Chemistry Third Edition Julia Burdge Lecture Power Points

Chemistry Third Edition Julia Burdge Lecture Power. Points Chapter 20 Nuclear Chemistry Copyright © 2012, The Mc. Graw-Hill Compaies, Inc. Permission required for reproduction or display.

CHAPTER 20. 1 20. 2 20. 3 20. 4 20. 5 20. 6 20. 7 20. 8 20 Nuclear Chemistry Nuclei and Nuclear Reactions Nuclear Stability Natural Radioactivity Nuclear Transmutation Nuclear Fission Nuclear Fusion Uses of Isotopes Biological Effects of Radiation 2

20. 1 Nuclei and Nuclear Reactions Topics Nuclei and Nuclear Reactions 3

20. 1 Nuclei and Nuclear Reactions All nuclei contain protons and neutrons. Radioactivity is the spontaneous emission of particles or electromagnetic radiation from unstable nuclei. All elements having an atomic number > 83 are unstable and radioactive. Nuclear transmutation results from the bombardment of nuclei by neutrons, protons, or other nuclei. 4

20. 1 Nuclei and Nuclear Reactions Radioactive decay and nuclear transmutation are nuclear reactions. 5

20. 1 Nuclei and Nuclear Reactions In nuclear equations, we must indicate the number of subatomic particles in every species. The superscript denotes the mass number. The subscript is the atomic number. 6

20. 1 Nuclei and Nuclear Reactions 7

20. 1 Nuclei and Nuclear Reactions In balancing nuclear equations, we must balance • the total of all atomic numbers • the total of all mass numbers for the products and reactants. We can identify the unknown species if we know the atomic numbers and mass numbers of all but one of the species in a nuclear equation. 8

SAMPLE PROBLEM 20. 1 Identify the missing species X in each of the following nuclear equations: 9

SAMPLE PROBLEM 20. 1 Strategy Setup a) 212 = (208 + mass number of X); mass number of X = 4. 84 = (82 + atomic number of X); atomic number of X = 2. b) 90 = (mass number of X + 0); mass number of X = 90. 10
![SAMPLE PROBLEM 20. 1 38 = [atomic number of X + (‒ 1)]; atomic SAMPLE PROBLEM 20. 1 38 = [atomic number of X + (‒ 1)]; atomic](http://slidetodoc.com/presentation_image_h2/b03cd7dbb8246aea8807eff2f82301b2/image-11.jpg)
SAMPLE PROBLEM 20. 1 38 = [atomic number of X + (‒ 1)]; atomic number of X = 39. c) Mass number of X = (18 + 0); mass number of X = 18. Atomic number of X = (8 + 1); atomic number of X = 9. Solution 11

20. 2 Nuclear Stability Topics Patterns of Nuclear Stability Nuclear Binding Energy 12

20. 2 Nuclear Stability Patterns of Nuclear Stability The nucleus occupies a small portion of the total volume of an atom, but it contains most of the atom’s mass. 13

20. 2 Nuclear Stability Patterns of Nuclear Stability The average atomic nucleus is roughly 9 × 1012 (or 9 trillion) times as dense as the densest element known! 14

20. 2 Nuclear Stability Patterns of Nuclear Stability From Coulomb’s law, we know that like charges repel and unlike charges attract one another. The stability of any nucleus is determined by the difference between coulombic repulsion and the short-range attraction. 15

20. 2 Nuclear Stability Patterns of Nuclear Stability The principal factor that determines nuclear stability is the neutron-to-proton ratio (n/p). For stable elements having low atomic number (≤ 20), the n/p value is close to 1. As atomic number increases, the n/p ratios also increases. 16

