Chemistry Third Edition Julia Burdge Lecture Power Points

Chemistry Third Edition Julia Burdge Lecture Power. Points Chapter 10 Gases Copyright © 2012, The Mc. Graw-Hill Compaies, Inc. Permission required for reproduction or display.

CHAPTER 10. 1 10. 2 10. 3 10. 4 10. 5 10. 6 10. 7 10 Gases Properties of Gases The Gas Laws The Ideal Gas Equation Reactions with Gaseous Reactants and Products Gas Mixtures The Kinetic Molecular Theory of Gases Deviation from Ideal Behavior 2

10. 1 Properties of Gases Topics Characteristics of Gases Gas Pressure: Definition and Units Calculation of Pressure Measurement of Pressure 3

10. 1 Properties of Gases Characteristics of Gases 1. A sample of gas assumes both the shape and volume of its container. 2. Gases are compressible. 3. The densities of gases are much smaller than those of liquids and solids and are highly variable depending on temperature and pressure. 4. Gases form homogeneous mixtures (solutions) with one another in any proportion. 4

10. 1 Properties of Gases Gas Pressure: Definition and Units Pressure is defined as the force applied per unit area: The SI unit of force is the newton (N), where The SI unit of pressure is the pascal (Pa), defined as 1 newton per square meter 5

10. 1 Properties of Gases Gas Pressure: Definition and Units 6

10. 1 Properties of Gases Calculation of Pressure 7

10. 1 Properties of Gases Measurement of Pressure A simple barometer, an instrument used to measure atmospheric pressure Standard atmospheric pressure (1 atm) was originally defined as the pressure that would support a column of mercury exactly 760 mm high at 0°C at sea level. 8

10. 1 Properties of Gases Measurement of Pressure The pressure exerted by a column of fluid is given by 9
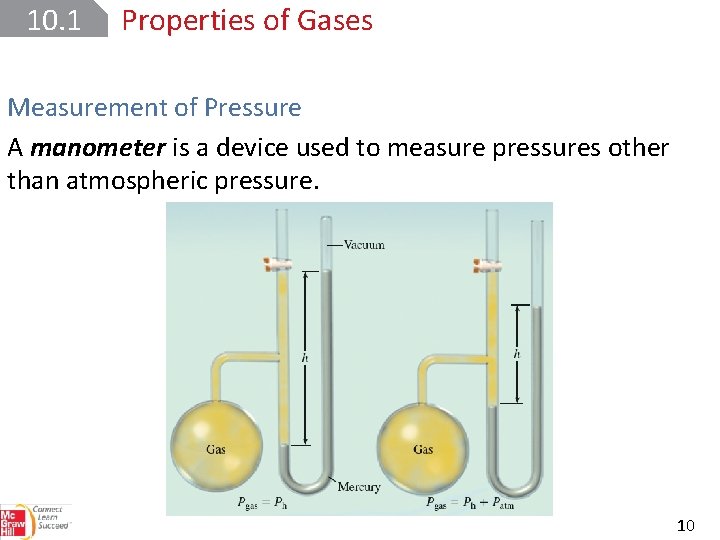
10. 1 Properties of Gases Measurement of Pressure A manometer is a device used to measure pressures other than atmospheric pressure. 10

SAMPLE PROBLEM 10. 1 Calculate the pressure exerted by a column of mercury 70. 0 cm high. Express the pressure in pascals and in atmospheres. The density of mercury is 13. 5951 g/cm 3. Setup Height must be expressed in meters and density must be expressed in kg/m 3. 11

