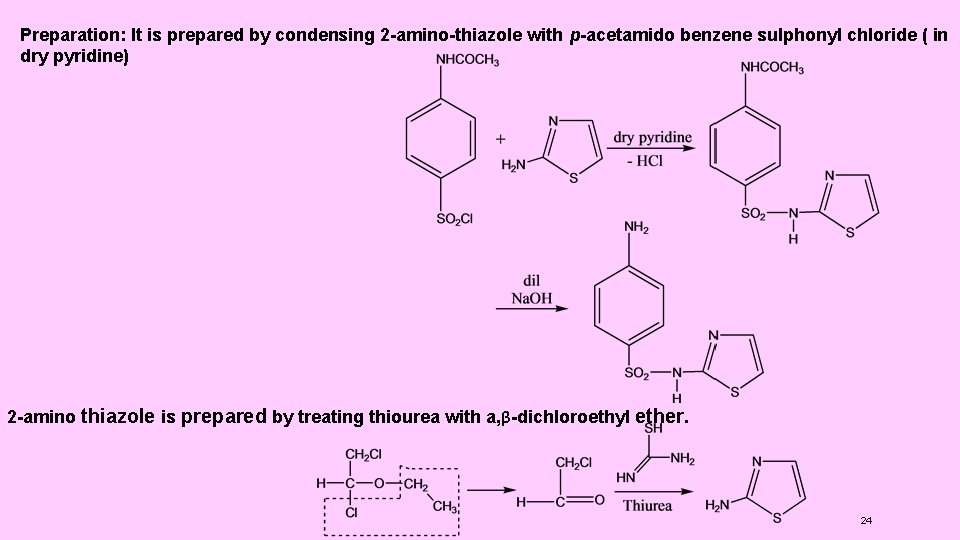
Preparation: It is prepared by condensing 2 -amino-thiazole with p-acetamido benzene sulphonyl chloride ( in dry pyridine) 2 -amino thiazole is prepared by treating thiourea with a, β-dichloroethyl ether. 24
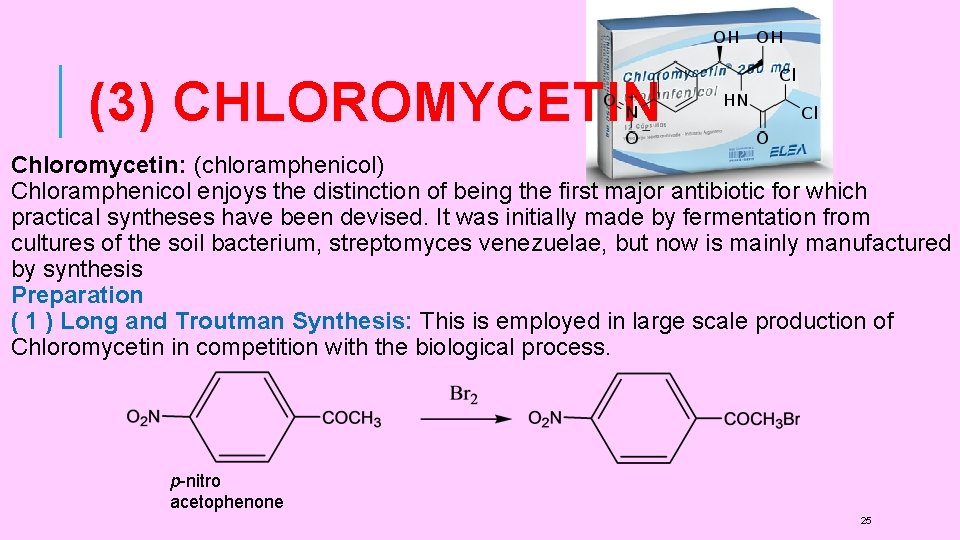
(3) CHLOROMYCETIN Chloromycetin: (chloramphenicol) Chloramphenicol enjoys the distinction of being the first major antibiotic for which practical syntheses have been devised. It was initially made by fermentation from cultures of the soil bacterium, streptomyces venezuelae, but now is mainly manufactured by synthesis Preparation ( 1 ) Long and Troutman Synthesis: This is employed in large scale production of Chloromycetin in competition with the biological process. p-nitro acetophenone 25
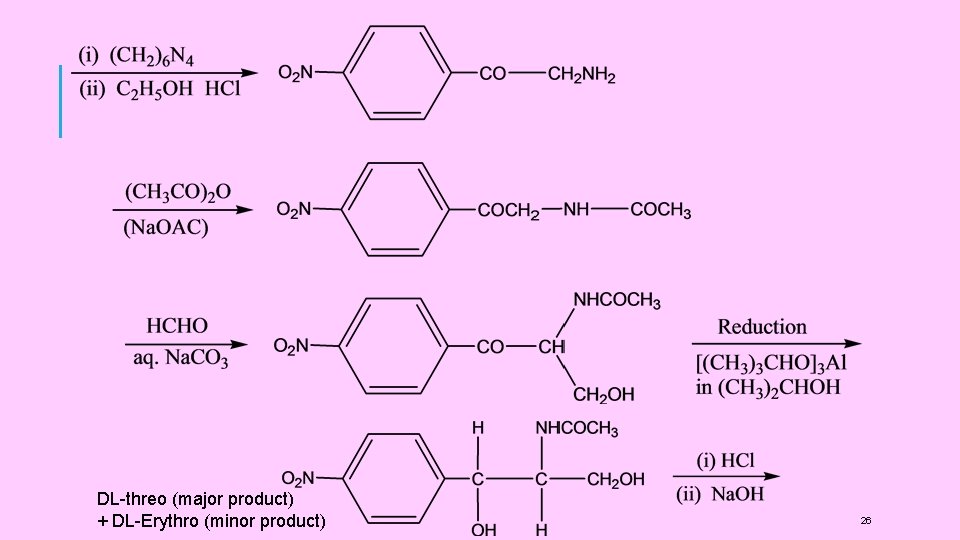
DL-threo (major product) + DL-Erythro (minor product) 26
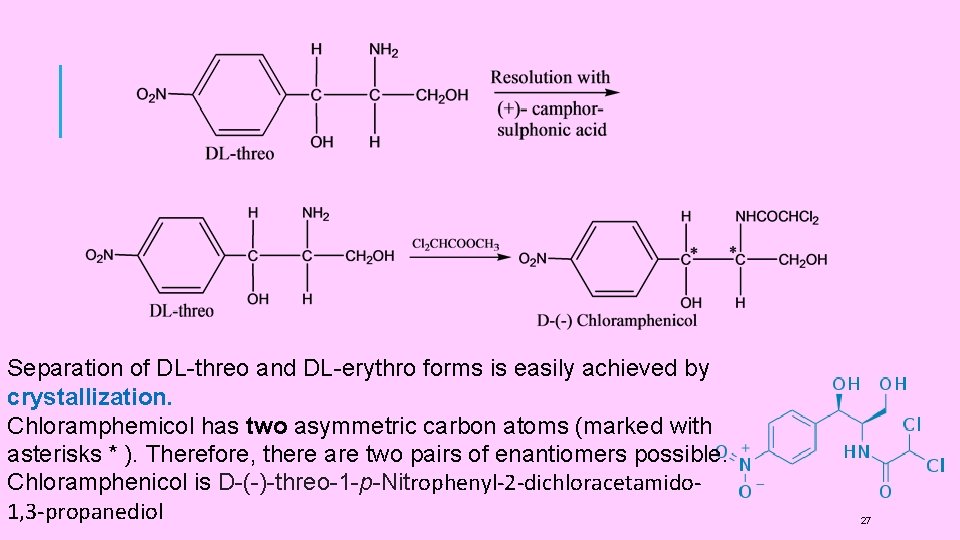