20. 2 Nuclear Stability Patterns of Nuclear Stability Rules for predicting stability: 1. There are more stable nuclei containing 2, 8, 20, 50, 82, or 126 protons or neutrons. These are called magic numbers. 2. There are many more stable nuclei with even numbers of both protons and neutrons than with odd numbers of these particles. 17
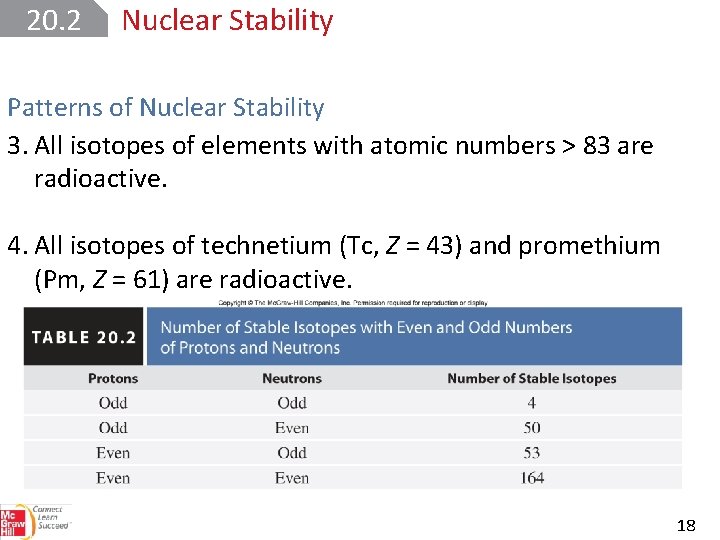
20. 2 Nuclear Stability Patterns of Nuclear Stability 3. All isotopes of elements with atomic numbers > 83 are radioactive. 4. All isotopes of technetium (Tc, Z = 43) and promethium (Pm, Z = 61) are radioactive. 18
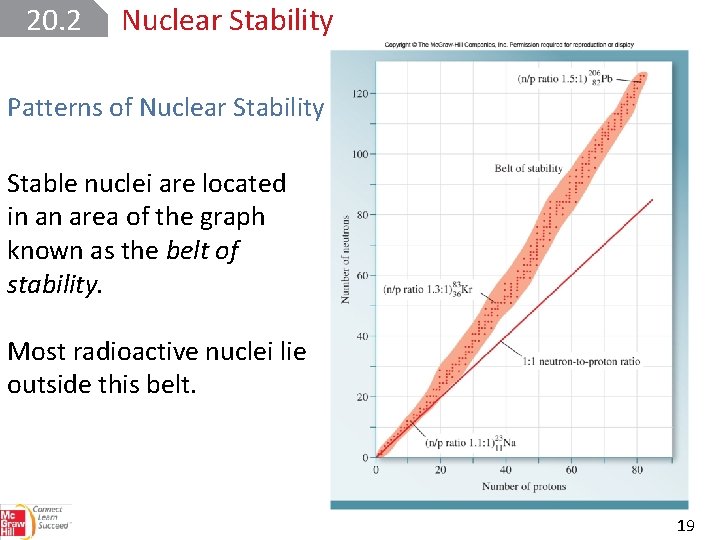
20. 2 Nuclear Stability Patterns of Nuclear Stability Stable nuclei are located in an area of the graph known as the belt of stability. Most radioactive nuclei lie outside this belt. 19

20. 2 Nuclear Stability Patterns of Nuclear Stability Above the belt of stability, nuclei have higher n/p ratios. To lower this ratio, these nuclei undergo β-particle emission: Increases number of protons; decreases number of neutrons. 20

20. 2 Nuclear Stability Patterns of Nuclear Stability Below the belt of stability, nuclei have lower n/p ratios. To increase this ratio, these nuclei may emit a positron, An example of positron emission is 21

20. 2 Nuclear Stability Patterns of Nuclear Stability A nucleus may also undergo electron capture, Electron capture is the capture of an electron—usually a 1 s electron—by the nucleus. Electron capture has the same net effect on the nucleus as positron emission, the atomic number decreases by 1. : 22

20. 2 Nuclear Stability Nuclear Binding Energy A quantitative measure of nuclear stability is the nuclear binding energy, which is the energy required to break up a nucleus into its component protons and neutrons. The masses of nuclei are always less than the sum of the masses of the nucleons, which is a general term for the protons and neutrons in a nucleus. 23

