Chemistry Third Edition Julia Burdge Lecture Power Points

Chemistry Third Edition Julia Burdge Lecture Power Points Chapter 19 Electrochemistry Copyright © 2012, The Mc. Graw-Hill Compaies, Inc. Permission required for reproduction or display.

• Electrochemistry Applications • • Waste water treatment Corrosion Energy storage Energy production Solar to electrical energy Production of chemicals Sensors

Electrochemistry 19. 1 19. 2 19. 3 19. 4 19. 5 19. 6 19. 7 19. 8 Balancing Redox Reactions Galvanic Cells Standard Reduction Potentials Spontaneity of Redox Reactions Under Standard. State Conditions Spontaneity of Redox Reactions Under Conditions Other Than Standard State Batteries Electrolysis Corrosion 3
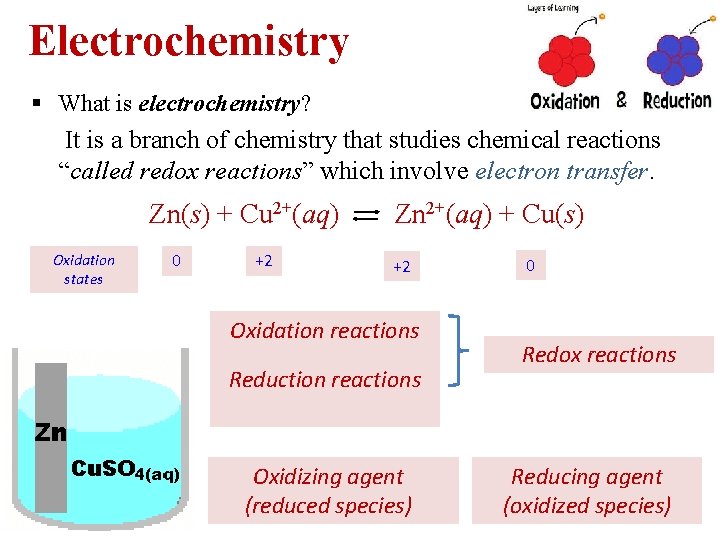
Electrochemistry § What is electrochemistry? It is a branch of chemistry that studies chemical reactions “called redox reactions” which involve electron transfer. Zn(s) + Cu 2+(aq) Oxidation states 0 +2 Zn 2+(aq) + Cu(s) +2 Oxidation reactions Reduction reactions Oxidizing agent (reduced species) 0 Redox reactions Reducing agent (oxidized species)
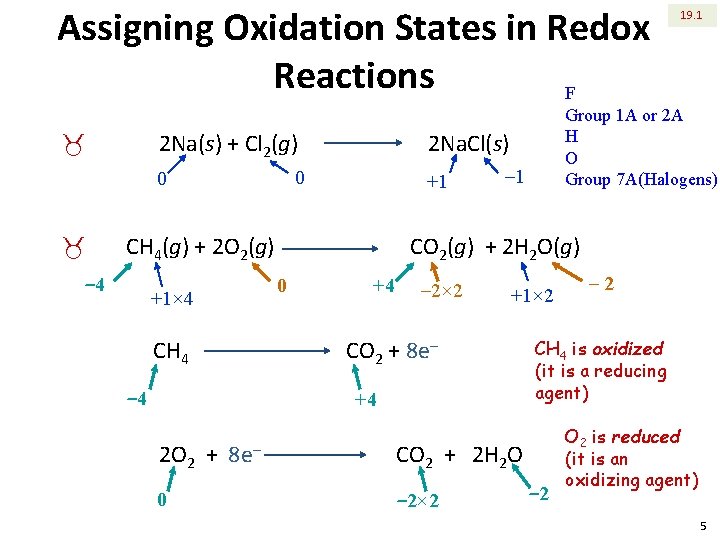
Assigning Oxidation States in Redox Reactions F 2 Na(s) + Cl 2(g) _ _ +1 CH 4(g) + 2 O 2(g) ‒ 4 CH 4 ‒ 1 CO 2(g) + 2 H 2 O(g) 0 +1× 4 +4 ‒ 2× 2 +1× 2 CO 2 + 8 e‒ ‒ 4 +4 2 O 2 + 0 8 e‒ Group 1 A or 2 A H O Group 7 A(Halogens) 2 Na. Cl(s) 0 0 19. 1 CH 4 is oxidized (it is a reducing agent) CO 2 + 2 H 2 O ‒ 2× 2 ‒ 2 O 2 is reduced (it is an oxidizing agent) 5

Balancing Redox Reactions in Media Cr 2 O 72 -(aq) + C 2 H 5 OH(l) Acidic Cr 3+(aq) + CO 2(g) 19. 1 H 2 O(l) H+(aq) Step 1: Write half equations (oxidation-reduction). Cr 2 O 72 -(aq) Cr 3+(aq) (reduction) +6 +3 C 2 H 5 OH(l) CO 2(g) -2 (oxidation) +4 Notice that we didn’t include H+ and H 2 O because they are going to be added at a later stage to balance the redox reaction. 6

Balancing Redox Reactions in Media Acidic 19. 1 Step 2: Balance all elements and charges for each half reaction. For the reduction half reaction: 6 e- + 14 H+ + Cr 2 O 72 - 2 Cr 3+ + 7 H 2 O a) Balance all elements except oxygen and hydrogen atoms. b) Balance oxygen with H 2 O. c) Balance hydrogen with H+. d) Balance charges using electrons. Similarly, for the oxidation half reaction: C 2 H 5 OH + 3 H 2 O 2 CO 2 + 12 H+ + 12 e 7