Separation of DL-threo and DL-erythro forms is easily achieved by crystallization. Chloramphemicol has two asymmetric carbon atoms (marked with asterisks * ). Therefore, there are two pairs of enantiomers possible. Chloramphenicol is D-(-)-threo-1 -p-Nitrophenyl-2 -dichloracetamido 1, 3 -propanediol 27

CHLOROMYCETIN: (CHLORAMPHENICOL) Use: Chloramphenicol is a broad spectrum antibiotic, active against both Gram positive and Gram negative bacteria, the typhus fever and certain viruses. Of the four stereoisomers, only D-threo form is active. It is a drug of choice for typhoid. Because of serious toxic effects its clinical use has been sharply curtailed. It is also interesting to note that Chloramphenicol is the first natural compound to contain a nitro group, the presence of — CHCl 2 group is also most unusual. 28

(4) SYNTHESIS OF PENICILLINS: (1) Synthesis of Penicillins V Sheehan et al, carried out the first successful synthesis of penicillins. He synthesised Penicillin V (Phenoxy methyl penicillin). It involved the following steps: (i) t-Butyl phthalimidomalonaldehydate(I) was prepared by condensing phthalimide with t-butyl chloroacetate, (Gabriel synthesis ), followed by formylation of a active methylene group (Claisen condensation ). (ii) Thus, in this compound (I), the amino group was protected by phthalimido group and one of the -COOH groups (the one later on involved in ring closure) was protected by tbutyl ester formation. (iii) The compound (I) was then condensed with D-penicillamine. (iv) Phthalimido group was then removed by reaction with hydrazine. The use of this reagent left the ester group intact. (v) Phenoxy-methyl group was introduced by treatment with phenoxyl acetyl chloride. (vi) t-butyl group was removed by hydrolysis with HC 1 to give a penicilloic acid (II) (vii) The final