SAMPLE PROBLEM 10. 1 Solution 12

10. 2 The Gas Laws Topics Boyle’s Law: The Pressure-Volume Relationship Charles’s and Gay-Lussac’s Law: The Temperature-Volume Relationship Avogadro’s Law: The Amount-Volume Relationship The Combined Gas Law: The Pressure-Temperature. Amount-Volume Relationship 13
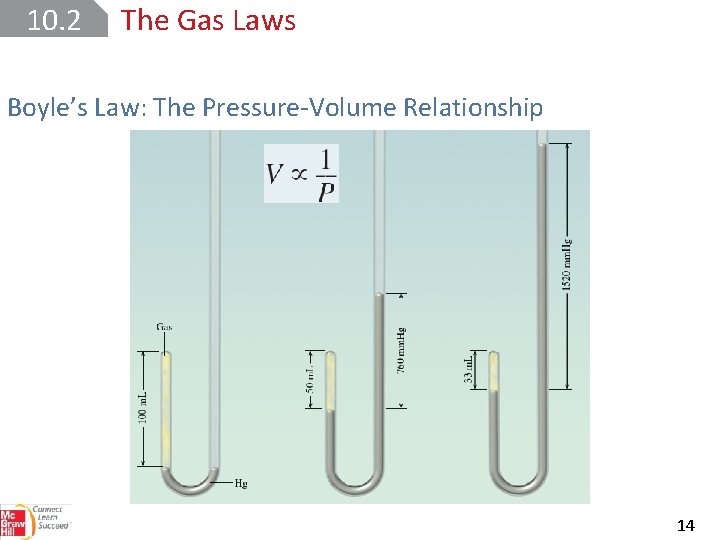
10. 2 The Gas Laws Boyle’s Law: The Pressure-Volume Relationship 14
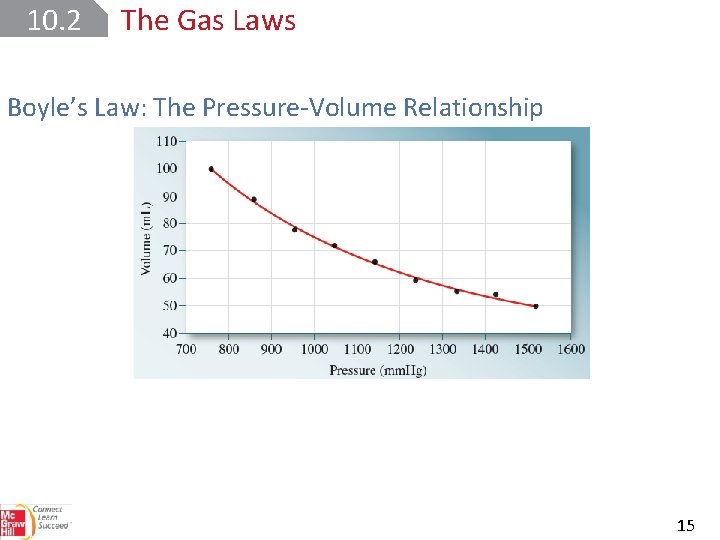
10. 2 The Gas Laws Boyle’s Law: The Pressure-Volume Relationship 15
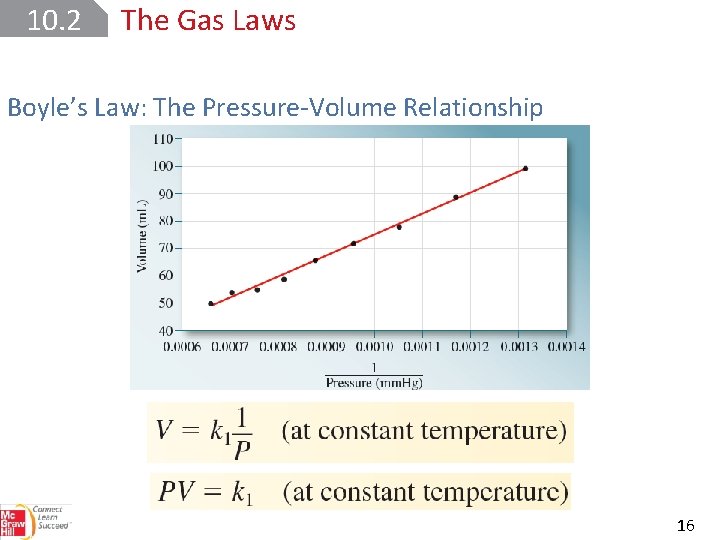
10. 2 The Gas Laws Boyle’s Law: The Pressure-Volume Relationship 16

10. 2 The Gas Laws Boyle’s Law: The Pressure-Volume Relationship 17

SAMPLE PROBLEM 10. 2 If a skin diver takes a breath at the surface, filling his lungs with 5. 82 L of air, what volume will the air in his lungs occupy when he dives to a depth where the pressure is 1. 92 atm? (Assume constant temperature and that the pressure at the surface is exactly 1 atm. ) Setup P 1 = 1. 00 atm, V 1 = 5. 82 L, and P 2 = 1. 92 atm. 18

SAMPLE PROBLEM 10. 2 Solution 19
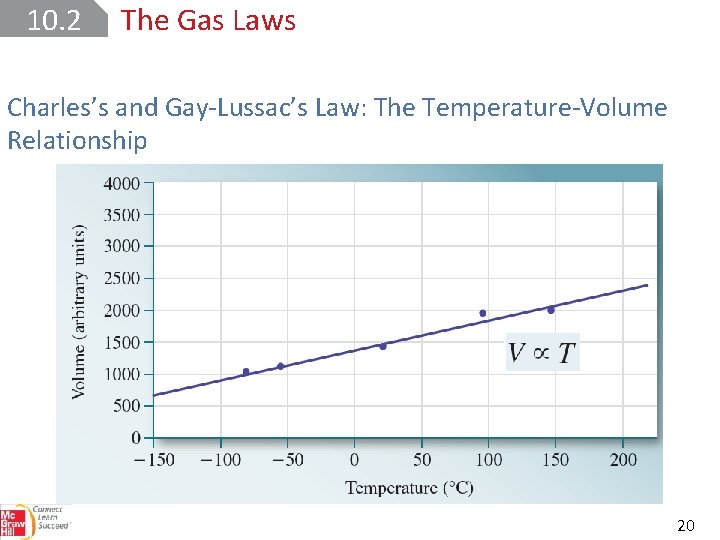
10. 2 The Gas Laws Charles’s and Gay-Lussac’s Law: The Temperature-Volume Relationship 20
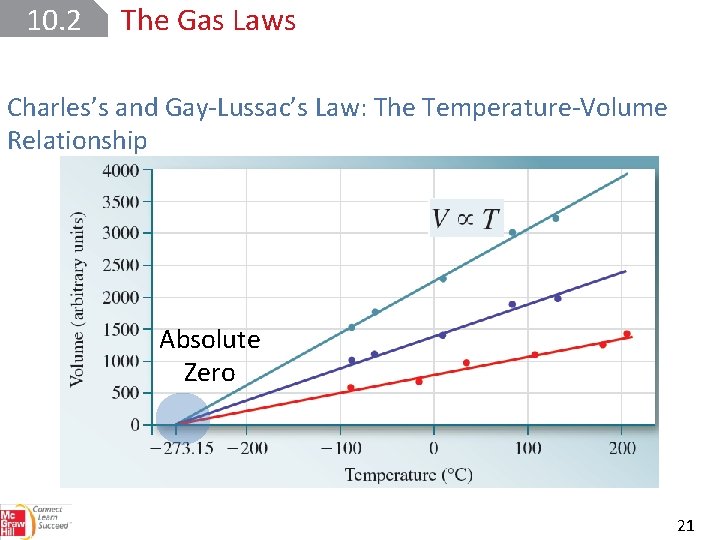
10. 2 The Gas Laws Charles’s and Gay-Lussac’s Law: The Temperature-Volume Relationship Absolute Zero 21

