Chemistry Third Edition Julia Burdge Lecture Power Points

Chemistry Third Edition Julia Burdge Lecture Power. Points Chapter 21 Environmental Chemistry Copyright © 2012, The Mc. Graw-Hill Compaies, Inc. Permission required for reproduction or display.

CHAPTER 21. 1 21. 2 21. 3 21. 4 21. 5 21. 6 21. 7 21. 8 21 Environmental Chemistry Earth’s Atmosphere Phenomena in the Outer Layers of the Atmosphere Depletion of Ozone in the Stratosphere Volcanoes The Greenhouse Effect Acid Rain Photochemical Smog Indoor Pollution 2

21. 1 Earth’s Atmosphere Topics Earth’s Atmosphere 3

21. 1 Earth’s Atmosphere Earth is unique in having an atmosphere that is chemically active and rich in oxygen. Primitive organisms used energy from the sun to break down CO 2 to obtain carbon, which they incorporated in their own cells in a process called photosynthesis. The major by-product of photosynthesis, is oxygen. Another important source of oxygen is the photodecomposition of water vapor by UV light. 4
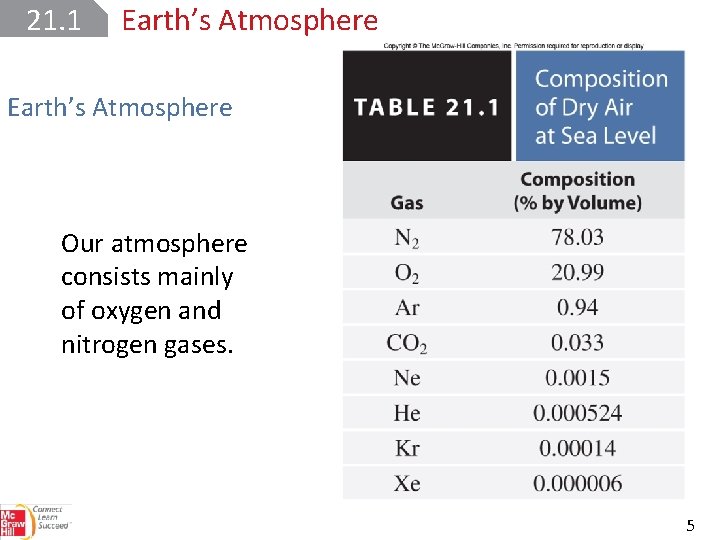
21. 1 Earth’s Atmosphere Our atmosphere consists mainly of oxygen and nitrogen gases. 5
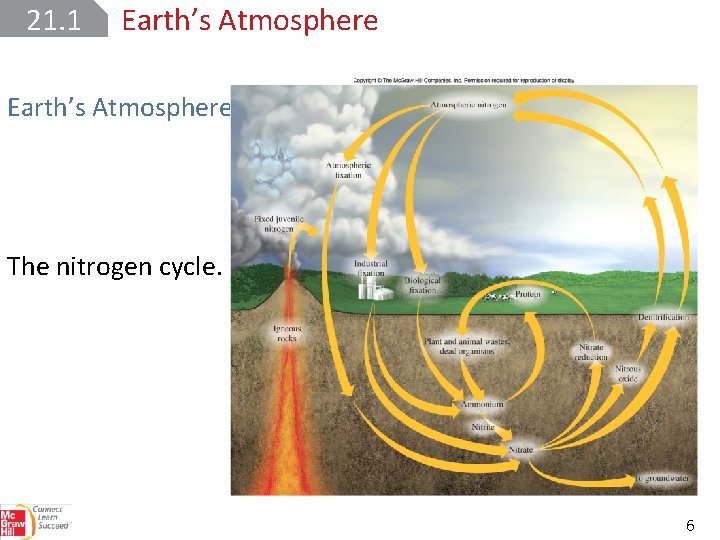
21. 1 Earth’s Atmosphere The nitrogen cycle. 6

21. 1 Earth’s Atmosphere The nitrogen cycle: Molecular nitrogen is a very stable molecule. Through nitrogen fixation ‒ the conversion of molecular nitrogen into nitrogen compounds ‒ atmospheric nitrogen gas is converted into nitrates and other compounds. 7

21. 1 Earth’s Atmosphere The nitrogen cycle: Lightening also produces nitrates from nitrogen gas 8

21. 1 Earth’s Atmosphere The nitrogen cycle: Nitric acid is converted to nitrate salts in the soil. Animals use nutrients from plants to make proteins and other essential biomolecules. Denitrification reverses nitrogen fixation to complete the cycle. 9
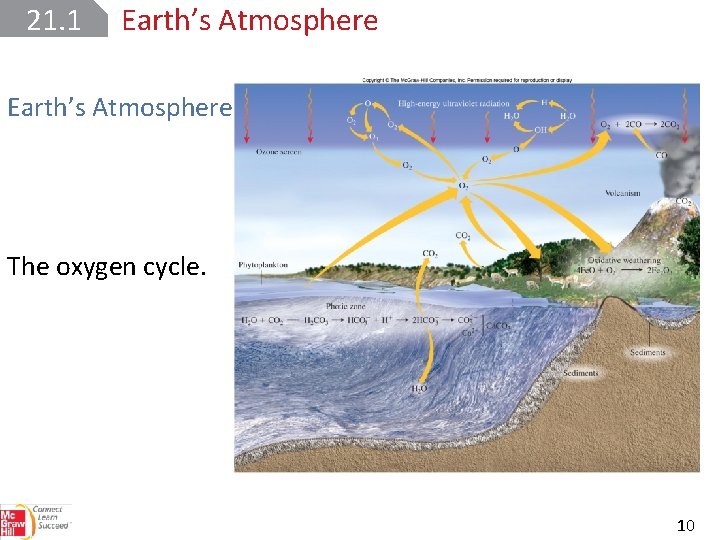
21. 1 Earth’s Atmosphere The oxygen cycle. 10