step of cyclisation ( formation of (β-lactam ring) was carried out on the KSalt of the penicilloic acid (II) with Dicyclohexyl carbodimide (DCC) to get Penicillin V 29
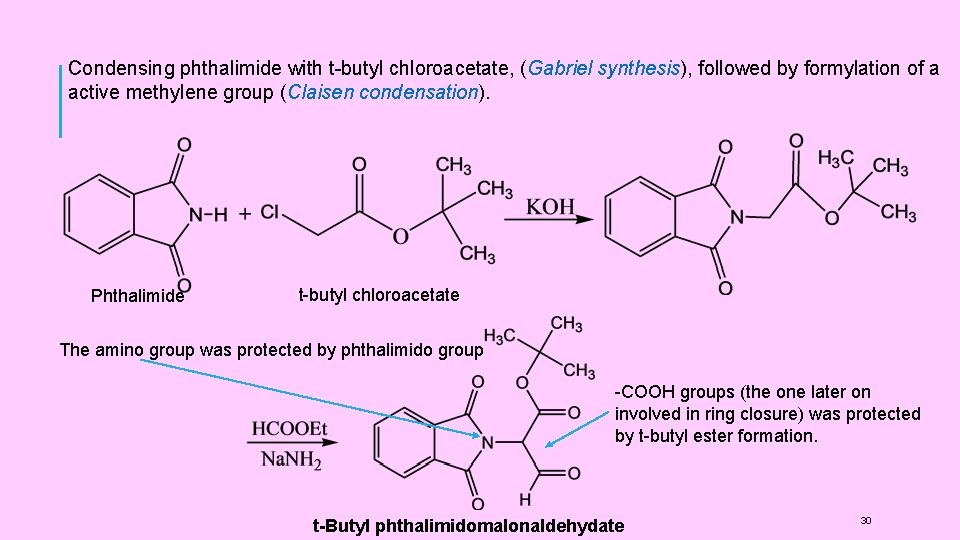
Condensing phthalimide with t-butyl chloroacetate, (Gabriel synthesis), followed by formylation of a active methylene group (Claisen condensation). Phthalimide t-butyl chloroacetate The amino group was protected by phthalimido group -COOH groups (the one later on involved in ring closure) was protected by t-butyl ester formation. t-Butyl phthalimidomalonaldehydate 30
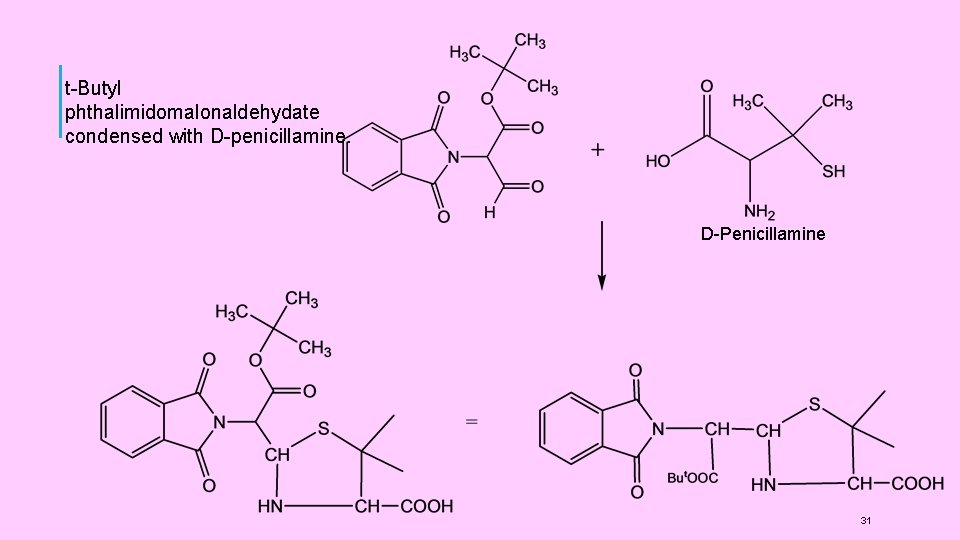
t-Butyl phthalimidomalonaldehydate condensed with D-penicillamine. D-Penicillamine 31
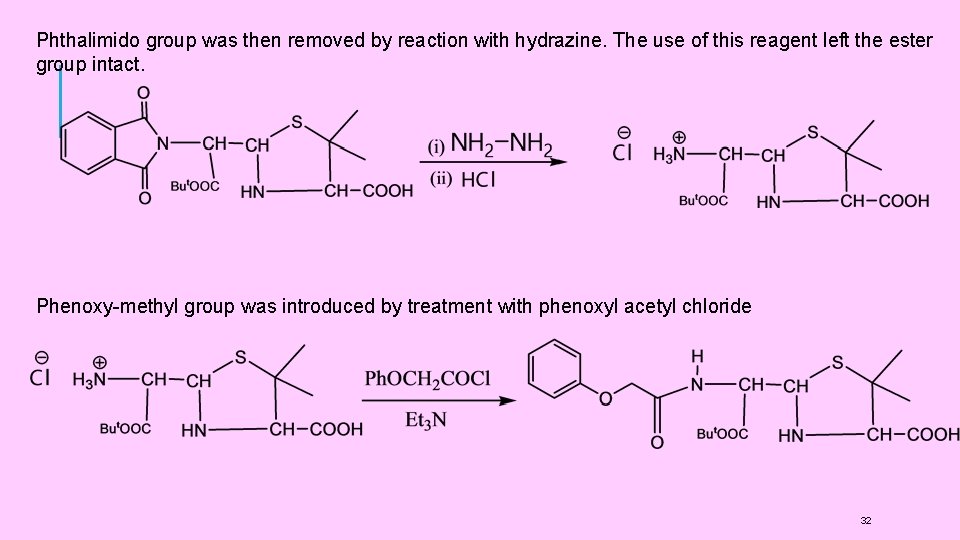
Phthalimido group was then removed by reaction with hydrazine. The use of this reagent left the ester group intact. Phenoxy-methyl group was introduced by treatment with phenoxyl acetyl chloride 32