10. 2 The Gas Laws Charles’s and Gay-Lussac’s Law: The Temperature-Volume Relationship All temperatures are in kelvins. 22

SAMPLE PROBLEM 10. 3 A sample of argon gas that originally occupied 14. 6 L at 25. 0°C was heated to 50. 0°C at constant pressure. What is its new volume? Setup T 1 = 298. 15 K, V 1 = 14. 6 L, and T 2 = 323. 15 K Solution 23

10. 2 The Gas Laws Avogadro’s Law: The Amount-Volume Relationship 24
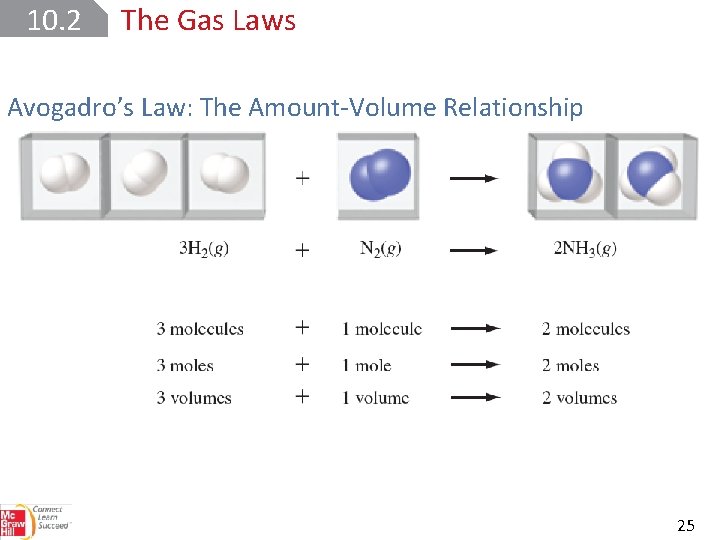
10. 2 The Gas Laws Avogadro’s Law: The Amount-Volume Relationship 25

SAMPLE PROBLEM 10. 4 If we combine 3. 0 L of NO and 1. 5 L of O 2, and they react according to the balanced equation what volume of NO 2 will be produced? (Assume that the reactants and product are all at the same temperature and pressure. ) Strategy Apply Avogadro’s law to determine the volume of a gaseous product. 26

SAMPLE PROBLEM 10. 4 Solution According to the balanced equation, the volume of NO 2 formed will be equal to the volume of NO that reacts. Therefore, 3. 0 L of NO 2 will form. 27

10. 2 The Gas Laws The Combined Gas Law: The Pressure-Temperature-Amount. Volume Relationship For a fixed amount of gas (n 1 = n 2): 28

SAMPLE PROBLEM 10. 5 If a child releases a 6. 25 -L helium balloon in the parking lot of an amusement park where the temperature is 28. 50°C and the air pressure is 757. 2 mm. Hg, what will the volume of the balloon be when it has risen to an altitude where the temperature is – 34. 35°C and the air pressure is 366. 4 mm. Hg? Setup T 1 = 301. 65 K, T 2 = 238. 80 K. 29

SAMPLE PROBLEM 10. 5 Solution 30

10. 3 The Ideal Gas Equation Topics Deriving the Ideal Gas Equation from the Empirical Gas Laws Applications of the Ideal Gas Equation 31