20. 2 Nuclear Stability Nuclear Binding Energy 24

20. 2 Nuclear Stability Nuclear Binding Energy The mass of 10 neutrons is 25

20. 2 Nuclear Stability Nuclear Binding Energy 26

20. 2 Nuclear Stability Nuclear Binding Energy We can calculate the amount of energy released: E is energy, m is mass, and c is the velocity of light Where, 27

20. 2 Nuclear Stability Nuclear Binding Energy The change in mass is or 28

20. 2 Nuclear Stability Nuclear Binding Energy Δm is a negative quantity. ΔE is also a negative quantity; energy is released to the surroundings. Calculate ΔE as follows: 29

20. 2 Nuclear Stability Nuclear Binding Energy The nuclear binding energy of the nucleus is 2. 3682 × 10‒ 11 J. For 1 mole of fluorine nuclei, the energy released is: The nuclear binding energy is 1. 4261 × 1010 k. J for 1 mole of fluorine-19 nuclei 30

20. 2 Nuclear Stability Nuclear Binding Energy When comparing the stability of any two nuclei, we must account for the fact that they have different numbers of nucleons. It makes more sense to compare nuclei using the nuclear binding energy per nucleon: 31

20. 2 Nuclear Stability Nuclear Binding Energy For the fluorine-19 nucleus, The greater the nuclear binding energy per nucleon, the more stable the nucleus. 32
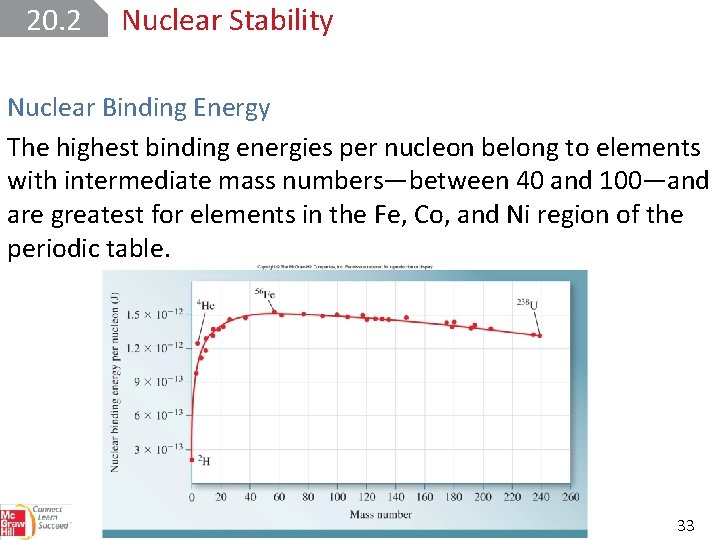
20. 2 Nuclear Stability Nuclear Binding Energy The highest binding energies per nucleon belong to elements with intermediate mass numbers—between 40 and 100—and are greatest for elements in the Fe, Co, and Ni region of the periodic table. 33

SAMPLE PROBLEM 20. 2 34

SAMPLE PROBLEM 20. 2 The mass of 74 neutrons is 35

SAMPLE PROBLEM 20. 2 The mass defect is 36

SAMPLE PROBLEM 20. 2 The energy released is The nuclear binding energy is 1. 71847 × 10‒ 10 J. The nuclear binding energy per nucleon is 37

20. 3 Natural Radioactivity Topics Kinetics of Radioactive Decay Dating Based on Radioactive Decay 38

20. 3 Natural Radioactivity Kinetics of Radioactive Decay The spontaneous emission by unstable nuclei of particles and/or electromagnetic radiation is known as radioactivity. The main types of radioactivity are: • the emission of α particles • the emission of β particles • the emission of γ rays, which are very-short-wavelength electromagnetic waves • the emission of positrons • electron capture 39

20. 3 Natural Radioactivity Kinetics of Radioactive Decay A radioactive decay series is a sequence of nuclear reactions that ultimately result in the formation of a stable isotope. Here is the uranium decay series: 40

20. 3 Natural Radioactivity Kinetics of Radioactive Decay It is important to balance the nuclear reaction for each step in a radioactive decay series. For example, the first step in the uranium decay series is the decay of uranium-238 to thorium-234, with the emission of an α particle: The next step is represented by 41