21. 1 Earth’s Atmosphere The oxygen cycle: Atmospheric oxygen is removed through respiration and various industrial processes, which produce carbon dioxide. Photosynthesis is the major mechanism by which molecular oxygen is regenerated from carbon dioxide and water. 11
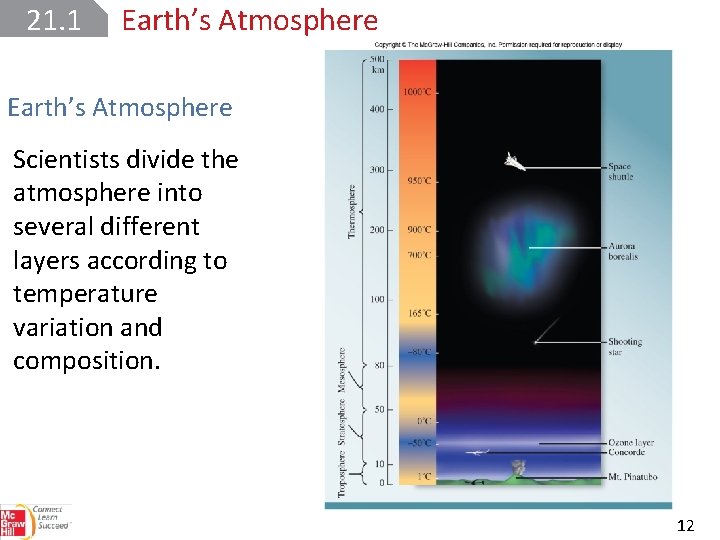
21. 1 Earth’s Atmosphere Scientists divide the atmosphere into several different layers according to temperature variation and composition. 12
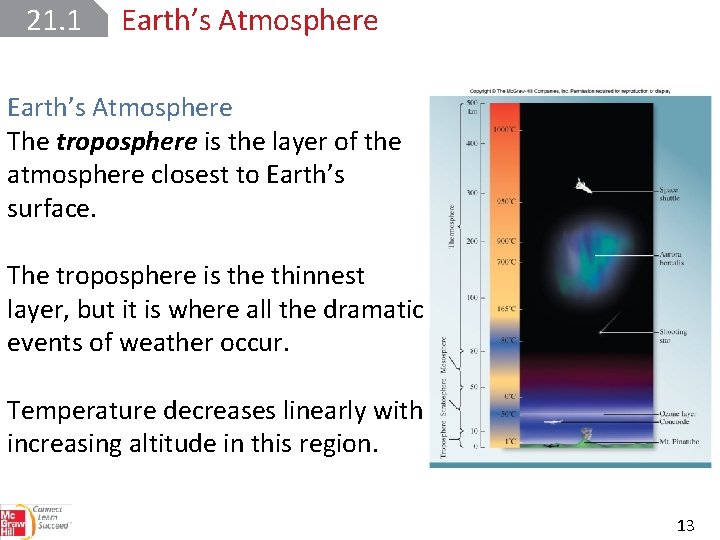
21. 1 Earth’s Atmosphere The troposphere is the layer of the atmosphere closest to Earth’s surface. The troposphere is the thinnest layer, but it is where all the dramatic events of weather occur. Temperature decreases linearly with increasing altitude in this region. 13
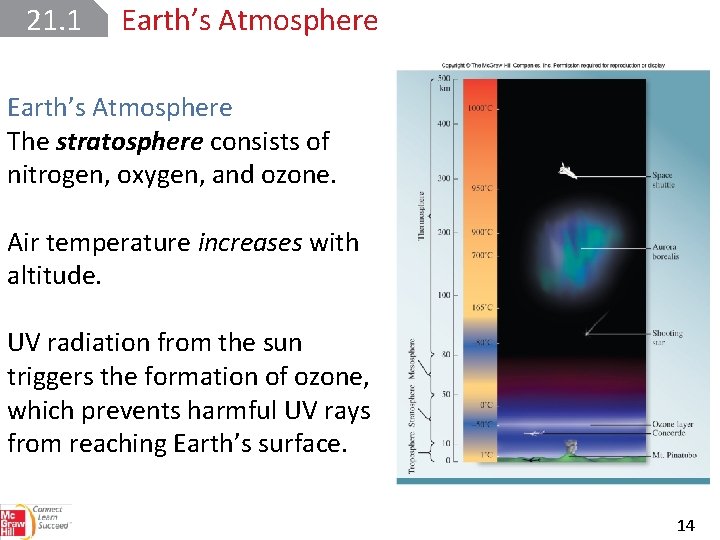
21. 1 Earth’s Atmosphere The stratosphere consists of nitrogen, oxygen, and ozone. Air temperature increases with altitude. UV radiation from the sun triggers the formation of ozone, which prevents harmful UV rays from reaching Earth’s surface. 14

21. 1 Earth’s Atmosphere In the mesosphere, the concentration of ozone and other gases is low. Temperature decreases with increasing altitude. 15

21. 1 Earth’s Atmosphere The thermosphere, or ionosphere, is the uppermost layer of the atmosphere. The increase in temperature in is the result of the bombardment of molecular oxygen and nitrogen and atomic species by energetic particles, from the sun. 16

21. 2 Phenomena in the Outer Layers of the Atmosphere Topics Aurora Borealis and Aurora Australis The Mystery Glow of Space Shuttles 17