10. 3 The Ideal Gas Equation Deriving the Ideal Gas Equation from the Empirical Gas Laws 32
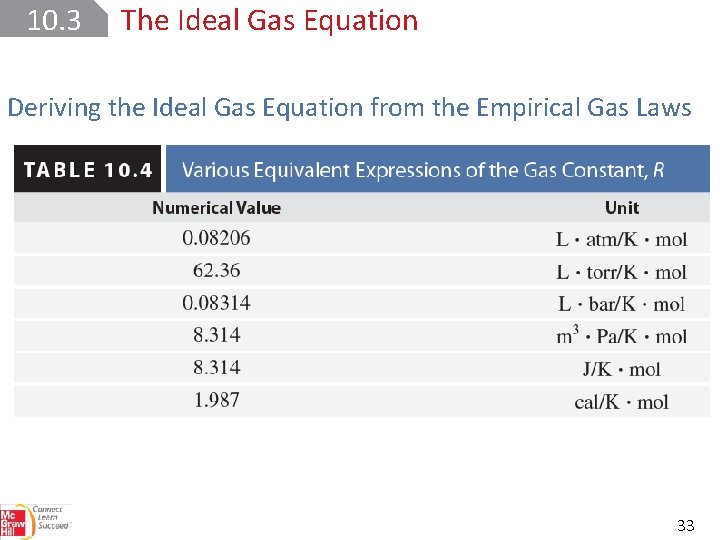
10. 3 The Ideal Gas Equation Deriving the Ideal Gas Equation from the Empirical Gas Laws 33

10. 3 The Ideal Gas Equation Deriving the Ideal Gas Equation from the Empirical Gas Laws The volume of 1 mole of an ideal gas at 0°C and 1 atm (conditions known as standard temperature and pressure (STP)) is : 34

SAMPLE PROBLEM 10. 6 Calculate the volume of a mole of ideal gas at room temperature (25°C) and 1 atm. Setup The data given are n = 1 mol, T = 298. 15 K, and P = 1. 00 atm. Because the pressure is expressed in atmospheres, we use R = 0. 08206 L · atm/K · mol to solve for volume in liters. 35

SAMPLE PROBLEM 10. 6 Solution 36

10. 3 The Ideal Gas Equation Applications of the Ideal Gas Equation 37

SAMPLE PROBLEM 10. 7 Carbon dioxide is effective in fire extinguishers partly because its density is greater than that of air, so CO 2 can smother the flames by depriving them of oxygen. (Air has a density of approximately 1. 2 g/L at room temperature and 1 atm. ) Calculate the density of CO 2 at room temperature (25°C) and 1. 0 atm. Setup The molar mass of CO 2 is 44. 01 g/mol. 38

SAMPLE PROBLEM 10. 7 Solution 39

10. 4 Reactions with Gaseous Reactants and Products Topics Calculating the Required Volume of a Gaseous Reactant Determining the Amount of Reactant Consumed Using Change in Pressure Predicting the Volume of a Gaseous Product 40

10. 4 Reactions with Gaseous Reactants and Products Calculating the Required Volume of a Gaseous Reactant At constant pressure and temperature: 41

10. 4 Reactions with Gaseous Reactants and Products Calculating the Required Volume of a Gaseous Reactant 42

SAMPLE PROBLEM 10. 9 Sodium peroxide (Na 2 O 2) is used to remove carbon dioxide from (and add oxygen to) the air supply in spacecrafts. It works by reacting with CO 2 in the air to produce sodium carbonate (Na 2 CO 3) and O 2. What volume (in liters) of CO 2 (at STP) will react with a kilogram of Na 2 O 2? 43

SAMPLE PROBLEM 10. 9 Setup The molar mass of Na 2 O 2 is 77. 98 g/mol (1 kg = 1000 g). (Treat the specified mass of Na 2 O 2 as an exact number. ) Solution 44

10. 4 Reactions with Gaseous Reactants and Products Determining the Amount of Reactant Consumed Using Change in Pressure 45

SAMPLE PROBLEM 10. 10 Another air-purification method for enclosed spaces involves the use of “scrubbers” containing aqueous lithium hydroxide, which reacts with carbon dioxide to produce lithium carbonate and water: Consider the air supply in a submarine with a total volume of 2. 5 × 105 L. The pressure is 0. 9970 atm, and the temperature is 25°C. If the pressure in the submarine drops to 0. 9891 atm as the result of carbon dioxide being consumed by an aqueous lithium hydroxide scrubber, how many moles of CO 2 are consumed? 46

SAMPLE PROBLEM 10. 10 Setup P = 0. 9970 atm – 0. 9891 atm = 7. 9 × 10– 3 atm. According to the problem statement, V = 2. 5 × 105 L and T = 298. 15 K. For problems in which P is expressed in atmospheres and V in liters, use R = 0. 08206 L · atm/K · mol. 47

SAMPLE PROBLEM 10. 10 Solution 48

10. 4 Reactions with Gaseous Reactants and Products Predicting the Volume of a Gaseous Product Using a combination of stoichiometry and the ideal gas equation, we can calculate the volume of gas that we expect to be produced in a chemical reaction. 49

SAMPLE PROBLEM 10. 11 The air bags in cars are inflated when a collision triggers the explosive, highly exothermic decomposition of sodium azide (Na. N 3): A typical driver-side air bag contains about 50 g of Na. N 3. Determine the volume of N 2 gas that would be generated by the decomposition of 50. 0 g of sodium azide at 85. 0°C and 1. 00 atm. 50

SAMPLE PROBLEM 10. 11 Setup The molar mass of Na. N 3 is 65. 02 g/mol. Solution 51