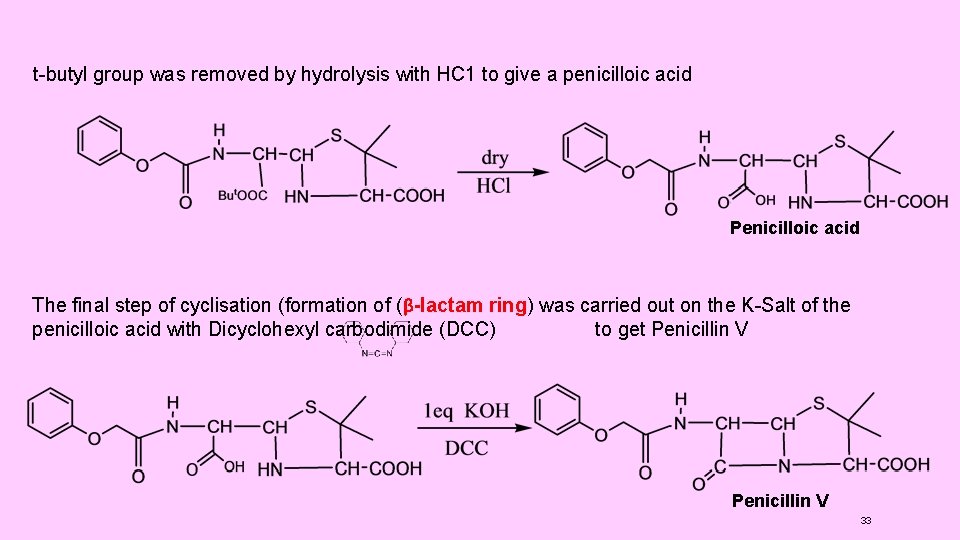
t-butyl group was removed by hydrolysis with HC 1 to give a penicilloic acid Penicilloic acid The final step of cyclisation (formation of (β-lactam ring) was carried out on the K-Salt of the penicilloic acid with Dicyclohexyl carbodimide (DCC) to get Penicillin V 33
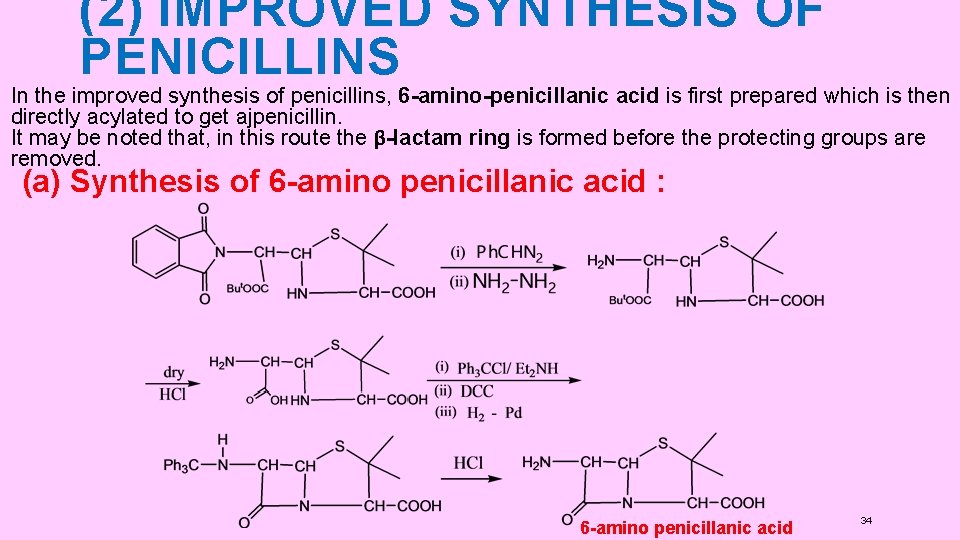
(2) IMPROVED SYNTHESIS OF PENICILLINS In the improved synthesis of penicillins, 6 -amino-penicillanic acid is first prepared which is then directly acylated to get ajpenicillin. It may be noted that, in this route the β-lactam ring is formed before the protecting groups are removed. (a) Synthesis of 6 -amino penicillanic acid : 6 -amino penicillanic acid 34
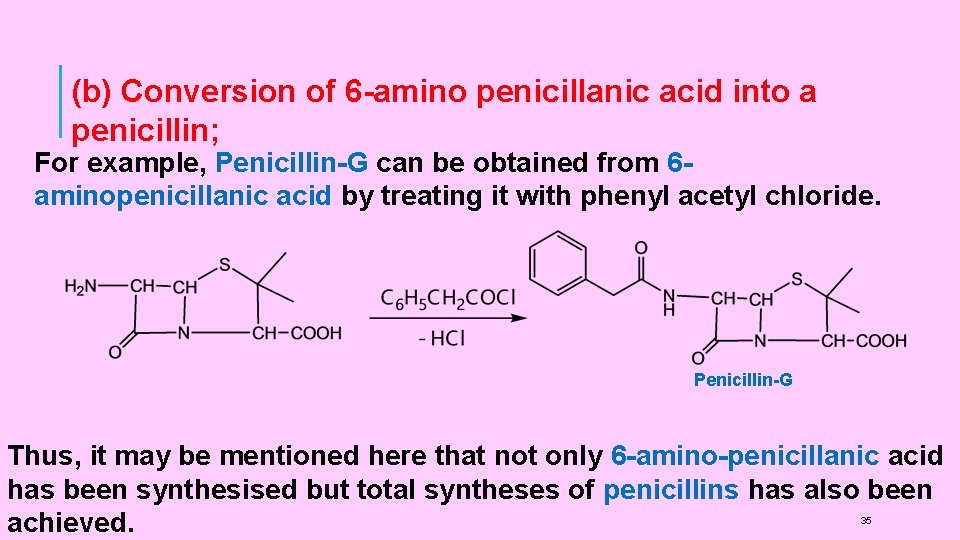
(b) Conversion of 6 -amino penicillanic acid into a penicillin; For example, Penicillin-G can be obtained from 6 aminopenicillanic acid by