21. 2 Phenomena in the Outer Layers of the Atmosphere Aurora Borealis and Aurora Australis Violent eruptions on the surface of the sun, called solar flares, result in the ejection of myriad electrons and protons into space, where provide us with spectacular celestial light shows known as auroras. 18

21. 2 Phenomena in the Outer Layers of the Atmosphere Aurora Borealis and Aurora Australis Electrons and protons collide with the molecules and atoms in Earth’s upper atmosphere, causing them to become ionized and electronically excited. The excited molecules and ions return to the ground state with the emission of light. An excited oxygen atom emits photons at wavelengths of 558 nm (green) and between 630 and 636 nm (red): 19

21. 2 Phenomena in the Outer Layers of the Atmosphere Aurora Borealis and Aurora Australis The blue and violet colors result from the transition in the ionized nitrogen molecule: Aurora borealis is the name given to this phenomenon in the Northern Hemisphere. In the Southern Hemisphere, it is called aurora australis. 20

SAMPLE PROBLEM 21. 1 The bond enthalpy of O 2 is 498. 7 k. J/mol. Calculate the maximum wavelength (in nm) of a photon that can cause the dissociation of an O 2 molecule. 21

SAMPLE PROBLEM 21. 1 Setup The conversion steps are: Solution The energy required to dissociate one O 2 molecule: 22

SAMPLE PROBLEM 21. 1 The energy of the photon is given by E = hν Calculate the wavelength of the photon, given by λ = c/ν as follows: Any photon with a wavelength of 240 nm or shorter can dissociate an O 2 molecule. 23

21. 2 Phenomena in the Outer Layers of the Atmosphere The Mystery Glow of Space Shuttles Astronauts noticed an eerie orange glow on the outside surface of their spacecraft 300 km above Earth. 24

21. 2 Phenomena in the Outer Layers of the Atmosphere The Mystery Glow of Space Shuttles It is believed that oxygen atoms interact with NO adsorbed on the shuttle’s surface to form electronically excited NO 2: As the NO 2* leaves the shell of the spacecraft, it emits photons at a wavelength of 680 nm (orange): 25

21. 3 Depletion of Ozone in the Stratosphere Topics Depletion of Ozone in the Stratosphere Polar Ozone Holes 26

21. 3 Depletion of Ozone in the Stratosphere The formation of ozone begins with the photodissociation of oxygen molecules by solar radiation The highly reactive O atoms combine with oxygen molecules to form ozone: where M is some inert substance such as N 2. 27

21. 3 Depletion of Ozone in the Stratosphere Ozone itself absorbs UV light between 200 and 300 nm: The process continues when O and O 2 recombine to form O 3. 28

21. 3 Depletion of Ozone in the Stratosphere Ozone acts as our protective shield against UV radiation. The formation and destruction of ozone is a dynamic equilibrium that maintains a constant concentration of ozone in the stratosphere. 29
21. 3 Depletion of Ozone in the Stratosphere Since the mid-1970 s scientists have been concerned about the harmful effects of CFCs on the ozone layer. 30

21. 3 Depletion of Ozone in the Stratosphere CFCs slowly diffuse unchanged to the stratosphere, where UV radiation causes them to decompose: The reactive chlorine atoms then undergo the following reactions: 31

21. 3 Depletion of Ozone in the Stratosphere The overall result is the net removal of an O 3 molecule from the stratosphere: The Cl atom plays the role of a catalyst. One Cl atom can destroy up to 100, 000 O 3 molecules! 32
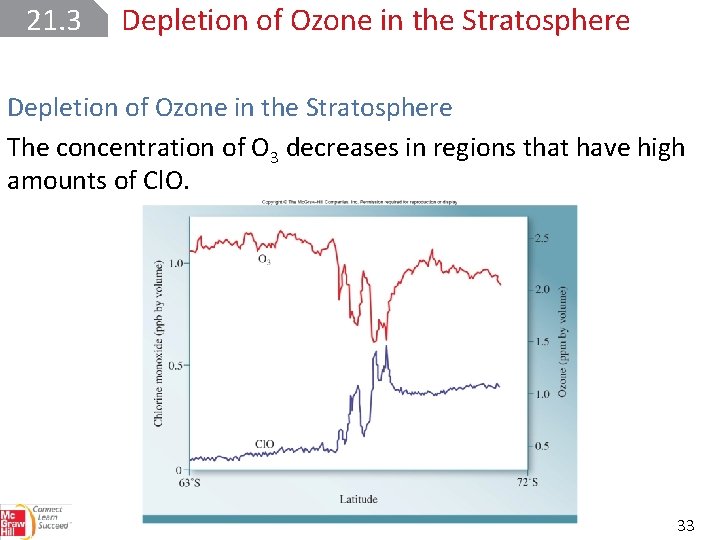
21. 3 Depletion of Ozone in the Stratosphere The concentration of O 3 decreases in regions that have high amounts of Cl. O. 33