10. 5 Gas Mixtures Topics Dalton’s Law of Partial Pressures Mole Fractions Using Partial Pressures to Solve Problems 52

10. 5 Gas Mixtures Dalton’s Law of Partial Pressures Dalton’s law of partial pressures states that the total pressure exerted by a gas mixture is the sum of the partial pressures exerted by each component of the mixture: 53

SAMPLE PROBLEM 10. 12 A 1. 00 -L vessel contains 0. 215 mole of N 2 gas and 0. 0118 mole of H 2 gas at 25. 5°C. Determine the partial pressure of each component and the total pressure in the vessel. Setup T = 298. 65 K. 54

SAMPLE PROBLEM 10. 12 Solution 55

10. 5 Gas Mixtures Mole Fractions 1. The mole fraction of a mixture component is always less than 1. 2. The sum of mole fractions for all components of a mixture is always 1. 3. Mole fraction is dimensionless. 56

10. 5 Gas Mixtures Mole Fractions 57

SAMPLE PROBLEM 10. 13 Calculate the mole fraction of NO in a 10. 00 -L gas cylinder at room temperature (25°C) that contains 6. 022 mol N 2 and in which the total pressure is 14. 75 atm. Setup The temperature is 298. 15 K. 58

SAMPLE PROBLEM 10. 13 Solution 59
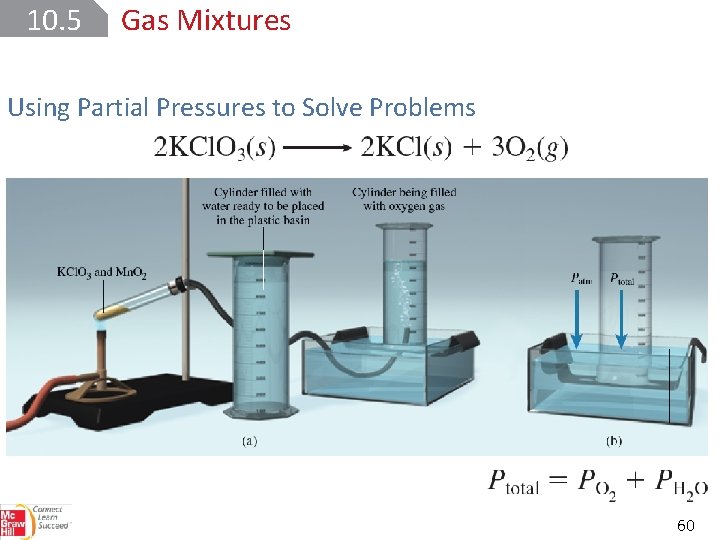
10. 5 Gas Mixtures Using Partial Pressures to Solve Problems 60
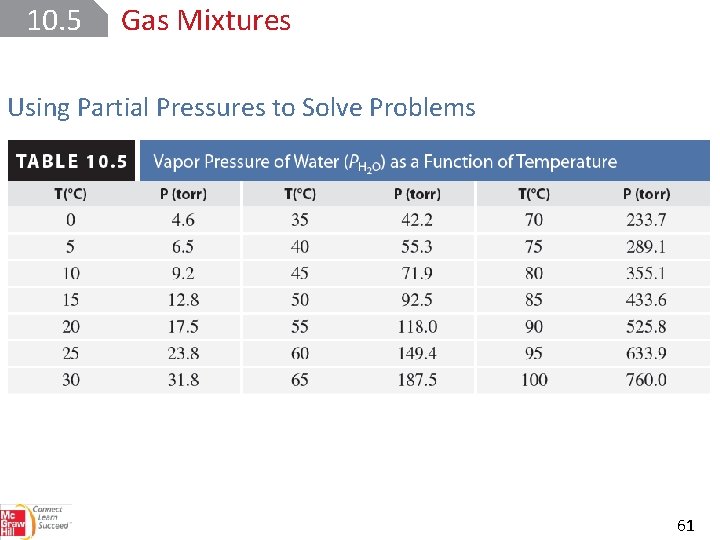
10. 5 Gas Mixtures Using Partial Pressures to Solve Problems 61

SAMPLE PROBLEM 10. 14 Calcium metal reacts with water to produce hydrogen gas : Determine the mass of H 2 produced at 25°C and 0. 967 atm when 525 m. L of the gas is collected over water Setup V = 0. 525 L and T = 298. 15 K. The partial pressure of water at 25°C is 23. 8 torr or 23. 8 torr · (1 atm/760 torr) = 0. 0313 atm. The molar mass of H 2 is 2. 016 g/mol. 62

SAMPLE PROBLEM 10. 14 Solution 63

10. 6 The Kinetic Molecular Theory of Gases Topics Application to the Gas Laws Molecular Speed Diffusion and Effusion 64

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws The kinetic molecular theory explains how the molecular nature of gases gives rise to their macroscopic properties. The basic assumptions of the kinetic molecular theory are as follows: 1. A gas is composed of particles that are separated by relatively large distances. The volume occupied by individual molecules is negligible. 65

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws 2. Gas molecules are constantly in random motion, moving in straight paths, colliding with the walls of their container and with one another in perfectly elastic collisions. (Energy is transferred but not lost in the collisions. ) 3. Gas molecules do not exert attractive or repulsive forces on one another. 66