20. 3 Natural Radioactivity Kinetics of Radioactive Decay 42

20. 3 Natural Radioactivity Kinetics of Radioactive Decay All radioactive decays obey first-order kinetics. The rate of radioactive decay at any time t is given by k is the first-order rate constant N is the number of radioactive nuclei present 43

20. 3 Natural Radioactivity Kinetics of Radioactive Decay The number of radioactive nuclei at time zero (N 0) and time t (Nt) is The corresponding half-life of the reaction is 44

20. 3 Natural Radioactivity Dating Based on Radioactive Decay The half-lives of radioactive isotopes have been used as “atomic clocks” to determine the ages of certain objects. The carbon-14 isotope is produced when atmospheric nitrogen is bombarded by cosmic rays: The radioactive carbon-14 isotope decays according to the equation 45

20. 3 Natural Radioactivity Dating Based on Radioactive Decay This is the basis of radiocarbon or “carbon-14” dating. To determine the age of an object, we measure the activity (disintegrations per second) of 14 C and compare it to the activity of 14 C in living matter. 46

SAMPLE PROBLEM 20. 3 A wooden artifact is found to have a 14 C activity of 9. 1 disintegrations per second. Given that the 14 C activity of an equal mass of fresh-cut wood has a constant value of 15. 2 disintegrations per second, determine the age of the artifact. The half-life of carbon-14 is 5715 years. 47

SAMPLE PROBLEM 20. 3 Strategy The activity of a radioactive sample is proportional to the number of radioactive nuclei. Setup 48

SAMPLE PROBLEM 20. 3 Solution The age of the artifact is 4. 2 × 103 years. Carbon dating cannot be used for objects older than about 60, 000 years (about 10 half-lives). 49

20. 3 Natural Radioactivity Dating Based on Radioactive Decay The uranium decay series is suitable for estimating the age of rocks found on Earth and of extraterrestrial objects. As a good approximation, we can assume that the half-life for the overall process is equal to the half-life of the first step: 50

20. 3 Natural Radioactivity Dating Based on Radioactive Decay After one half-life, half of the original uranium-238 has been converted to lead-206 51

SAMPLE PROBLEM 20. 4 A rock is found to contain 5. 51 mg of 238 U and 1. 63 mg of 206 Pb. Determine the age of the rock (t 1/2 of 238 U = 4. 51 × 109 yr). 52

SAMPLE PROBLEM 20. 4 Setup The original mass of 238 U was 5. 51 mg + 1. 88 mg = 7. 39 mg. 53

SAMPLE PROBLEM 20. 4 Solution The rock is 1. 9 billion years old. 54

20. 4 Nuclear Transmutation Topics Nuclear Transmutation 55

20. 4 Nuclear Transmutation The following experiment suggested the possibility of producing radioactivity artificially. This reaction demonstrated the feasibility of converting one element into another, by the process of nuclear transmutation. Nuclear transmutation differs from radioactive decay in that transmutation is brought about by the collision of two particles. 56

20. 4 Nuclear Transmutation 57

SAMPLE PROBLEM 20. 5 58

SAMPLE PROBLEM 20. 5 Check your work by summing the mass numbers and the atomic numbers on both sides of the equation. 59

20. 4 Nuclear Transmutation Particle accelerators made it possible to synthesize the transuranium elements, elements with atomic numbers greater than 92. Neptunium (Z = 93) was first prepared in 1940. Since then, 24 other transuranium elements have been synthesized. 60
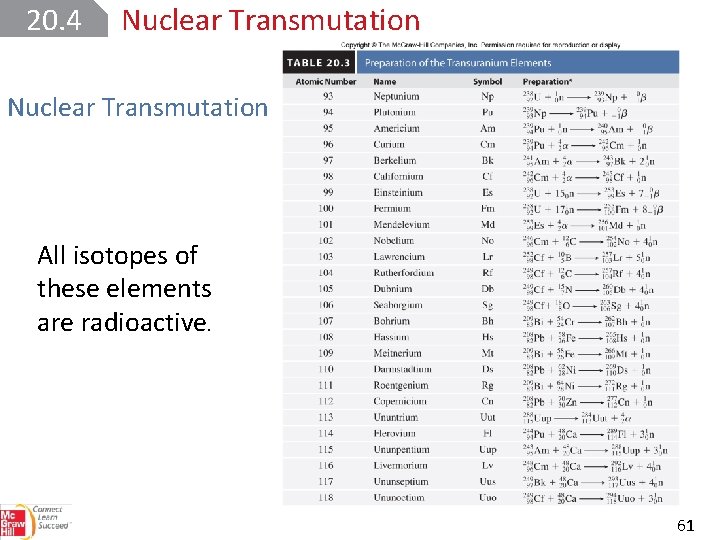
20. 4 Nuclear Transmutation All isotopes of these elements are radioactive. 61