21. 3 Depletion of Ozone in the Stratosphere Another group of compounds that can destroy stratospheric ozone are the nitrogen oxides, generally denoted as NOx. NO is the catalyst and NO 2 is the intermediate. 34
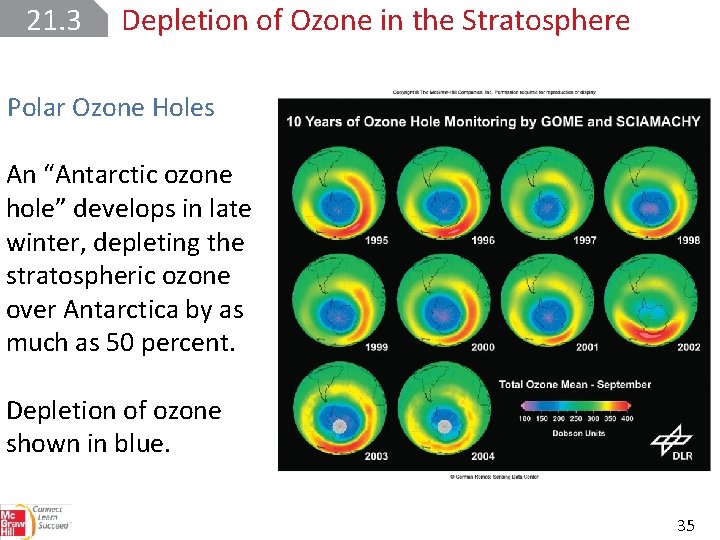
21. 3 Depletion of Ozone in the Stratosphere Polar Ozone Holes An “Antarctic ozone hole” develops in late winter, depleting the stratospheric ozone over Antarctica by as much as 50 percent. Depletion of ozone shown in blue. 35

21. 3 Depletion of Ozone in the Stratosphere Polar Ozone Holes A stream of air known as the “polar vortex” circles Antarctica in winter. Air trapped within this vortex becomes extremely cold during the polar night. This leads to the formation of ice particles known as polar stratospheric clouds (PSCs). 36

21. 3 Depletion of Ozone in the Stratosphere Polar Ozone Holes PSCs provide a surface for reactions converting HCl and Cl. NO 2 to more reactive chlorine molecules: By early spring, the sunlight splits molecular chlorine into chlorine atoms, which then attack ozone. The situation is not as severe in the warmer Arctic region, where the vortex does not persist quite as long. 37

21. 3 Depletion of Ozone in the Stratosphere Polar Ozone Holes Nations throughout the world have acknowledged the need to drastically curtail or totally stop the production of CFCs. An intense effort is under way to find CFC substitutes that are not harmful to the ozone layer. 38

21. 4 Volcanoes Topics Volcanoes 39

21. 4 Volcanoes Volcanic eruptions are instrumental in forming large parts of Earth’s crust. Molten rock, called magma, in the upper mantle, rises to the surface and generates volcanic eruptions. 40

21. 4 Volcanoes An active volcano emits gases, liquids, and solids. The gases spewed into the atmosphere include primarily N 2, CO 2, HCl, HF, H 2 S, and water vapor. Volcanoes are the source of about two-thirds of the sulfur in the air. 41
21. 4 Volcanoes At high temperatures, the hydrogen sulfide gas given off by a volcano is oxidized by air: Some of the SO 2 is reduced by more H 2 S from the volcano to elemental sulfur and water: 42

21. 4 Volcanoes The rest of the SO 2 is released into the atmosphere, where it reacts with water to form acid rain. Aerosols given off by volcanic eruptions • destroy ozone • affect climate, creating a localized cooling effect. 43

21. 5 The Greenhouse Effect Topics The Greenhouse Effect 44

21. 5 The Greenhouse Effect The greenhouse effect describes the trapping of heat near Earth’s surface by gases in the atmosphere, particularly carbon dioxide. The transfer of CO 2 to and from the atmosphere is an essential part of the carbon cycle. CO 2 is produced when any form of carbon or a carboncontaining compound is burned in an excess of oxygen. 45
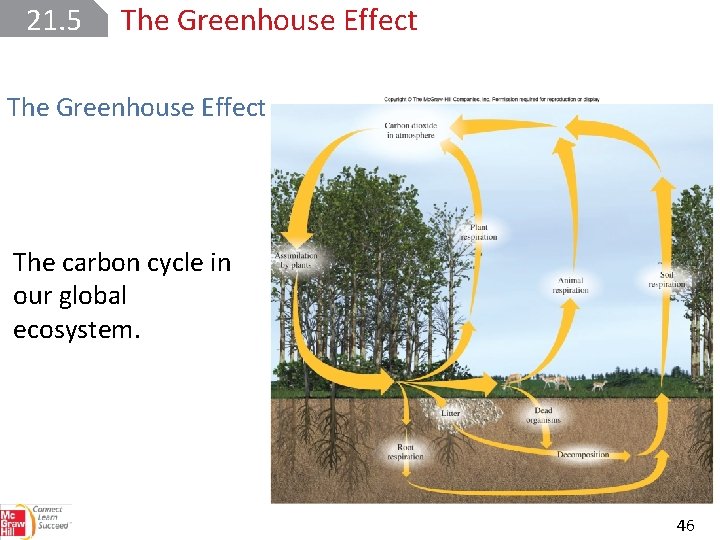
21. 5 The Greenhouse Effect The carbon cycle in our global ecosystem. 46

21. 5 The Greenhouse Effect Sources of CO 2: Carbonates can give off CO 2 when heated or treated with acid: Carbon dioxide is also a by-product of the fermentation of sugar: 47

21. 5 The Greenhouse Effect Sources of CO 2: Animals respire and release CO 2 as an end product of metabolism: Another major source of CO 2 is volcanic activity. 48

21. 5 The Greenhouse Effect Removal of CO 2: Carbon dioxide is removed from the atmosphere by photosynthetic plants and certain microorganisms: After plants and animals die, the carbon in their tissues is oxidized to CO 2 and returns to the atmosphere. There is a dynamic equilibrium between atmospheric CO 2 and carbonates in the oceans and lakes. 49