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws 4. The average kinetic energy, of gas molecules in a sample is proportional to the absolute temperature: The mean square speed : 67

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws Kinetic molecular theory enables us to understand some of the properties and behavior of gases in the following ways. Compressibility Gases are compressible because molecules in the gas phase are separated by large distances (assumption 1) and can be moved closer together by decreasing the volume occupied by a sample. 68

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws Boyle’s Law (V 1/P) The pressure exerted by a gas is the result of the collisions of gas molecules with the walls of their container (assumption 2). The magnitude of the pressure depends on both the frequency of collision and the speed of molecules when they collide with the walls. Decreasing the volume occupied by a sample of gas increases the frequency of these collisions, thus increasing the pressure. 69

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws Charles’s Law (V T) Heating a sample of gas increases its average kinetic energy (assumption 4). An increase in average kinetic energy must be accompanied by an increase in the mean square speed of the molecules. In other words, heating a sample of gas makes the gas molecules move faster. 70

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws Avogadro’s Law (V n) Because the magnitude of the pressure exerted by a sample of gas depends on the frequency of the collisions with the container wall, the presence of more molecules would cause an increase in pressure. 71

10. 6 The Kinetic Molecular Theory of Gases Application to the Gas Laws Dalton’s Law of Partial Pressures (Ptotal Pi) Gas molecules do not attract or repel one another (assumption 3), so the pressure exerted by one gas is unaffected by the presence of another gas. Consequently, the total pressure exerted by a mixture of gases is simply the sum of the partial pressures of the individual components in the mixture 72

10. 6 The Kinetic Molecular Theory of Gases Molecular Speed root-mean-square (rms) speed (urms). 73
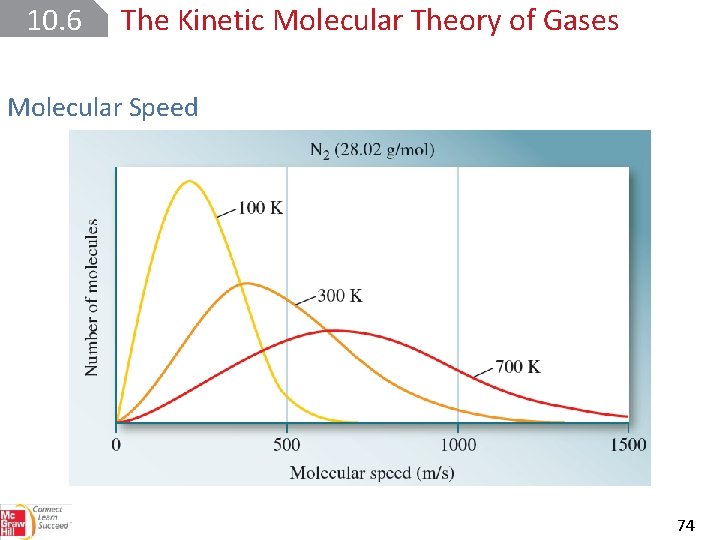
10. 6 The Kinetic Molecular Theory of Gases Molecular Speed 74
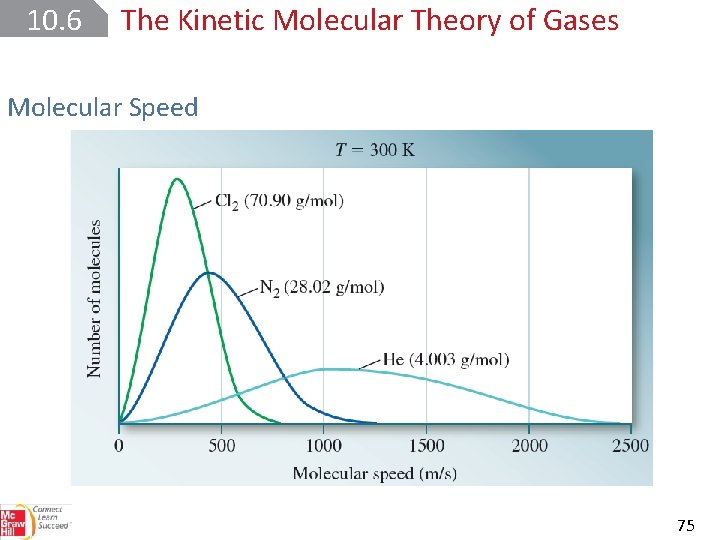
10. 6 The Kinetic Molecular Theory of Gases Molecular Speed 75

10. 6 The Kinetic Molecular Theory of Gases Molecular Speed 76

SAMPLE PROBLEM 10. 16 Determine how much faster a helium atom moves, on average, than a carbon dioxide molecule at the same temperature. Setup The molar masses of He and CO 2 are 4. 003 and 44. 01 g/mol, respectively. 77