Balancing Redox Reactions in Media Acidic 19. 1 Step 3: Equalize the number of electrons in both balanced half reactions. 12 e- + 28 H+ + 2 Cr 2 O 72 - 4 Cr 3+ + 14 H 2 O Step 4: Add up the two half reactions: 12 e- + 28 H+ + 2 Cr 2 O 72 C 2 H 5 OH + 3 H 2 O 16 H+ + 2 Cr 2 O 72 - +C 2 H 5 OH The reduction half reaction after being multiplied by 2 4 Cr 3+ + 14 H 2 O 2 CO 2 + 12 H+ + 12 e 4 Cr 3+ + 11 H 2 O + 2 CO 2 Step 5: Check that elements and charges in the final reaction are balanced. 8

Balancing Redox Reactions in Basic Media – Ag(s) + CN (aq) + O 2(g) Step 1: Ag + CN– O 2 basic 19. 1 Ag(CN)2–(aq) + H 2 O(l) Ag(CN)2– (oxidation) H 2 O(l) (reduction) Step 2: Ag + 2 CN– O 2 + 4 H+ + 4 e– Ag(CN)2– + e– 2 H 2 O Step 3: 4 Ag + 8 CN– O 2 + 4 H+ + 4 e– 4 Ag(CN)2– + 4 e– 2 H 2 O 9

Balancing Redox Reactions in Basic Media Step 4: 4 Ag + 8 CN– + O 2 + 4 H+ 19. 1 4 Ag(CN)2– + 2 H 2 O But in alkaline, we have HOStep 5: – + + 4 OH O 4 Ag + 8 CN– + O 2 + 4 H 4 H 2 4 Ag(CN)2– +2 H 2 O + 4 OH– Step 6: Eliminate H 2 O molecules from both sides: 4 Ag + 8 CN– + O 2 + 2 H 2 O 4 Ag(CN)2– + 4 OH– Step 7: Recheck the balance of elements and charges. 10
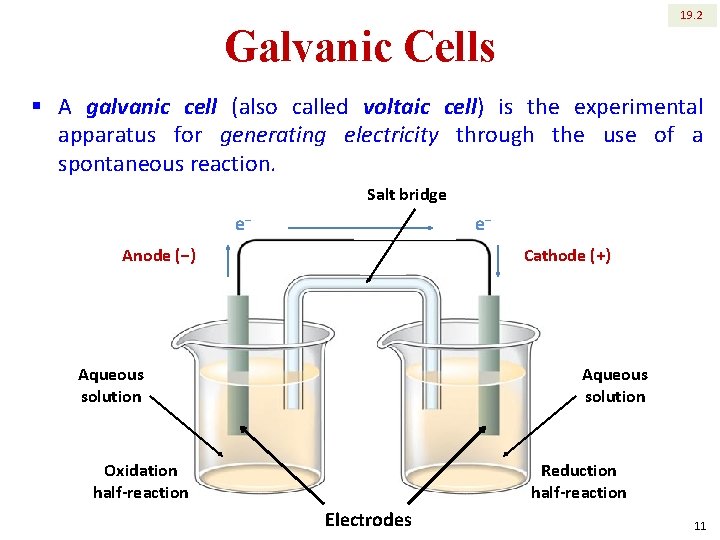
19. 2 Galvanic Cells § A galvanic cell (also called voltaic cell) is the experimental apparatus for generating electricity through the use of a spontaneous reaction. Salt bridge e‒ e‒ Anode (‒) Cathode (+) Aqueous solution Oxidation half-reaction Reduction half-reaction Electrodes 11
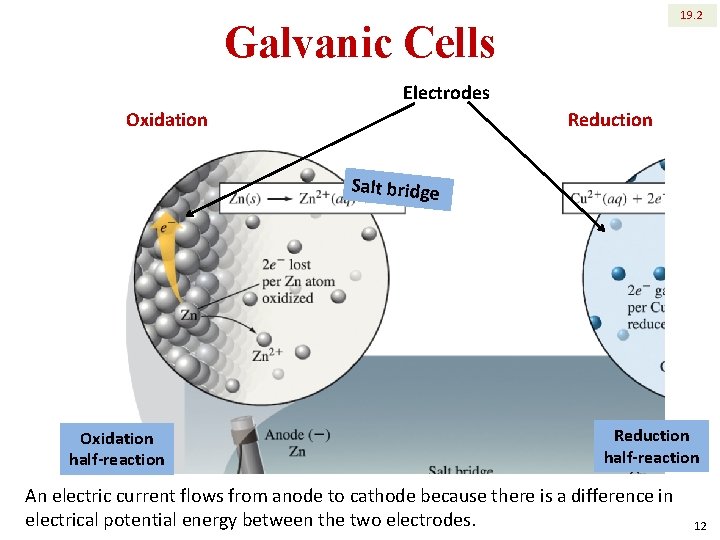
19. 2 Galvanic Cells Electrodes Reduction Oxidation Salt bridge Oxidation half-reaction Reduction half-reaction An electric current flows from anode to cathode because there is a difference in electrical potential energy between the two electrodes. 12
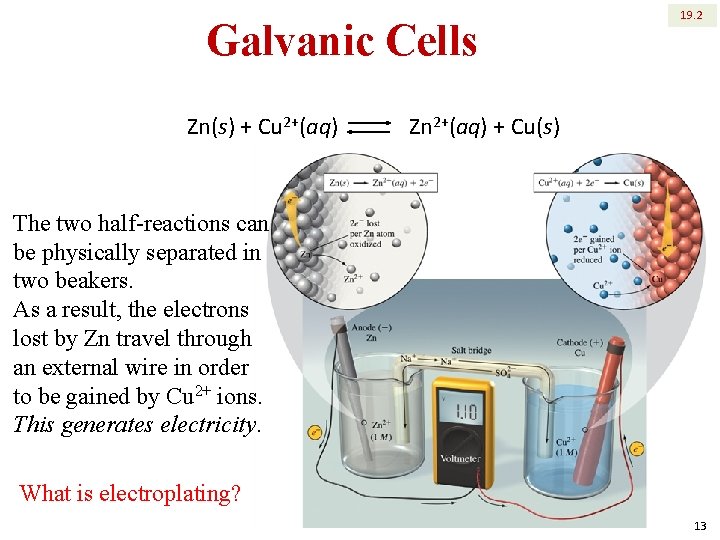
Galvanic Cells Zn(s) + Cu 2+(aq) 19. 2 Zn 2+(aq) + Cu(s) The two half-reactions can be physically separated in two beakers. As a result, the electrons lost by Zn travel through an external wire in order to be gained by Cu 2+ ions. This generates electricity. What is electroplating? 13
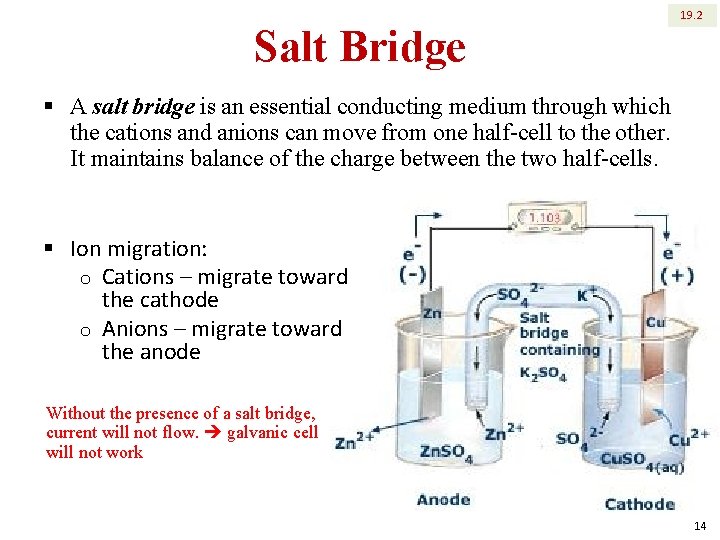
Salt Bridge 19. 2 § A salt bridge is an essential conducting medium through which the cations and anions can move from one half-cell to the other. It maintains balance of the charge between the two half-cells. § Ion migration: o Cations – migrate toward the cathode o Anions – migrate toward the anode Without the presence of a salt bridge, current will not flow. galvanic cell will not work 14
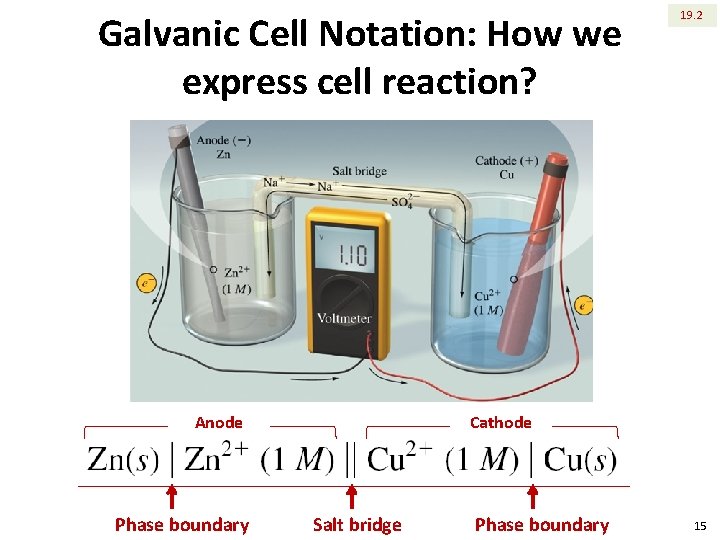
Galvanic Cell Notation: How we express cell reaction? Anode Phase boundary 19. 2 Cathode Salt bridge Phase boundary 15