treating it with phenyl acetyl chloride. Penicillin-G Thus, it may be mentioned here that not only 6 -amino-penicillanic acid has been synthesised but total syntheses of penicillins has also been achieved. 35

It is also known as Anaesthesin BENZOCAINE ( ETHYL- p AMINO-BENZOATE ) Preparation: It is conveniently prepared from p - nitrobenzoic acid by the following (i)methods. p - nitrobenzoic acid is first reduced with tin and hydrochloric acid to p - amino benzoic acid. The latter is then esterified with ethyl alcohol in the presence of hydrogen chloride. Benzocaine 36
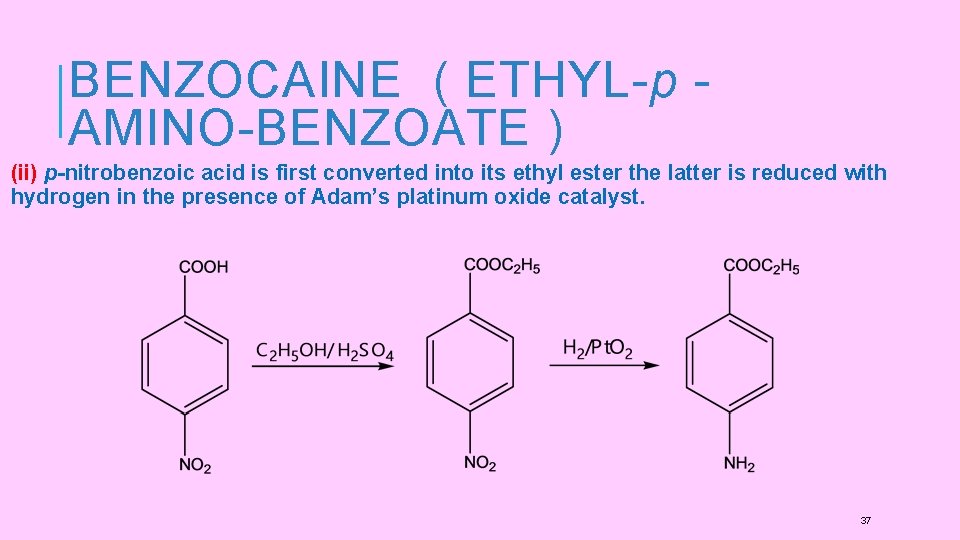
BENZOCAINE ( ETHYL- p AMINO-BENZOATE ) (ii) p-nitrobenzoic acid is first converted into its ethyl ester the latter is reduced with hydrogen in the presence of Adam’s platinum oxide catalyst. 37