21. 5 The Greenhouse Effect Much of the solar radiant energy received by Earth is in the visible region of the spectrum. Thermal radiation emitted by Earth’s surface is characterized by wavelengths in the IR region. The outgoing IR radiation can be absorbed by water and carbon dioxide, but not by nitrogen and oxygen. 50
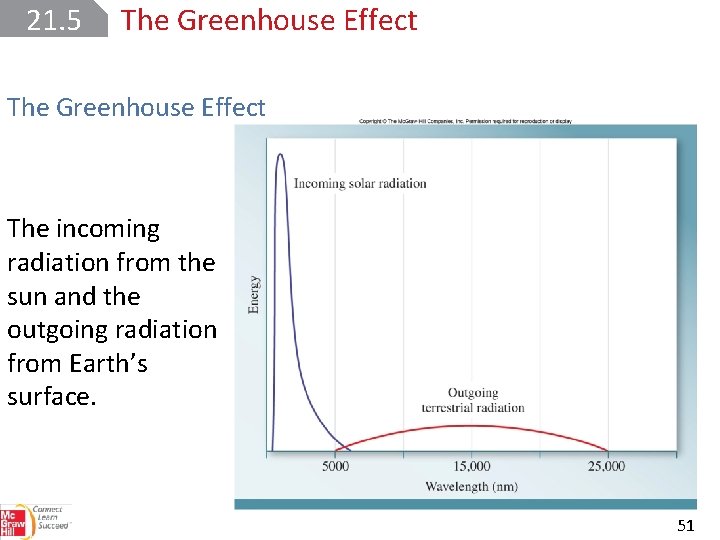
21. 5 The Greenhouse Effect The incoming radiation from the sun and the outgoing radiation from Earth’s surface. 51

21. 5 The Greenhouse Effect All molecules vibrate. The energy associated with molecular vibration is quantized. 52

21. 5 The Greenhouse Effect To absorb a photon in the IR region, the dipole moment of the molecule must change during the course of a vibration. If the molecule is homonuclear, there can be no change in the dipole moment; the molecule has a zero dipole moment. Such molecules are IR-inactive because they cannot absorb IR radiation. 53

21. 5 The Greenhouse Effect All heteronuclear diatomic molecules are IR-active; they can absorb IR radiation because their dipole moments change. A polyatomic molecule can vibrate in more than one way. H 2 O is IR active Water, for example, can vibrate in three different ways. 54
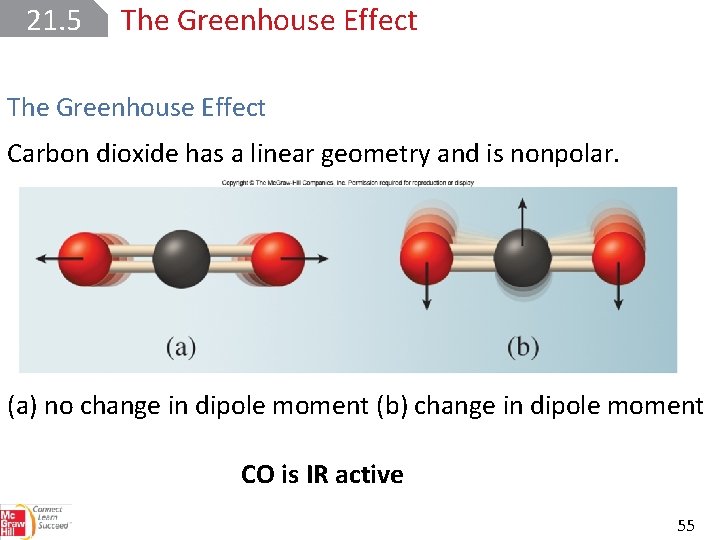
21. 5 The Greenhouse Effect Carbon dioxide has a linear geometry and is nonpolar. (a) no change in dipole moment (b) change in dipole moment CO is IR active 55