19. 2 Cell Potential § The cell potential, cell voltage, Electromotive force, (E) is the difference in electrical potential between the cathode and the anode half-cells. § Ecell = Ecathode - Eanode § Cell potential is: o concentration dependent. o temperature dependent. o and nature of reactants. The cell potential is measured using a voltmeter with units of volts (V). 16

What we learned in last class… • • Oxidation & reduction Oxidizing agent & reducing agent Balancing Redox reactions Galvanic cell (Design, Anode, Cathode, Salt bridge, etc) • Galvanic cell notation • Cell potential (Ecell = Ecathode - Eanode) • Standard reduction potential

What we hope to learn today… • • Standard reduction potential Standard Hydrogen Electrode (SHE) Identifying cathode and anode Predicting the occurrence of redox reactions • Electrochemistry & Thermodynamics

Standard Reduction Potential 19. 3 § The standard reduction potential (E ) is the potential associated with a reduction half-reaction at an electrode when the ion concentration is 1 M and the gas pressure is 1 atm. The standard reduction potential (E ) for the standard hydrogen electrode (SHE) is assigned the value 0 V. shown as reduction § The relative standard reduction potentials of other halfreactions are measured relative to the SHE potential. 19

Standard Hydrogen Electrode 19. 3 Hydrogen/Pt electrode can be used as a reference for other electrodes. 2 H+ (aq) + 2 e- H 2 (g) § The platinum (Pt) electrode is used to: o provide a surface on which the oxidation of H 2 can take place. o serve as an electrical conductor. § Under standard state condition at 25 C , the reduction potential of H+ is defined as exactly zero. The standard hydrogen electrode (SHE) By definition 20
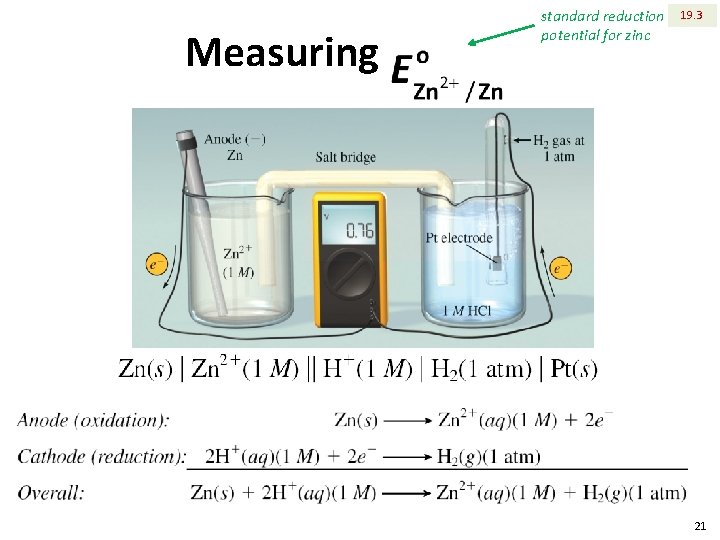
Measuring standard reduction potential for zinc 19. 3 21
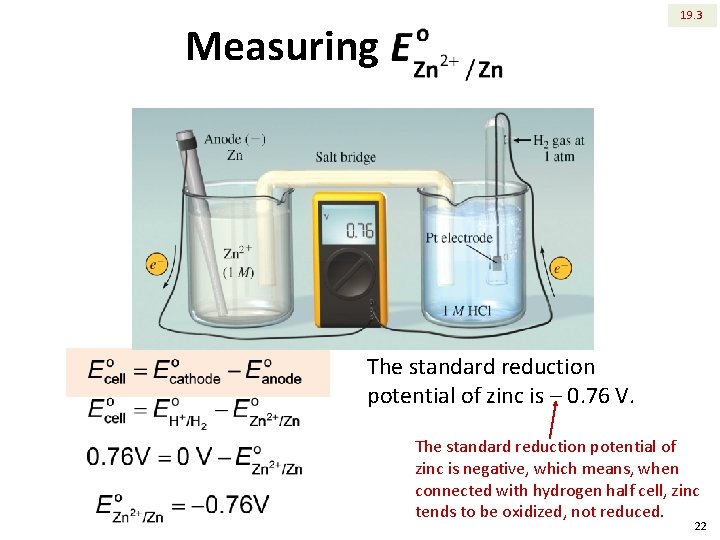
19. 3 Measuring The standard reduction potential of zinc is ‒ 0. 76 V. The standard reduction potential of zinc is negative, which means, when connected with hydrogen half cell, zinc tends to be oxidized, not reduced. 22
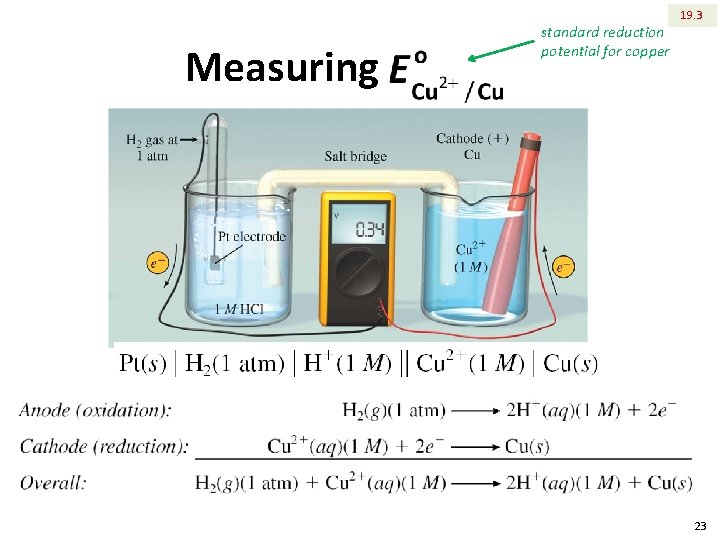
Measuring standard reduction potential for copper 19. 3 23
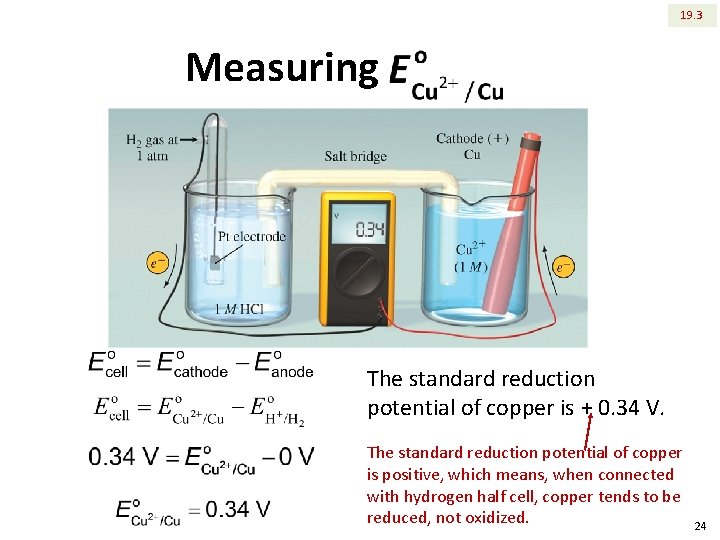
19. 3 Measuring The standard reduction potential of copper is + 0. 34 V. The standard reduction potential of copper is positive, which means, when connected with hydrogen half cell, copper tends to be reduced, not oxidized. 24

19. 3 E = + 0. 34 V Cathode 2 H+ (aq) + 2 e‒ H 2 (g) E = 0 V Anode Zn 2+ (aq) + 2 e‒ Zn (s) E = ‒ 0. 76 V Easier to be oxidized Cu 2+ (aq) + 2 e‒ Cu (s) Easier to be reduced Comparison of E Values 25

