Chemistry Topic Chemical Bonding Chemical Bonding Introduction F

Chemistry Topic: Chemical Bonding
Chemical Bonding

Introduction F What will happen when two magnets are put together? Why? F MAGNETIC FORCE causes the 2 magnets to stick together.

Bonding F ELECTROSTATIC FORCE causes 2 atoms to join together. F The forces between 2 atoms are called BONDING.
Types of Bonding

Ionic Bond F What is the chemical formula of sodium chloride? F Na. Cl F Is Na. Cl neutral or a charge carrier? F Neutral

Ionic Bond (cont’d) F What type of particles are present in Na. Cl? F Ions F Are they neutral or charged? F Charged

Ionic Bond (cont’d) F Which F F is positively / negatively charged? Sodium ion, Na+, — positive – Chloride ion, Cl , — negative

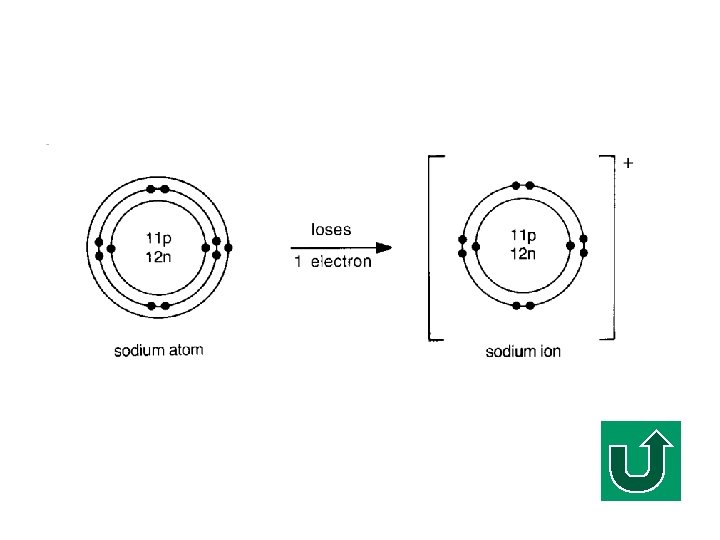
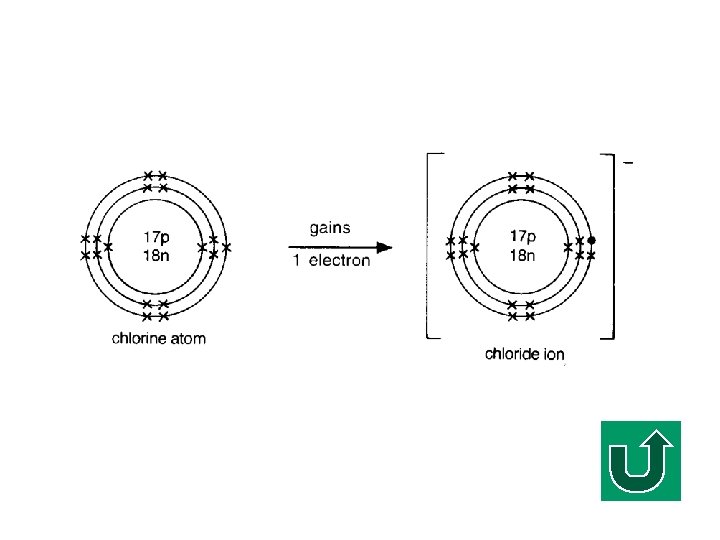
Definition of an Ionic Bond F An IONIC BOND is the strong nondirectional electrostatic force of attraction between oppositely charged ions. FA pure ionic bond is formed by the COMPLETE transfer of one or more electrons from one atom to another.

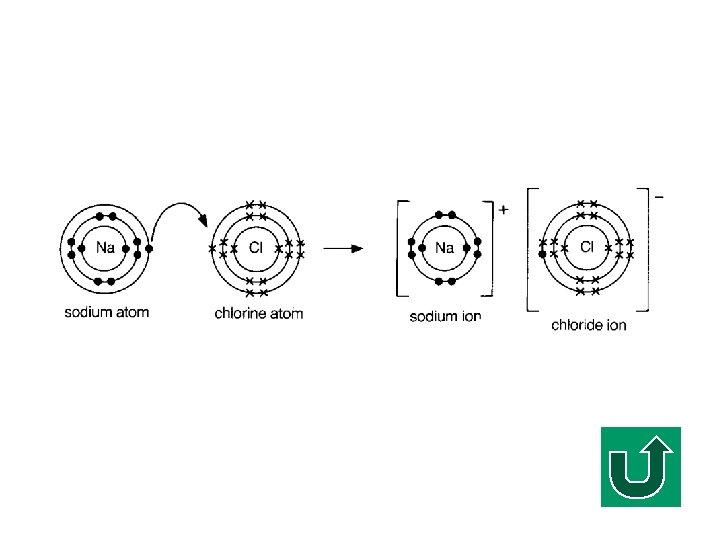
Electron Transfer in Na. Cl F Na is a metal, Cl is a non-metal. F Generally, the electrons are transferred from a metal to a non-metal, i. e. from Na to Cl. F Why?