20. 4 Nuclear Transmutation Tritium decays with the emission of β particles: 62

20. 4 Nuclear Transmutation Positively charged must have considerable kinetic energy to overcome the electrostatic repulsion between themselves and the target nuclei. A particle accelerator uses electric and magnetic fields to increase the kinetic energy of charged species so that a reaction will occur. 63
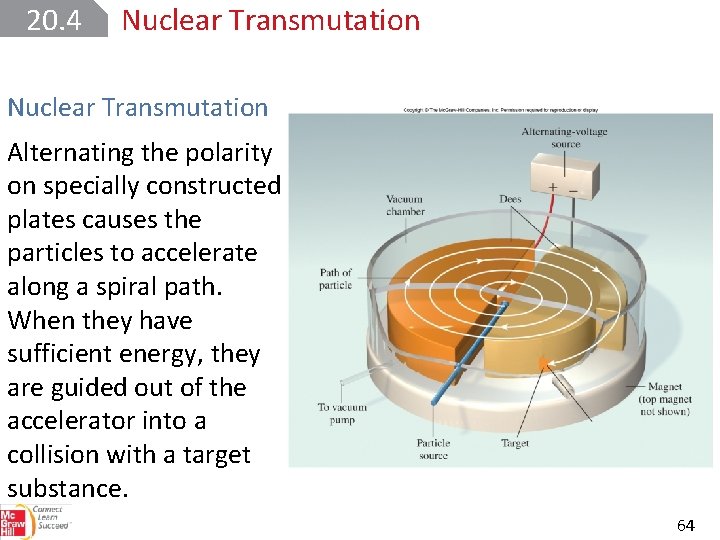
20. 4 Nuclear Transmutation Alternating the polarity on specially constructed plates causes the particles to accelerate along a spiral path. When they have sufficient energy, they are guided out of the accelerator into a collision with a target substance. 64

20. 4 Nuclear Transmutation Particles may also be accelerated along a linear path. 65

20. 5 Nuclear Fission Topics Nuclear Fission 66

20. 5 Nuclear Fission Nuclear fission is the process in which a heavy nucleus (mass number > 200) divides to form smaller nuclei of intermediate mass and one or more neutrons. This process releases a large amount of energy. Uranium-235 undergoes nuclear fission when bombarded with slow neutrons 67

20. 5 Nuclear Fission The difference between the binding energies of the reactants and products is (1. 23 × 10‒ 10 + 1. 92 × 10‒ 10 ) J ‒ (2. 82 × 10‒ 10 ) J, or 3. 3 × 10‒ 11 J per uranium-235 nucleus. For 1 mole of uranium-235, the energy released would be (3. 3 × 10‒ 11 ) (6. 02 × 1023), or 2. 0 × 1013 J. This is an extremely exothermic reaction. 68