19. 3 of a Zinc-Copper Cell 26

Predicting the Occurrence of Redox Reactions
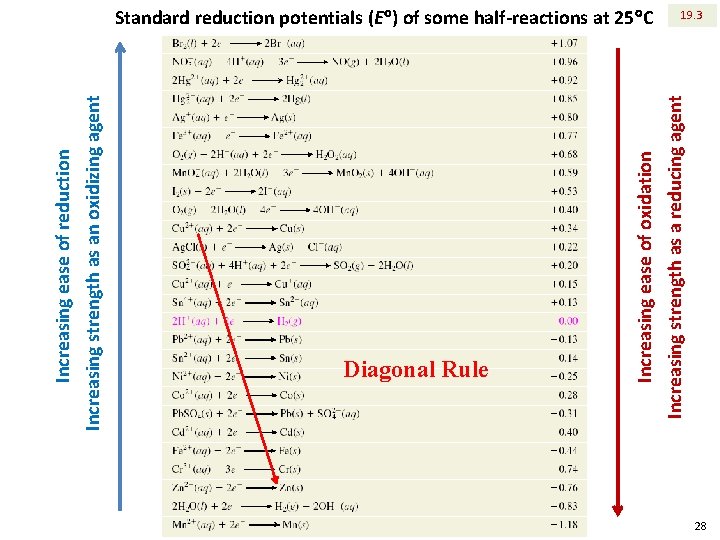
Increasing ease of reduction Increasing strength as an oxidizing agent Diagonal Rule Increasing ease of oxidation Increasing strength as a reducing agent Standard reduction potentials (E ) of some half-reactions at 25 C 19. 3 28

Predicting the Occurrence of Redox Reactions 19. 3 § Example: Determine whether redox reaction will occur or not at 25 C when lead metal (Pb) is added to: (a) 1. 0 M solution of Ni. Cl 2. (b) 1. 0 M solution of HCl. Also, write cell notation, calculate cell potential, write overall reaction? Pb 2+ (aq) + 2 e Pb(s) Cl 2 (gas) + 2 e 2 Cl-(aq) Ni 2+(aq) + 2 e Ni (s) 2 H+(aq) + 2 e H 2(gas) - 0. 13 V + 1. 36 V - 0. 25 V 0. 00 V Cl 2 (gas) + 2 e 2 Cl-(aq) 2 H+(aq) + 2 e H 2(gas) Pb 2+ (aq) + 2 e Pb(s) Ni 2+(aq) + 2 e Ni (s) + 1. 36 V 0. 00 V - 0. 13 V - 0. 25 V 29

Solution The proposed reaction will occur only if Thus, the redox reaction will NOT occur. The proposed reaction will occur only if Thus, the redox reaction will occur Cell notation: Pb(s) | Pb 2+(1 M) || 2 H+(1 M) | H 2(g) Cell potential: E°cathode - E°anode = 0 - ( -0. 13 V) = 0. 13 V Overall reaction: Pb(s) + 2 H+ Pb 2+ + H 2(g)

19. 3 Calculating the Standard Cell Potential ( ) § Example: Write the overall cell reaction and calculate the standard cell potential of the following cell? Zn(s) | Zn 2+(1 M) | | Ag+(1 M) | Ag(s) EZn 2+/Zn(s) = - 0. 76 V EAg+/Ag(s) = + 0. 80 V 31

Balanced equation of the overall cell reaction 2 Ag+ (aq) + 2 e‒ 2 Ag (s) Zn (s) 2 e‒ + Zn 2+ (aq) Zn (s) + 2 Ag+ (aq) Zn 2+ (aq) + 2 Ag (s) 32

Cell Potential Zn (s) + 2 Ag+ (aq) Zn 2+ (aq) + 2 Ag (s) Note: Standard reduction potential is an intensive property (like temperature and density), and it is not an extensive property (like mass and volume). In other words, it does NOT depend on the amounts of substances involved in the cell.

Example Determine the following for a Cadmium – Lead cell. - Redox reaction will occur or not ECd 2+/Cd(s) = - 0. 40 V 0 - E cell EPb 2+/Pb(s) = - 0. 13 V - Cell notation - Balanced equation of the overall cell reaction Note: Cadmium – Lead cell means - Cd electrode in 1. 0 M Cd(NO 3)2 and Lead electrode in 1. 0 M Pb(NO 3)2)?

E 0 cell Pb 2+ + 2 e- Pb(s) E = ‒ 0. 13 V Cd 2+ + 2 e- Cd(s) E = ‒ 0. 40 V (We have Pb 2+ and Cd(s), so the reaction will occur) Cell notation Cd(s) | Cd 2+ (1 M) || Pb 2+ (1 M) | Pb(s)

Balanced equation of the overall cell reaction Pb 2+ + 2 e. Cd(s) Pb(s) Reduction Cd 2+ + 2 e- Oxidation + Pb 2+ Cd 2+ + Pb(s)

What we discussed in last lecture… • • Standard reduction potential Standard Hydrogen Electrode (SHE) Identifying cathode and anode Predicting the occurrence of redox reactions

Today… • Electrochemistry & Thermodynamics (ΔG o or ΔG ) • Electrochemistry & Equilibrium constant
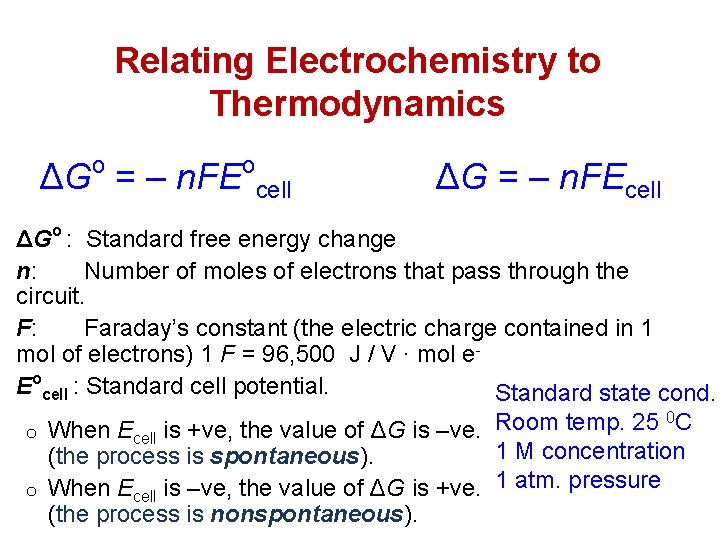
Relating Electrochemistry to Thermodynamics o ΔG = ‒ n. FE o cell ΔG = ‒ n. FEcell ΔGo : Standard free energy change n: Number of moles of electrons that pass through the circuit. F: Faraday’s constant (the electric charge contained in 1 mol of electrons) 1 F = 96, 500 J / V ∙ mol e. Eocell : Standard cell potential. Standard state cond. 0 C Room temp. 25 o When Ecell is +ve, the value of ΔG is ‒ve. 1 M concentration (the process is spontaneous). o When Ecell is ‒ve, the value of ΔG is +ve. 1 atm. pressure (the process is nonspontaneous).