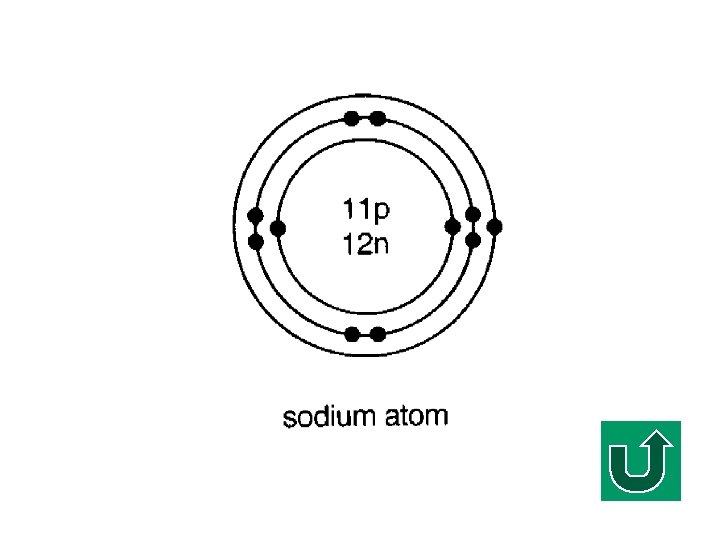
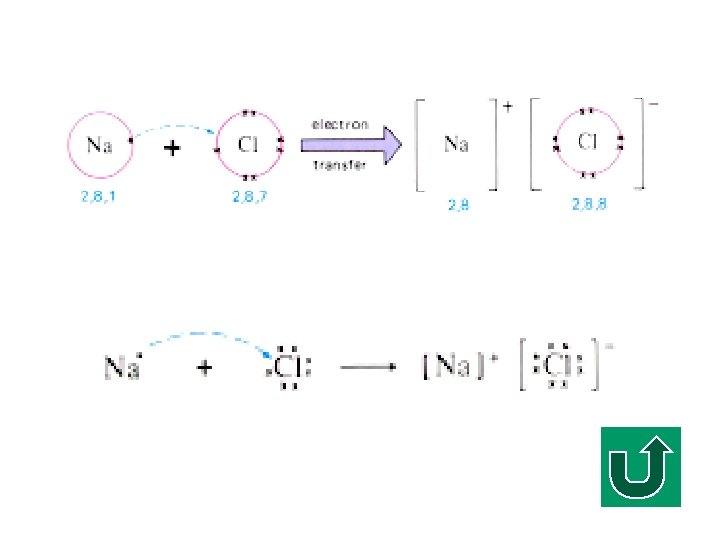
Electronic Configuration of Na F What is the electronic configuration of Na? F (2, 8, 1) F It tends to lose ONE electron to make it stable.
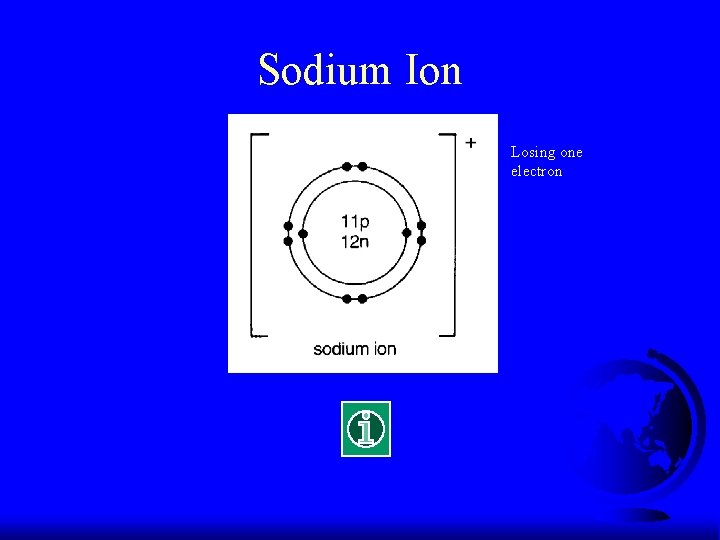
Sodium Ion Losing one electron