20. 5 Nuclear Fission During uranium-235 fission, more neutrons are produced than are originally captured in the process. This property makes possible a nuclear chain reaction, which is a self-sustaining sequence of nuclear fission reactions. If there is not enough uranium-235 to capture the neutrons, the chain reaction will not occur and the mass of the sample is said to be subcritical. 69
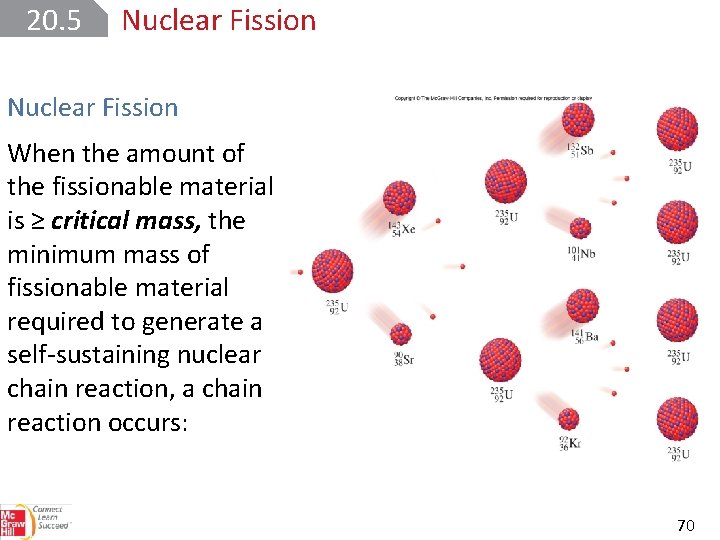
20. 5 Nuclear Fission When the amount of the fissionable material is ≥ critical mass, the minimum mass of fissionable material required to generate a self-sustaining nuclear chain reaction, a chain reaction occurs: 70
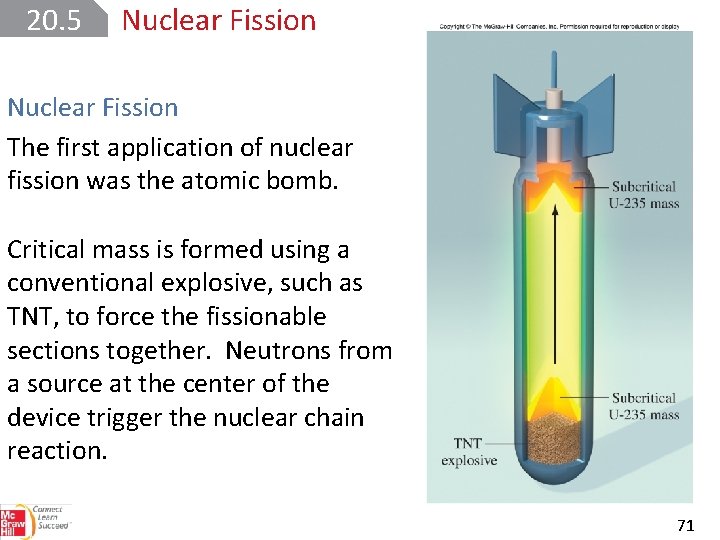
20. 5 Nuclear Fission The first application of nuclear fission was the atomic bomb. Critical mass is formed using a conventional explosive, such as TNT, to force the fissionable sections together. Neutrons from a source at the center of the device trigger the nuclear chain reaction. 71

20. 5 Nuclear Fission Nuclear fission is also used in the generation of electricity. Slow neutrons split uranium-235 nuclei more efficiently than fast ones. For greater efficiency, neutrons must be slowed down. To accomplish this goal, scientists use moderators, which are substances that can reduce the kinetic energy of neutrons. 72

20. 5 Nuclear Fission 73
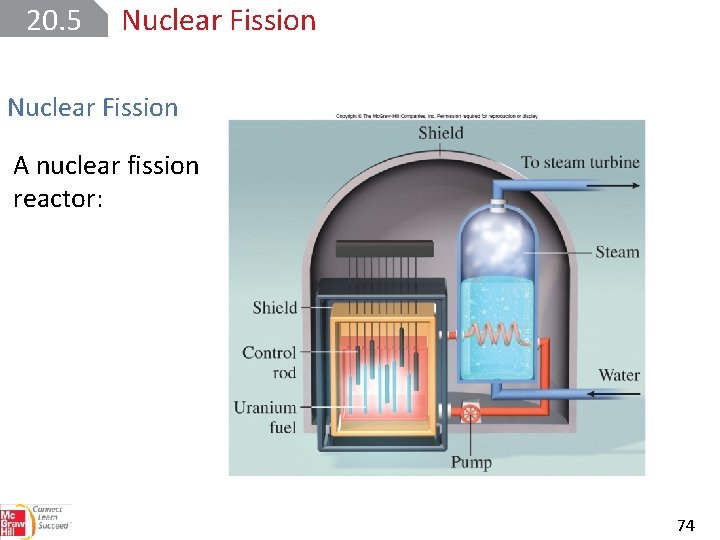
20. 5 Nuclear Fission A nuclear fission reactor: 74