19. 4 Relationship between and ΔG ΔGo = ‒ n. FEocell § Example: Give the balanced equation and determine the standard free energy change (ΔG ) for the following reaction at 25 C : 2 Al(s) + 3 Mn 2+(aq)(1. 0 M) 2 Al 3+(aq) (1. 0 M) + 3 Mn(s) The half-cell reactions: reduction: 3 Mn 2+(aq) + 62 e- 3 Mn(s) oxidation: 2 Al (s) 2 Al 3+(aq) + 63 eΔGo = ‒ n. FEocell = Eocathode – Eoanode = – 1. 18 V – (– 1. 66 V) = + 0. 48 ΔGo = ‒ n. FEocell = ‒ (6 e-)(9. 65 × 104 J/V∙mol e-)(0. 48 V) = ‒ 2. 78 × 105 J/mol = ‒ 2. 78 × 102 k. J/mol (product favored) 40

19. 4 Relationship between and K ΔGo = ‒ RT ln. K (from Chapter 18) ΔGo = ‒ n. FEocell Combining the above two equations: ‒ RT ln. K = ‒ n. FEocell Solving for Eocell : At 25 C: 41

19. 4 Relationship between and Kc § Example: Calculate the equilibrium constant for the following reaction at 25 C : 2 Al (s) + 3 Mn 2+(aq)(1. 0 M) 2 Al 3+(aq) (1. 0 M) + 3 Mn (s) Eocell = – 1. 18 V – (– 1. 66 V) = + 0. 48 Very large or very small K, which can be difficult to determine, is easily calculated using electrochemistry!! Remember that product will be favored (very large Kc value)! 42

Nonstandard States 19. 5 § The spontaneity of a chemical reaction at a condition other than the standard states (a nonstandard state means that concentrations are NOT 1 M) can be predicted using Nernst Equation. Since then: ΔGo = ‒ n. FEo and ΔG = ‒ n. FE ΔG = ΔGo + RT ln. Q (from Chapter 18) ‒ n. FE = ‒ n. FEo + RT ln. Q dividing the whole equation by ‒ n. F ; and setting T = 298 K : or 43
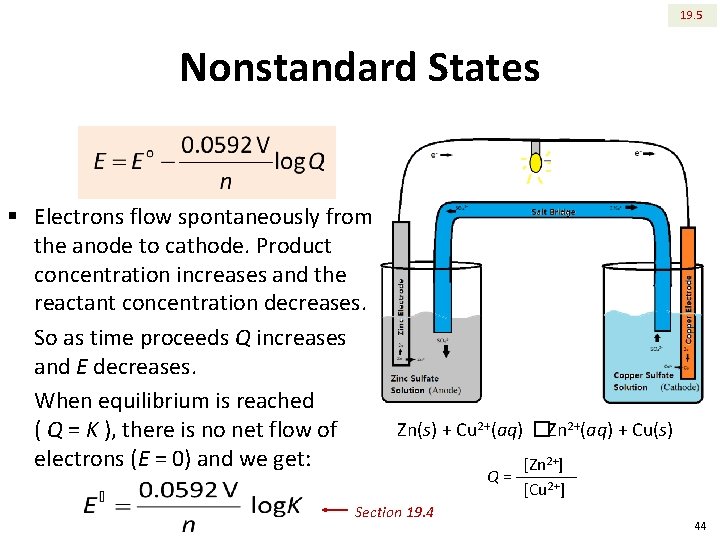
19. 5 Nonstandard States § Electrons flow spontaneously from the anode to cathode. Product concentration increases and the reactant concentration decreases. So as time proceeds Q increases and E decreases. When equilibrium is reached Zn(s) + Cu 2+(aq) �Zn 2+(aq) + Cu(s) ( Q = K ), there is no net flow of electrons (E = 0) and we get: [Zn 2+] Q= Section 19. 4 [Cu 2+] 44

19. 5 Conditions other than Standard State Example Predict whether the following reaction will occur spontaneously as written at 298 K: assuming [Co 2+] = 0. 15 M and [Fe 2+] = 0. 68 M. ECo 2+/Co(s) = - 0. 28 V EFe 2+/Fe(s) = - 0. 44 V 45