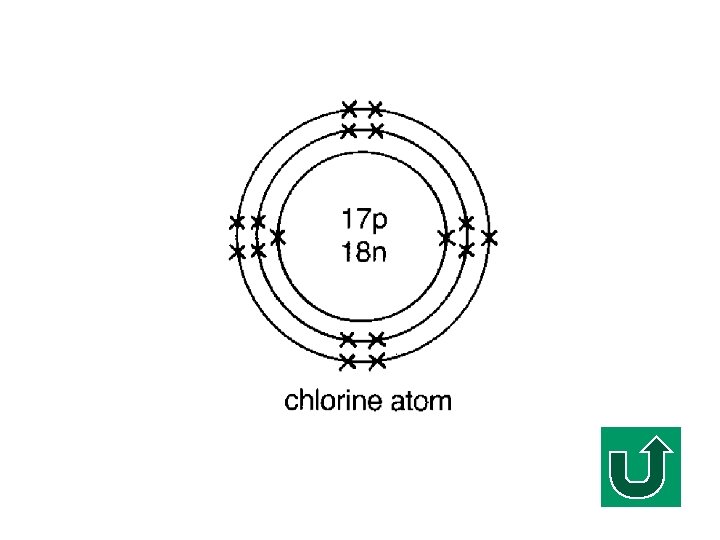
Electronic Configuration of Cl F What is the electronic configuration of Cl? F (2, 8, 7) F It tends to gain ONE electron to make it stable.
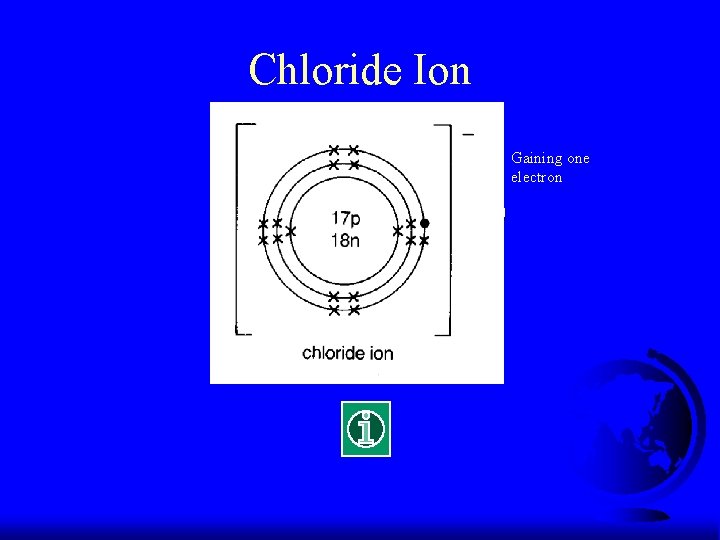
Chloride Ion Gaining one electron

Electron Diagram of Na. Cl F The electron is transferred from sodium atom to chlorine atom. F It is also called an electron ‘dot/cross’ diagram (or simply electron diagram) F Simplified notation

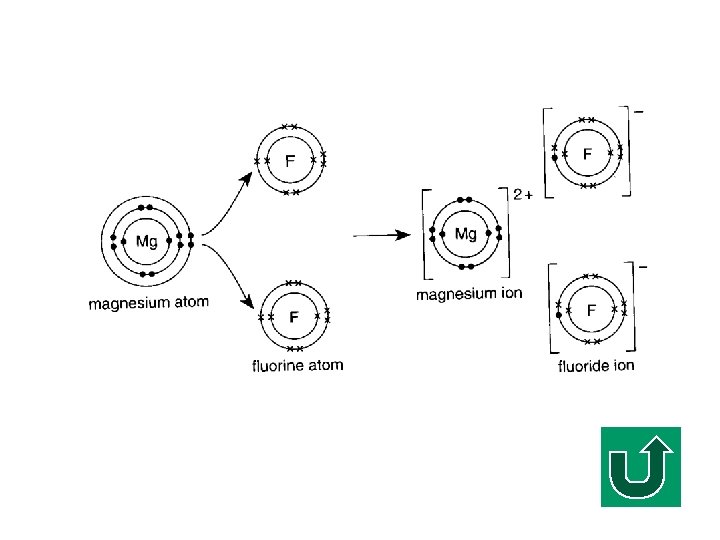
Another Example Let’s consider another example, magnesium fluoride. F What is the chemical formula of magnesium fluoride? F Mg. F 2

Another Example (cont’d) F Mg FF loses 2 electrons. gains 1 electron.

Another Example (cont’d) F How many atoms of Mg and F are involved in this electron transfer process? F 1 Mg atom, 2 F atoms F So, totally 3 atoms are involved in this process.

Magnesium Fluoride Mg. F 2 Prediction the electron diagram of Mg. F 2

Homework F Draw the electronic diagram of the following compounds. – Potassium chloride – Magnesium bromide – Aluminum oxide

- Slides: 28