20. 5 Nuclear Fission The nuclear fuel consists of uranium, usually in the form of an oxide, U 3 O 8 that is enriched in uranium-235. The factor limiting the rate of the reaction is the number of neutrons present. This can be controlled by lowering cadmium or boron control rods between the fuel elements. These rods capture neutrons 75

20. 5 Nuclear Fission Another type of nuclear reactor uses D 2 O, or heavy water, as the moderator. Deuterium absorbs neutrons much less efficiently than does ordinary hydrogen. The reactor is more efficient and does not require enriched uranium. However, D 2 O must be prepared which can be very expensive considering the amount of water used in a nuclear reactor. 76

20. 5 Nuclear Fission A breeder reactor uses uranium fuel, but unlike a conventional nuclear reactor, it produces more fissionable materials than it uses. When uranium-238 is bombarded with fast neutrons, the following reactions take place 77

20. 5 Nuclear Fission In a typical breeder reactor, the nonfissionable uranium-238 is transmuted into the fissionable isotope plutonium-239 The stockpile of fissionable material can be steadily increased. It takes about 7 to 10 years to regenerate the sizable amount of material needed to refuel the original reactor and to fuel another reactor of comparable size. This interval is called the doubling time. 78

20. 5 Nuclear Fission Disadvantages of Breeder Reactors: • Very expensive compared to conventional reactors. • There also more technical difficulties associated with the construction of such reactors. • Risk of accidents • Radioactive waste disposal 79

20. 6 Nuclear Fusion Topics Nuclear Fusion 80

20. 6 Nuclear Fusion Nuclear fusion is the combining of small nuclei into larger ones. When two light nuclei combine to form a larger, more stable nucleus, an appreciable amount of energy will be released in the process. Research is focused on the harnessing of nuclear fusion for the production of energy. 81

20. 6 Nuclear Fusion The sun is made up mostly of hydrogen and helium. Nuclear fusion occurs constantly in the sun where temperatures reach about 15 million degrees Celsius. 82

20. 6 Nuclear Fusion Because fusion reactions take place only at very high temperatures, they are often called thermonuclear reactions. These temperatures are a major concern in choosing the proper nuclear fusion process for energy production. Some promising reactions are listed here: 83

20. 6 Nuclear Fusion Advantage of nuclear fusion: • Fuels are cheap and almost inexhaustible. • The process produces little radioactive waste. No danger of a meltdown. 84

20. 6 Nuclear Fusion Many technical difficulties due to the extreme temperature. At temperatures of about 100 million degrees Celsius, molecules exist in a state of matter called plasma: a gaseous mixture of positive ions and electrons. No solid container can exist at such temperatures to contain the plasma. 85

20. 6 Nuclear Fusion One approach to solving this problem is to use magnetic confinement. Plasma moves through a doughnut-shaped tunnel, confined by a complex magnetic field. 86

20. 6 Nuclear Fusion The hydrogen bomb, also called a thermonuclear bomb, contains solid lithium deuteride (Li. D). The detonation of a hydrogen bomb occurs in two stages— first a fission reaction and then a fusion reaction. The required temperature for fusion is achieved with an atomic bomb. 87

20. 6 Nuclear Fusion Immediately after the atomic bomb explodes, the following fusion reactions occur, releasing vast amounts of energy: 88

20. 7 Uses of Isotopes Topics Chemical Analysis Isotopes in Medicine 89

20. 7 Uses of Isotopes Chemical Analysis The formula of the thiosulfate ion is S 2 O 32‒. Chemists were uncertain as to whether the two sulfur atoms occupied equivalent positions in the ion. The thiosulfate ion is prepared by When thiosulfate is treated with dilute acid, the reaction is reversed 90