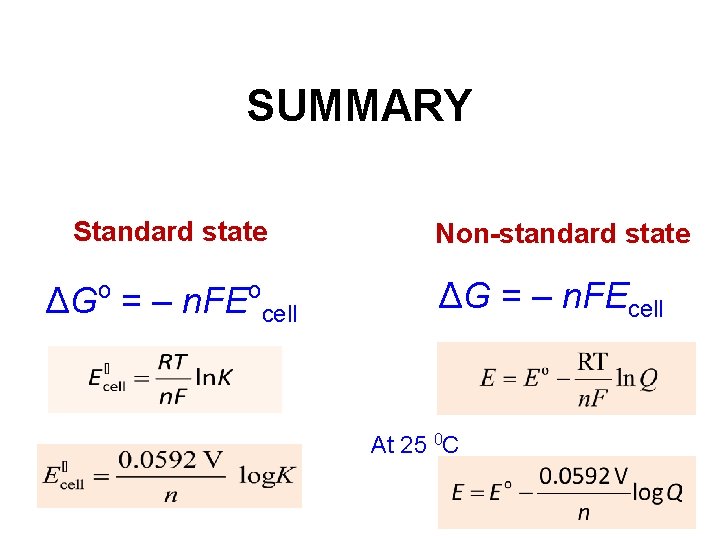
SUMMARY Standard state o ΔG = ‒ n. FE o cell Non-standard state ΔG = ‒ n. FEcell At 25 0 C
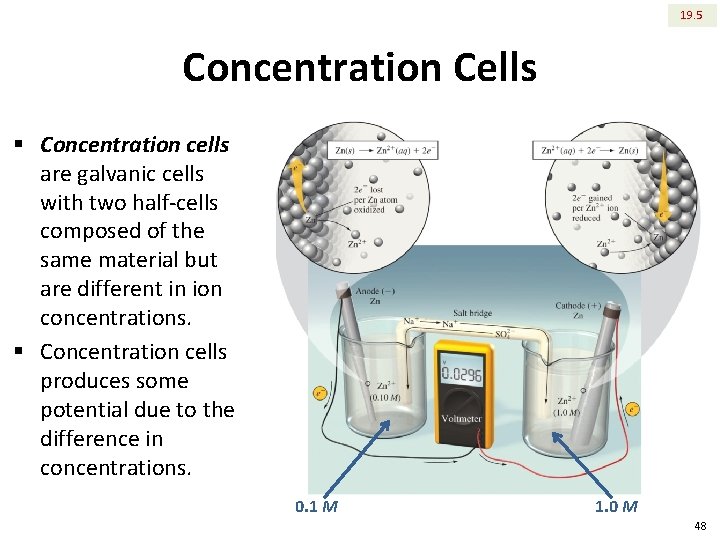
19. 5 Concentration Cells § Concentration cells are galvanic cells with two half-cells composed of the same material but are different in ion concentrations. § Concentration cells produces some potential due to the difference in concentrations. 0. 1 M 1. 0 M 48

19. 5 Concentration Cells Oxidation: Reduction: Overall: Zn(s) �Zn 2+(aq) (0. 10 M) + 2 e. Zn 2+(aq) (1. 0 M) + 2 e- �Zn(s) Zn 2+(aq) (1. 0 M) � Zn 2+(aq) (0. 10 M) § The cell potential is: equal to zero E of the concentration cell above is 0. 030 V. In general, concentration cells have small potentials. o As the process continues, E gradually decreases. When [Zn 2+]dilute becomes equal to [Zn 2+]conc. then E becomes zero. o 49
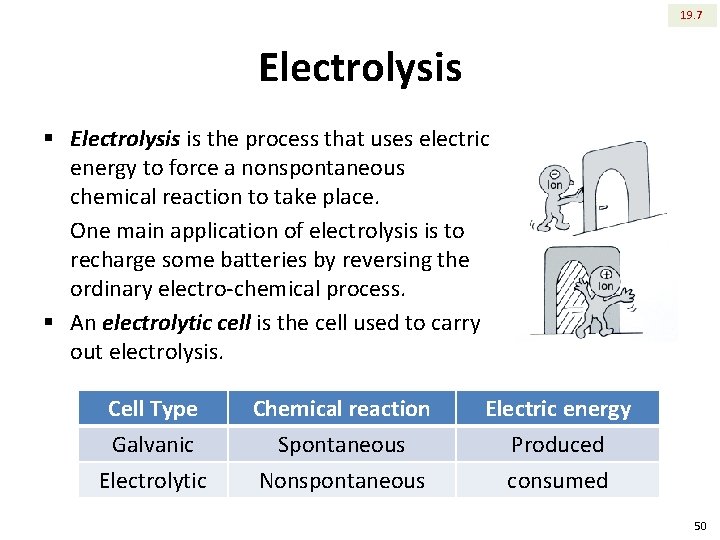
19. 7 Electrolysis § Electrolysis is the process that uses electric energy to force a nonspontaneous chemical reaction to take place. One main application of electrolysis is to recharge some batteries by reversing