SAMPLE PROBLEM 10. 16 Solution On average, He atoms move 3. 316 times faster than CO 2 molecules at the same temperature. 78
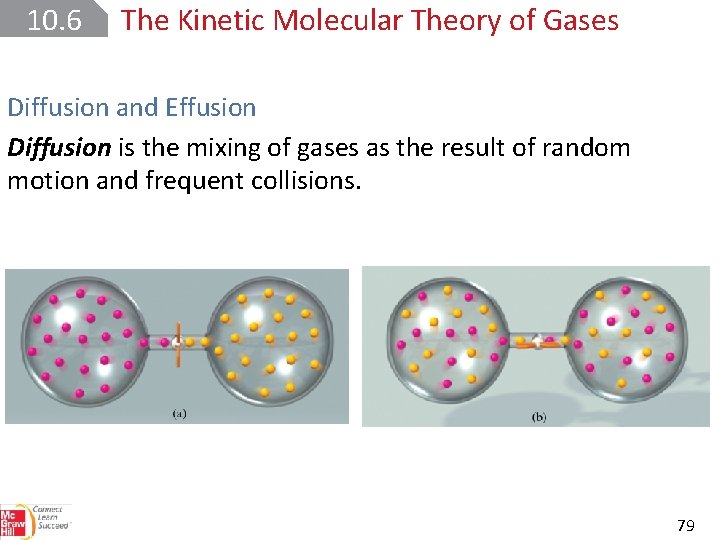
10. 6 The Kinetic Molecular Theory of Gases Diffusion and Effusion Diffusion is the mixing of gases as the result of random motion and frequent collisions. 79
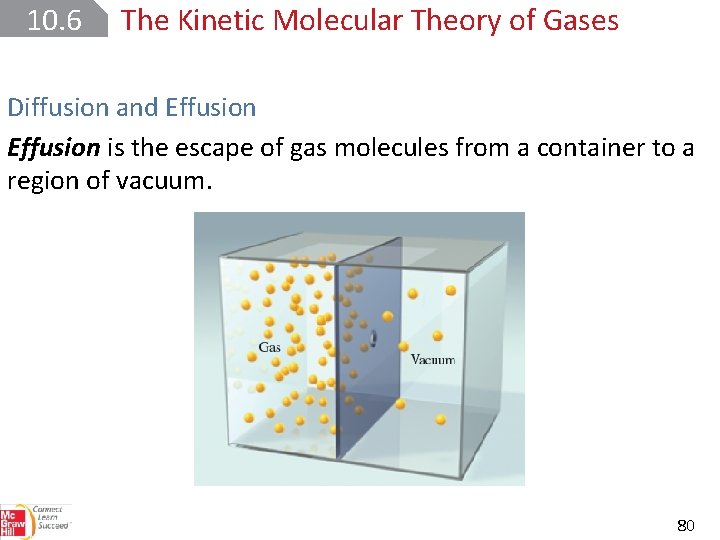
10. 6 The Kinetic Molecular Theory of Gases Diffusion and Effusion is the escape of gas molecules from a container to a region of vacuum. 80

10. 6 The Kinetic Molecular Theory of Gases Diffusion and Effusion Graham’s law states that the rate of diffusion or effusion of a gas is inversely proportional to the square root of its molar mass: 81

10. 7 Deviation from Ideal Behavior Topics Factors That Cause Deviation from Ideal Behavior The van der Waals Equation 82

10. 7 Deviation from Ideal Behavior Factors That Cause Deviation from Ideal Behavior At high pressures, gas molecules are relatively close together. We can assume that gas molecules occupy no volume only when the distances between molecules are large. When the distances between molecules are reduced, the volume occupied by each individual molecule becomes more significant. 83

10. 7 Deviation from Ideal Behavior Factors That Cause Deviation from Ideal Behavior At low temperatures, gas molecules are moving more slowly. We can assume that there are no intermolecular forces between gas molecules, either attractive or repulsive, when the gas molecules are moving very fast and the magnitude of their kinetic energies is much larger than the magnitude of any intermolecular forces. When molecules move more slowly, they have lower kinetic energies and the magnitude of the forces between them becomes more significant. 84
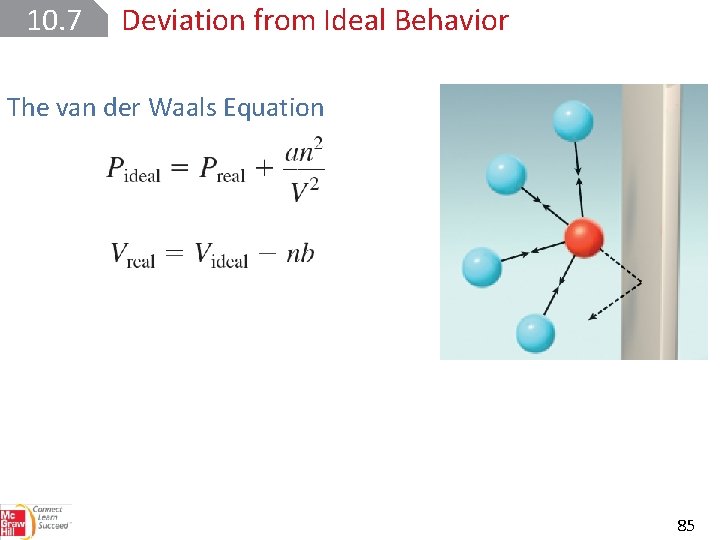
10. 7 Deviation from Ideal Behavior The van der Waals Equation 85