21. 5 The Greenhouse Effect Receiving a photon in the IR region, a molecule of H 2 O or CO 2 is promoted to a higher vibrational energy level: These excited molecules lose their excess energy either by collision with other molecules or by spontaneous emission of radiation. 56
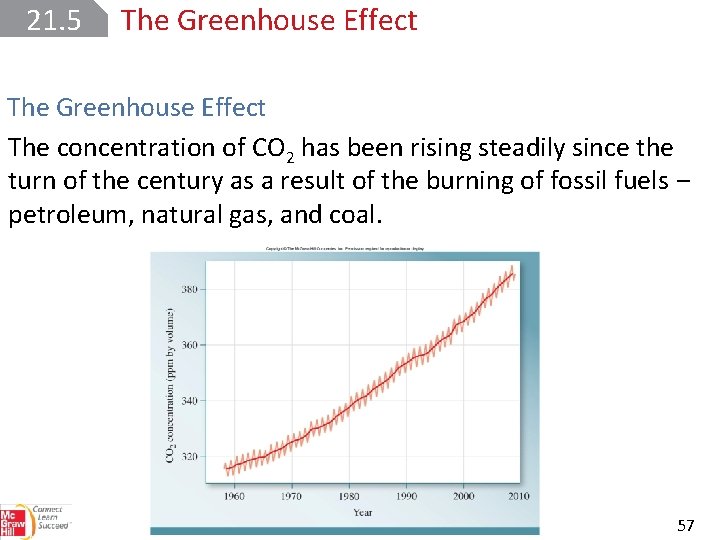
21. 5 The Greenhouse Effect The concentration of CO 2 has been rising steadily since the turn of the century as a result of the burning of fossil fuels ‒ petroleum, natural gas, and coal. 57
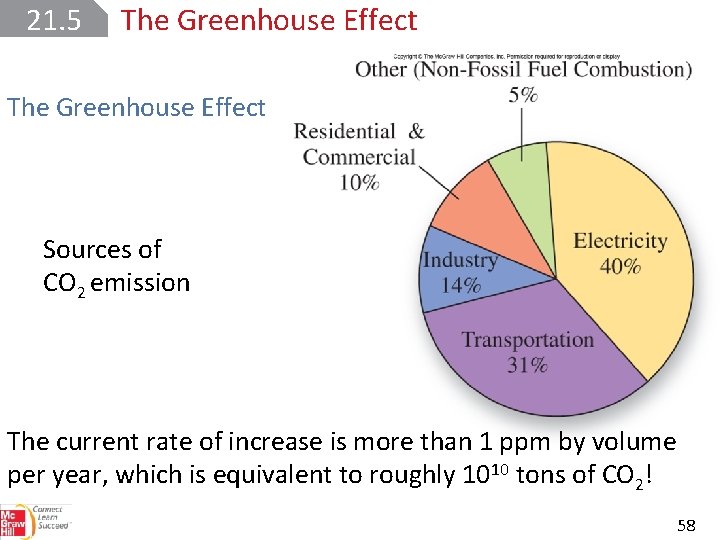
21. 5 The Greenhouse Effect Sources of CO 2 emission The current rate of increase is more than 1 ppm by volume per year, which is equivalent to roughly 1010 tons of CO 2! 58
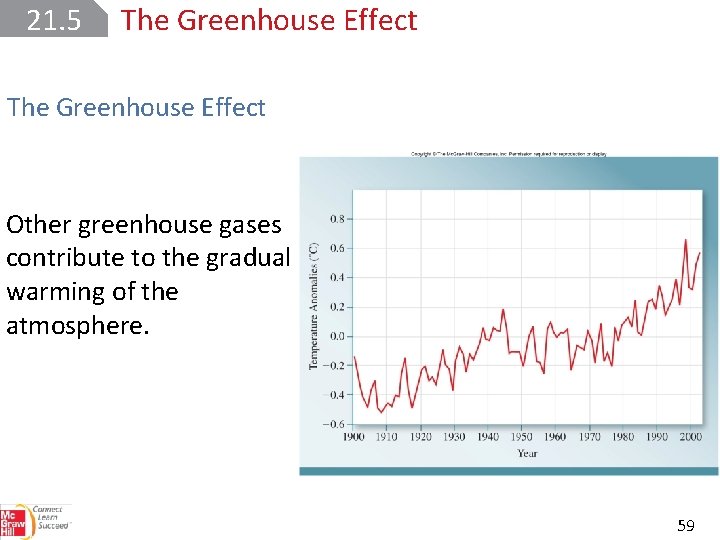
21. 5 The Greenhouse Effect Other greenhouse gases contribute to the gradual warming of the atmosphere. 59
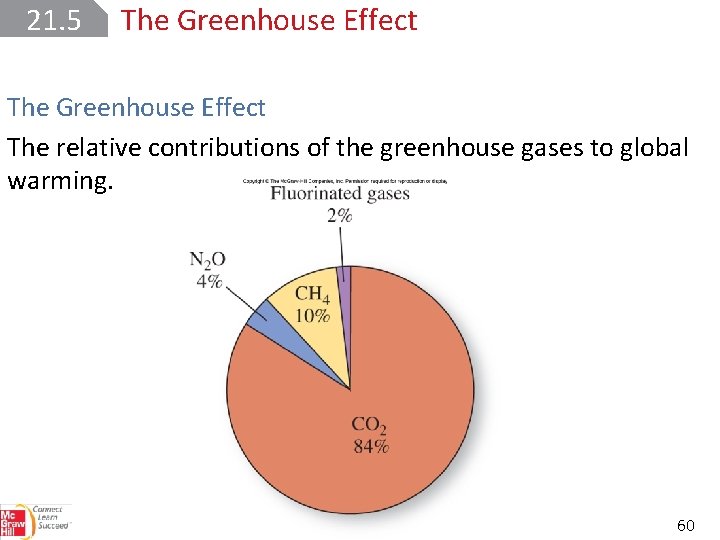
21. 5 The Greenhouse Effect The relative contributions of the greenhouse gases to global warming. 60

21. 5 The Greenhouse Effect To combat the greenhouse effect, we must lower carbon dioxide emissions. This can be done by • improving energy efficiency in automobiles households • developing nonfossil fuel energy sources • phasing out CFCs • recovery of methane gas generated at landfills • reduction of natural gas leakages • preservation forests 61

SAMPLE PROBLEM 21. 2 Which of the following qualify as greenhouse gases: CO, NO 2, Cl 2, H 2, Ne? 62

SAMPLE PROBLEM 21. 2 Strategy The molecule must possess a dipole moment or some of its vibrational motions must generate a temporary dipole moment. Setup The necessary conditions rule out homonuclear diatomic molecules and atomic species. 63

SAMPLE PROBLEM 21. 2 Solution Only CO, NO, and NO 2, which are all polar molecules, qualify as greenhouse gases. Both Cl 2 and H 2 are homonuclear diatomic molecules, and Ne is atomic. These three species are all IR-inactive. 64
21. 6 Acid Rain Topics Acid Rain 65

21. 6 Acid Rain Every year acid rain causes hundreds of millions of dollars’ worth of damage to stone buildings and statues throughout the world. The term stone leprosy is used by some environmental chemists to describe the corrosion of stone by acid rain. Acid rain is also toxic to vegetation and aquatic life. 66