the ordinary electro-chemical process. § An electrolytic cell is the cell used to carry out electrolysis. Cell Type Galvanic Electrolytic Chemical reaction Spontaneous Nonspontaneous Electric energy Produced consumed 50
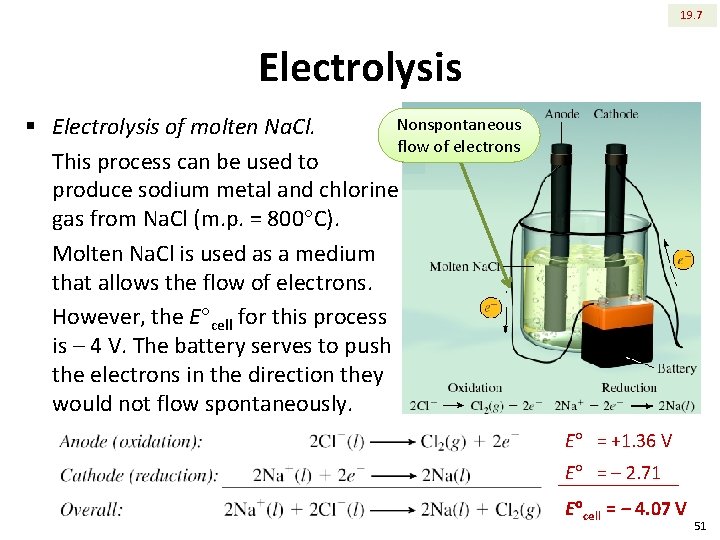
19. 7 Electrolysis Nonspontaneous § Electrolysis of molten Na. Cl. flow of electrons This process can be used to produce sodium metal and chlorine gas from Na. Cl (m. p. = 800 C). Molten Na. Cl is used as a medium that allows the flow of electrons. However, the E cell for this process is ‒ 4 V. The battery serves to push the electrons in the direction they would not flow spontaneously. E = +1. 36 V E = ‒ 2. 71 E cell = ‒ 4. 07 V 51
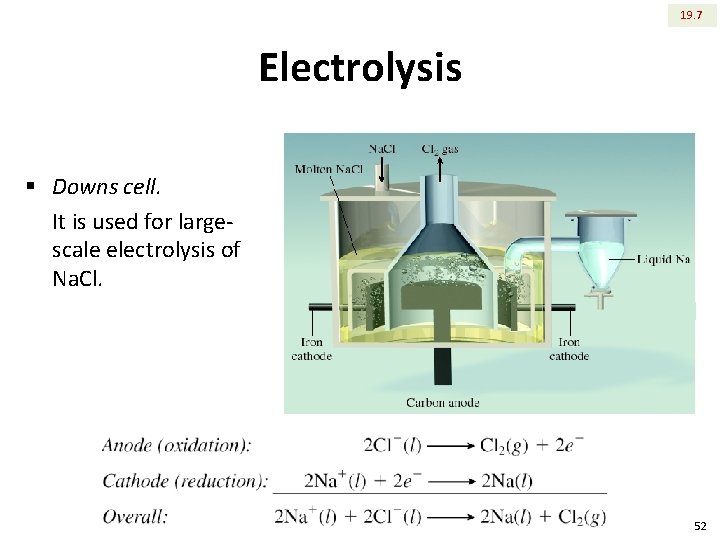
19. 7 Electrolysis § Downs cell. It is used for largescale electrolysis of Na. Cl. 52
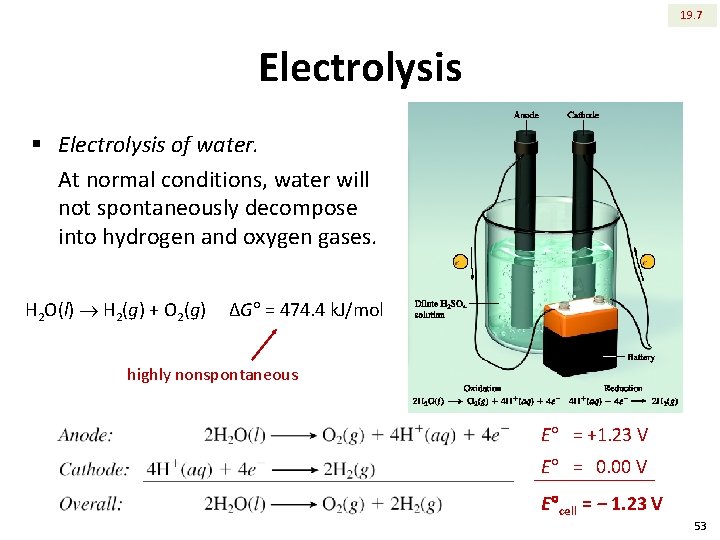
19. 7 Electrolysis § Electrolysis of water. At normal conditions, water will not spontaneously decompose into hydrogen and oxygen gases. H 2 O(l) H 2(g) + O 2(g) ΔG = 474. 4 k. J/mol highly nonspontaneous E = +1. 23 V E = 0. 00 V E cell = ‒ 1. 23 V 53