20. 7 Uses of Isotopes Chemical Analysis The sulfur-35 isotope acts as a “label” for S atoms. All the labels are found in the sulfur precipitate; none of them appears in the final sulfite ions. Therefore, the two atoms of sulfur in S 2 O 32‒ are not structurally equivalent. The structure is not 91

20. 7 Uses of Isotopes Chemical Analysis Isotopes, especially radioactive isotopes that are used to trace the path of the atoms of an element in a chemical or biological process, are called tracers. 92
20. 7 Uses of Isotopes in Medicine Tracers are used for diagnosis in medicine. Sodium-24 injected into the bloodstream can detect possible constrictions or obstructions in the circulatory system. Iodine-131 has been used to test the activity of the thyroid gland. Iodine-123 is used to image the brain. 93

20. 7 Uses of Isotopes in Medicine Technetium is one of the most useful elements in nuclear medicine. All its isotopes are radioactive. In the laboratory it is prepared by the nuclear reactions “m” denotes that Tc is in its excited nuclear state 94
20. 7 Uses of Isotopes in Medicine By detecting the γ rays emitted by 99 m. Tc, doctors can obtain images of organs such as the heart, liver, and lungs. A major advantage of using radioactive isotopes as tracers is that they are easy to detect using a Geiger counter. 95

20. 8 Biological Effects of Radiation Topics Biological Effects of Radiation 96

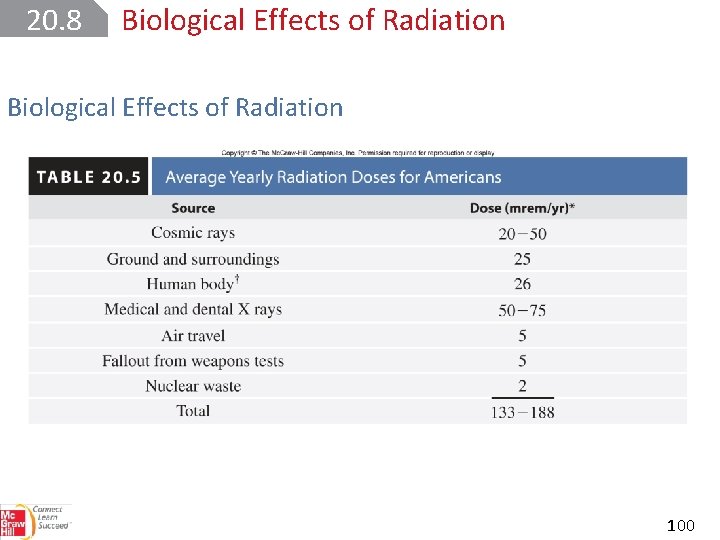
20. 8 Biological Effects of Radiation The fundamental unit of radioactivity is the curie (Ci). 1 Ci corresponds to exactly 3. 70 × 1010 nuclear disintegrations per second. A millicurie (m. Ci) is one-thousandth of a curie. 10 m. Ci of a carbon-14 sample is the quantity that undergoes (10 × 1023)(3. 70 × 1010 ) = 3. 70 × 108 disintegrations per second. 97

20. 8 Biological Effects of Radiation The intensity of radiation depends on: • the number of disintegrations • the energy and type of radiation emitted The rad (radiation absorbed dose) is the amount of radiation that results in the absorption of 1 × 10‒ 5 J per gram of irradiated material. 98

20. 8 Biological Effects of Radiation The rad is often multiplied by a factor called the RBE (relative biological effectiveness). The product is called a rem (roentgen equivalent for man): Penetrating power of nuclear radiation: α<β<γ 99
20. 8 Biological Effects of Radiation 100

20. 8 Biological Effects of Radiation can remove electrons from atoms and molecules in its path, leading to the formation of ions and radicals. Radicals (also called free radicals) are molecular fragments having one or more unpaired electrons; they are usually short lived and highly reactive. In tissues, free radicals attack cell membranes and a host of organic compounds. 101

20. 8 Biological Effects of Radiation damage to living systems is generally classified as somatic or genetic. Somatic injuries are those that affect the organism during its own lifetime. Genetic damage means inheritable changes or gene mutations. 102
- Slides: 102