21. 6 Acid Rain 67
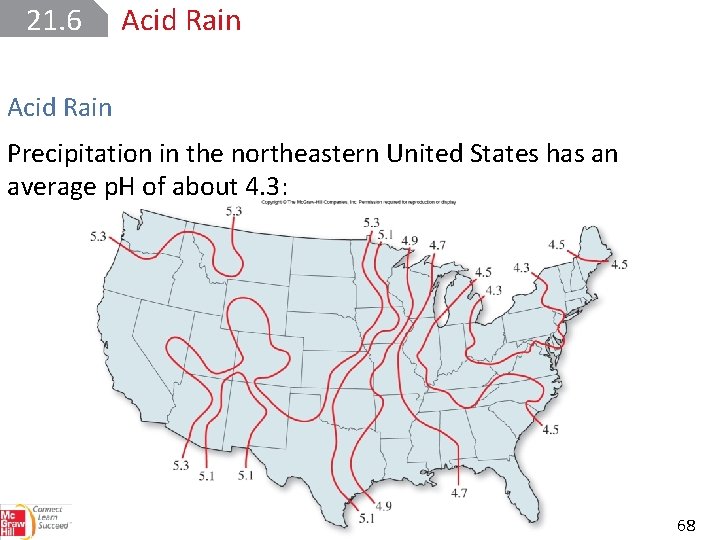
21. 6 Acid Rain Precipitation in the northeastern United States has an average p. H of about 4. 3: 68

21. 6 Acid Rain Sulfur dioxide (SO 2) and, to a lesser extent, nitrogen oxides from auto emissions are believed to be responsible for the high acidity of rainwater. Acidic oxides, such as SO 2, react with water to give the corresponding acids. There are several sources of atmospheric SO 2, including volcanic eruptions. 69

21. 6 Acid Rain Many metals exist combined with sulfur in nature. Extracting the metals often entails smelting, or roasting, the ores—that is, heating the metal sulfide in air to form the metal oxide and SO 2. For example, The metal oxide can be reduced more easily than the sulfide to the free metal. 70

21. 6 Acid Rain The burning of fossil fuels in industry, in power plants, and in homes accounts for most of the SO 2 emitted to the atmosphere. 50 million to 60 million tons of SO 2 are released into the atmosphere each year! Almost all oxidized to H 2 SO 4 in the form of aerosol, which ends up in acid rain. 71

21. 6 Acid Rain The mechanism for the conversion of SO 2 to H 2 SO 4 is quite complex and not fully understood. The reaction is believed to be initiated by the hydroxyl radical (OH): The HOSO 2 radical is further oxidized to SO 3: 72

21. 6 Acid Rain The sulfur trioxide formed would then rapidly react with water to form sulfuric acid: Eventually, the acid rain can corrode limestone and marble. 73

21. 6 Acid Rain There are two ways to minimize the effects of SO 2 pollution: 1. remove sulfur from fossil fuels before combustion (difficult) 2. remove SO 2 as it is formed (limestone) 74

21. 6 Acid Rain Limestone is injected into the power plant boiler or furnace along with the coal. 75

21. 6 Acid Rain At high temperatures, the following decomposition occurs: The quicklime reacts with SO 2 to form calcium sulfite and some calcium sulfate: 76

21. 6 Acid Rain Quicklime is also added to lakes and soils in a process called liming to reduce their acidity. 77

21. 7 Photochemical Smog Topics Photochemical Smog 78

21. 7 Photochemical Smog The word smog was coined to describe the combination of smoke and fog that shrouded London during the 1950 s. The primary cause of this noxious cloud was sulfur dioxide. Photochemical smog is formed by the reactions of automobile exhaust in the presence of sunlight. 79

21. 7 Photochemical Smog Automobile exhaust consists mainly of NO, CO, and various unburned hydrocarbons. These gases are called primary pollutants because they set in motion a series of photochemical reactions that produce secondary pollutants. It is the secondary pollutants—chiefly NO 2 and O 3—that are responsible for the buildup of smog. 80

21. 7 Photochemical Smog NO is the product of the reaction between atmospheric N 2 and O 2 at high temperatures inside an automobile engine: Nitric oxide is then oxidized to nitrogen dioxide: 81

21. 7 Photochemical Smog Sunlight causes the photochemical decomposition of NO 2 into NO and O: Atomic oxygen is a highly reactive species that can initiate the formation of ozone: where M is some inert substance such as N 2. 82

21. 7 Photochemical Smog Ozone attacks the C=C linkage in rubber: This reaction cause automobile tires to crack. Similar reactions are also damaging to lung tissues and other biological substances. 83
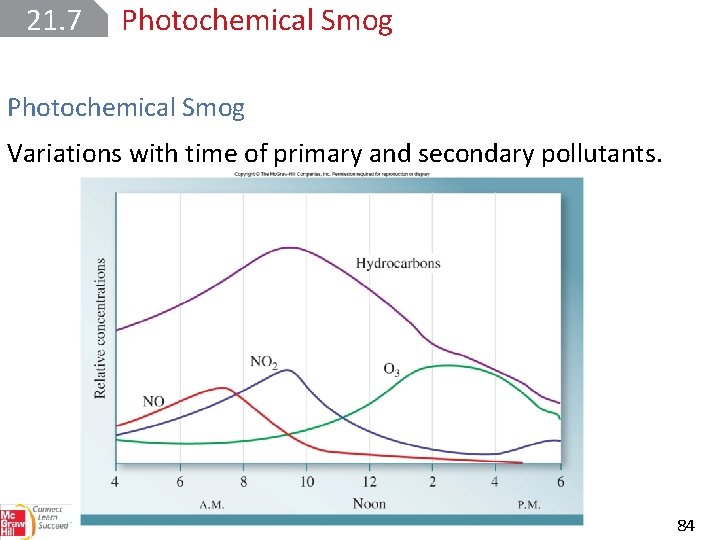
21. 7 Photochemical Smog Variations with time of primary and secondary pollutants. 84