19. 7 Electrolysis § Electrolysis of water. Pure water does not have sufficient ions, so 0. 1 M H 2 SO 4 solution is used to conduct electric current and establish the circuit. There is no net consumption of H 2 SO 4 in the over all reaction. 54

19. 7 Electrolysis of an Aqueous Sodium Chloride Solution Thus, the half-cell reactions in the electrolysis of aqueous sodium chloride are 55
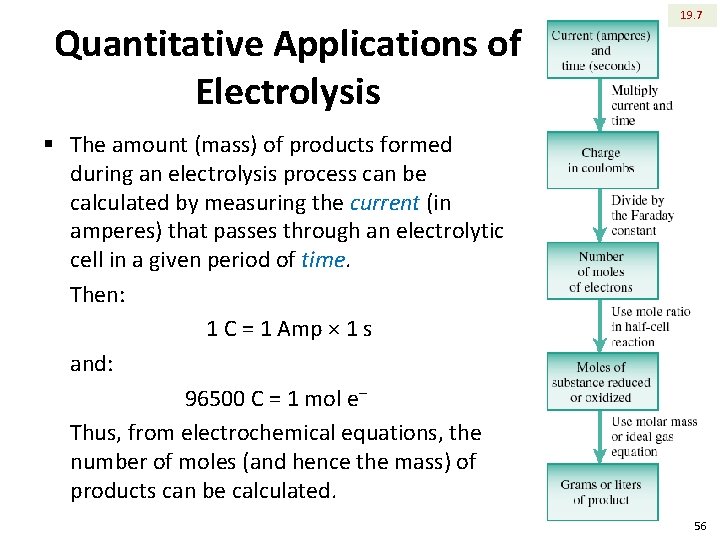
Quantitative Applications of Electrolysis 19. 7 § The amount (mass) of products formed during an electrolysis process can be calculated by measuring the current (in amperes) that passes through an electrolytic cell in a given period of time. Then: 1 C = 1 Amp × 1 s and: 96500 C = 1 mol e‒ Thus, from electrochemical equations, the number of moles (and hence the mass) of products can be calculated. 56

19. 7 Quantitative Applications of Electrolysis Consider an electrolytic cell in which molten Ca. Cl 2 is separated into its constituent elements, Ca and Cl 2. Suppose a current of 0. 452 A is passed through the cell for 1. 50 h. How much product will be formed at each electrode? 57

19. 7 Electrolysis Quantitative Applications of Electrolysis 58
- Slides: 58