CHLORPROPAMIDE (i) Preparation: Synthesis: The synthesis of chlorpropamide has been carried out by reacting p-chlorosulfonamide with n-propylisocyanate in the presence of mild base (Scheme 5 a). Side effects: Renal impairment and hypertension other than hypoglycemia is reported. Metabolism: The half life period of chlorpropamide is relatively longer because of p-chloro substituent which protects the p - position from metabolic oxidation. 20% of a dose is excreted unchanged and remaining 80% is metabolized by omega and omega-1 type oxidation (Scheme 5 b). 38
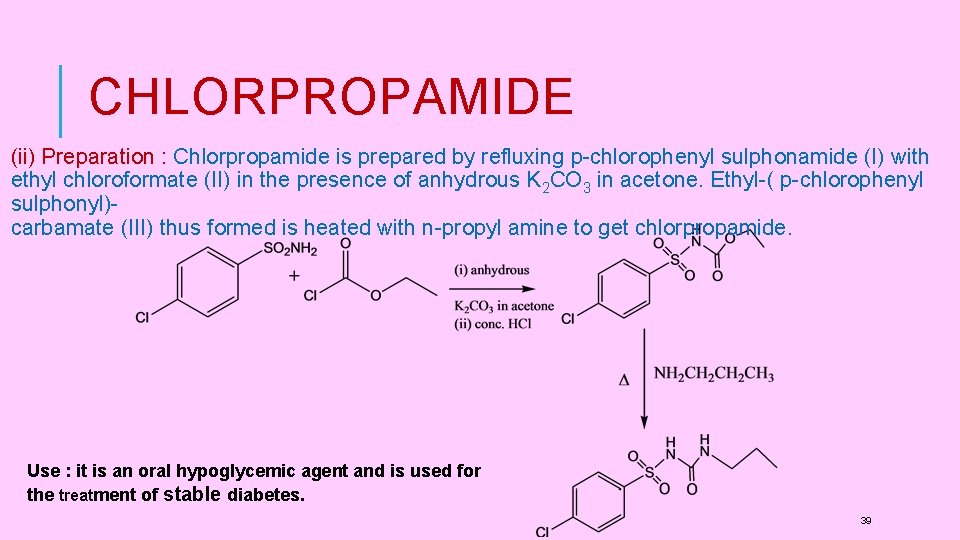
CHLORPROPAMIDE (ii) Preparation : Chlorpropamide is prepared by refluxing p-chlorophenyl sulphonamide (I) with ethyl chloroformate (II) in the presence of anhydrous K 2 CO 3 in acetone. Ethyl-( p-chlorophenyl sulphonyl)carbamate (III) thus formed is heated with n-propyl amine to get chlorpropamide. Use : it is an oral hypoglycemic agent and is used for the treatment of stable diabetes. 39

THANK YOU 40
- Slides: 40