10. 7 Deviation from Ideal Behavior The van der Waals Equation 86
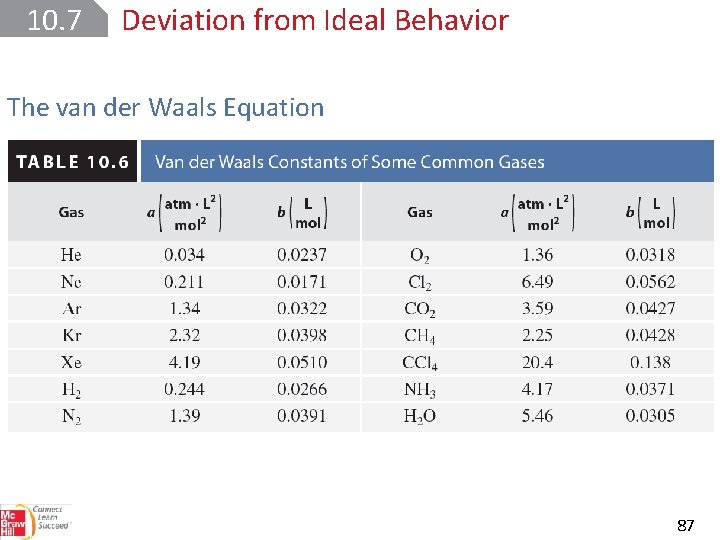
10. 7 Deviation from Ideal Behavior The van der Waals Equation 87

SAMPLE PROBLEM 10. 17 A sample of 3. 50 moles of NH 3 gas occupies 5. 20 L at 47°C. Calculate the pressure of the gas (in atm) using (a) the ideal gas equation and (b) the van der Waals equation. Setup T = 320. 15 K, a = 4. 17 atm · L/mol 2, and b = 0. 0371 L/mol. 88

SAMPLE PROBLEM 10. 17 Solution 89

10. 7 Deviation from Ideal Behavior The van der Waals Equation One way to measure a gas’s deviation from ideal behavior is to determine its compressibility factor, Z, where Z = PV/RT. For one mole of an ideal gas, Z is equal to 1 at all pressures and temperatures. 90
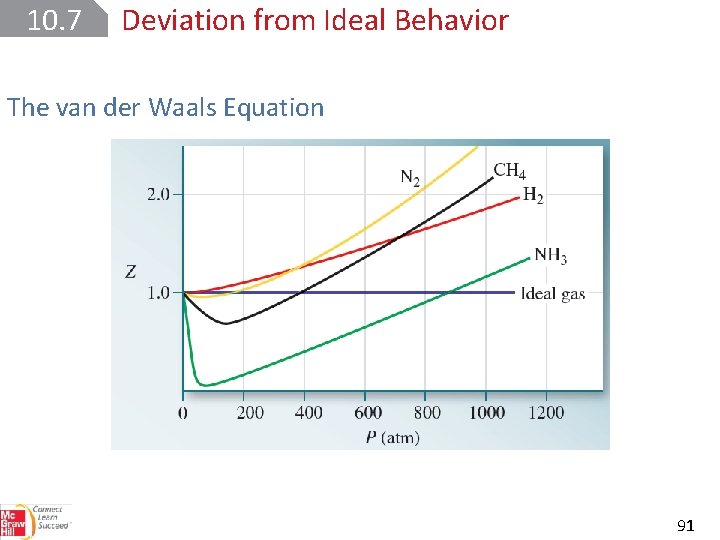
10. 7 Deviation from Ideal Behavior The van der Waals Equation 91
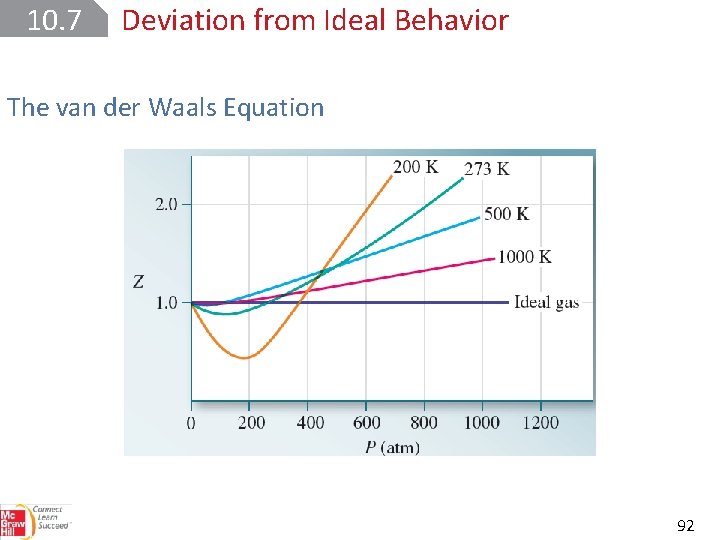
10. 7 Deviation from Ideal Behavior The van der Waals Equation 92
- Slides: 92