21. 7 Photochemical Smog The oxidation of hydrocarbons produces various organic intermediates, which are less volatile than the hydrocarbons themselves. These substances condense into small droplets of liquid called aerosols. This interaction makes the air look hazy. 85

21. 7 Photochemical Smog Major efforts have been made to reduce the buildup of primary pollutants. Most automobiles now are quipped with catalytic converters designed to oxidize CO and unburned hydrocarbons to CO 2 and H 2 O and to reduce NO and NO 2 to N 2 and O 2. 86

21. 8 Indoor Pollution Topics The Risk from Radon Carbon Dioxide and Carbon Monoxide Formaldehyde 87

21. 8 Indoor Pollution The Risk from Radon is a member of Group 8 A (the noble gases). It is an intermediate product of the radioactive decay of uranium-238. All isotopes of radon are radioactive, but radon-222 is the most hazardous because it has the longest half-life— 3. 8 days. Radon, is generated mostly from the phosphate minerals of uranium. 88
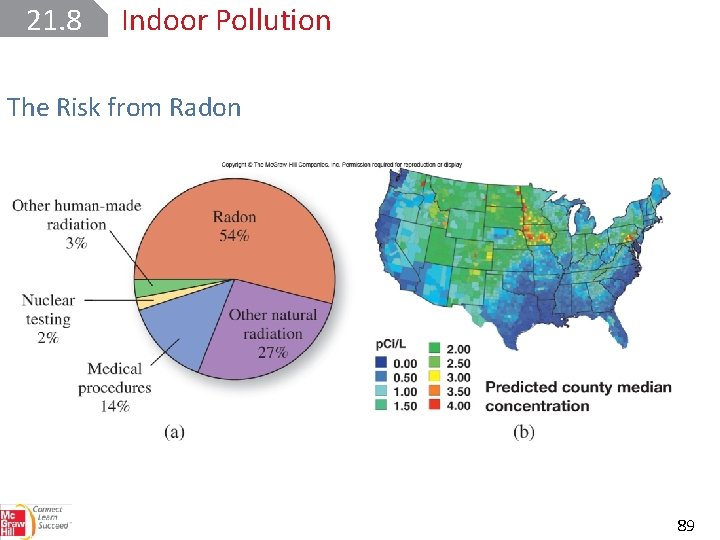
21. 8 Indoor Pollution The Risk from Radon 89

21. 8 Indoor Pollution The Risk from Radon High levels of radon have been detected in homes built on reclaimed land above uranium mill tailing deposits. The colorless, odorless, and tasteless radon gas enters a building through tiny cracks in the basement floor. Radon-222 is an α-emitter. 90

21. 8 Indoor Pollution The Risk from Radon When it decays, it produces radioactive polonium-214 and polonium-218, which can build up to high levels in an enclosed space. These solid radioactive particles are inhaled into the lungs and deposited in the respiratory tract. Over a long period of time, the α particles emitted by polonium and its decay products can cause lung cancer. 91

21. 8 Indoor Pollution The Risk from Radon The first step combat radon pollution indoors is to measure the radon level in the basement with a reliable test kit. If the radon level is unacceptably high, then the house must be regularly ventilated. 92

SAMPLE PROBLEM 21. 3 The half-life of Rn-222 is 3. 8 days. Starting with 1. 0 g of Rn-222, how much will be left after 10 half-lives? 93

SAMPLE PROBLEM 21. 3 Strategy All radioactive decays obey first-order kinetics, making the half-life independent of the initial concentration. Setup Because the question involves an integral number of halflives, we can deduce the amount of Rn-222 remaining. 94

SAMPLE PROBLEM 21. 3 Solution After one half-life, the amount of Rn left is 0. 5 × 1. 0 g, or 0. 5 g. After two half-lives, only 0. 25 g of Rn remains. Generalizing the fraction of the isotope left after n half-lives as (1/2)n, where n = 10, we write 95

21. 8 Indoor Pollution Carbon Dioxide and Carbon Monoxide Both carbon dioxide (CO 2) and carbon monoxide (CO) are products of combustion. Indoor sources of these gases are • gas cooking ranges • woodstoves • space heaters • tobacco smoke • human respiration • exhaust fumes from cars in garages 96

21. 8 Indoor Pollution Carbon Dioxide and Carbon Monoxide Carbon dioxide is not a toxic gas, but it does have an asphyxiating effect. Adequate ventilation is the solution to CO 2 pollution. 97

21. 8 Indoor Pollution Carbon Dioxide and Carbon Monoxide CO is highly poisonous. The toxicity of CO lies in its unusual ability to bind very strongly to hemoglobin, the oxygen carrier in blood. The affinity of hemoglobin for CO is about 200 times greater than it is for O 2. The first-aid response to CO poisoning is to remove the victim immediately to an area with a plentiful oxygen supply. 98

21. 8 Indoor Pollution Formaldehyde (CH 2 O) is a liquid used as a preservative for laboratory specimens. Industrially, formaldehyde resins are used as bonding agents in building and furniture construction materials. Low concentrations of formaldehyde in the air can cause drowsiness, nausea, headaches, and other respiratory ailments. 99

21. 8 Indoor Pollution Formaldehyde Because formaldehyde is a reducing agent, devices have been constructed to remove it by means of a redox reaction. Proper ventilation is the best way to remove formaldehyde. 100
- Slides: 100