Chemistry The study of matter and the changes





















- Slides: 25

Chemistry: “The study of matter and the changes it undergoes” Classifying Matter and the Changes Matter Undergoes

Types of Changes Matter can Undergo: �Physical – a change in a substance that does NOT alter its fundamental properties (e. g. boiling point, melting point, color, chemical reactivity)

Examples of Physical Changes • Phase change such as boiling or melting • Formation of a solution (salt water)

Examples of Physical Changes • Breaking or tearing

Types of Changes Matter can Undergo: �Chemical – a change in a substance that does alter its fundamental properties (b. p. , m. p. color, chemical reactivity)

Examples of Chemical Changes • sodium + chlorine → sodium chloride • Burning wood

Video links • Link to breaking glass • Reaction of sodium and chlorine • Note: Color change and/or dramatic difference in appearance and/or large energy changes usually indicates chemical change.

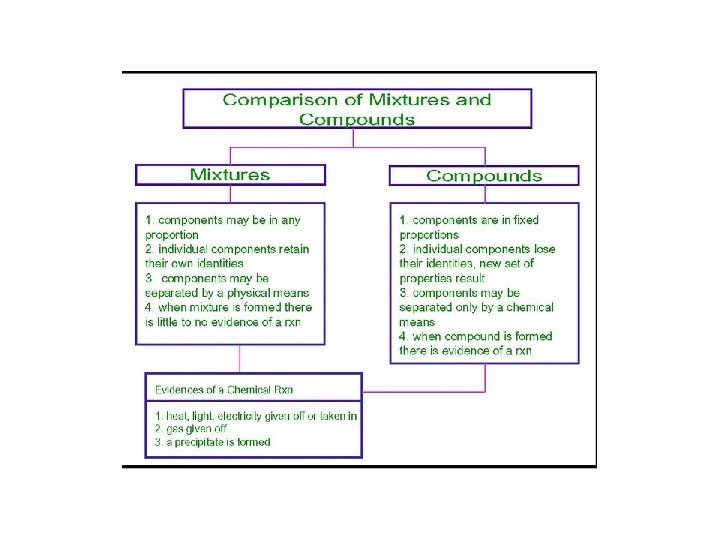
Importance of Chemical vs Physical Processes in classifying compound vs mixture • Substances that contain more than one type of atom are classified as either a compound (if the atoms can only be separated by a chemical reaction) or a mixture (if the atoms can only be separated by a physical process. .

Homework 1 -1 : Classify each change as chemical (C) or physical (P) • p. 67 11, 12, 18 abcdejk • 11) Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these characteristics of mercury physical or chemical properties? PHYSICAL • 12) If liquid elemental mercury is heated in oxygen, the volume of the shiny liquid decreases and a reddish orange solids forms in its place. Do these characteristics represent a chemical change or a physical change? CHEMICAL •

Homework 1 -1 18 abcde • 18 a) A fireplace poker glows red when you heat it in the fire. P • 18 b) A marshmallow gets black when toasted too long in a campfire. C • 18 c) Hydrogen peroxide dental strips will make your teeth whiter. C • 18 d) If you wash your jeans with chlorine bleach, they will fade. C

Homework 1 -1 18 ejk • 18 e) If you spill some nail polish remover on your skin it will evaporate quickly. P • 18 j) A log of wood is chopped up with an axe into smaller pieces of wood. P • 18 k) A log of wood is burned in the fire place. C

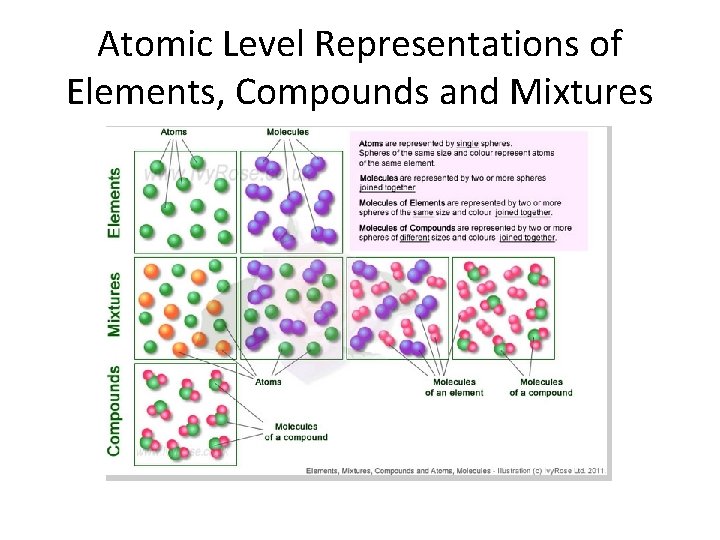
Classifying Matter � ELEMENT – a substance composed of atoms of the SAME type � COMPOUND – a substance composed of two or more different types of elements CHEMICALLY BONDED together � MIXTURE – a substance composed of two or more different substances that can only be separated by PHYSICAL means

Classification of Matter Atom – smallest chemical building block Molecule – 2 or more atoms bonded together Homogeneous – uniform throughout Heterogeneous – different – nonuniform throughout

Atomic Level Representations of Elements, Compounds and Mixtures

ELEMENT – substance composed of 1 type of atom • Cannot be broken down into simpler substance by either chemical or physical process • Element symbols are listed on periodic table

Examples of Elements • Hydrogen (H): • Copper (Cu): • Oxygen (O): • Iron (Fe):

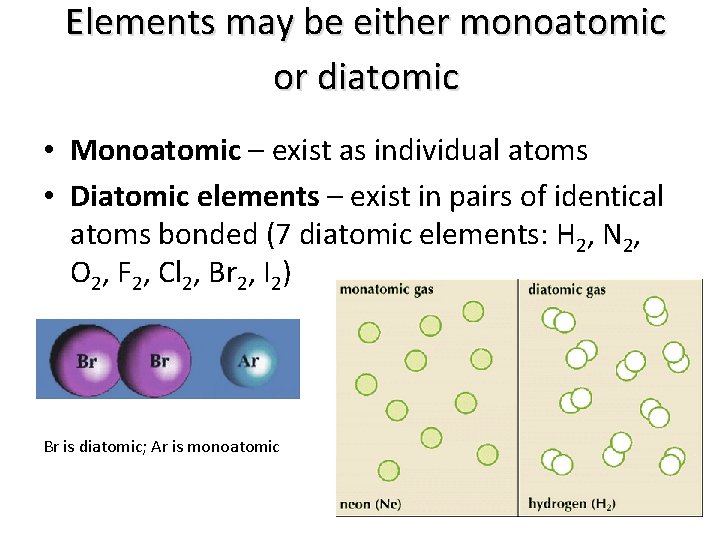
Elements may be either monoatomic or diatomic • Monoatomic – exist as individual atoms • Diatomic elements – exist in pairs of identical atoms bonded (7 diatomic elements: H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2) Br is diatomic; Ar is monoatomic

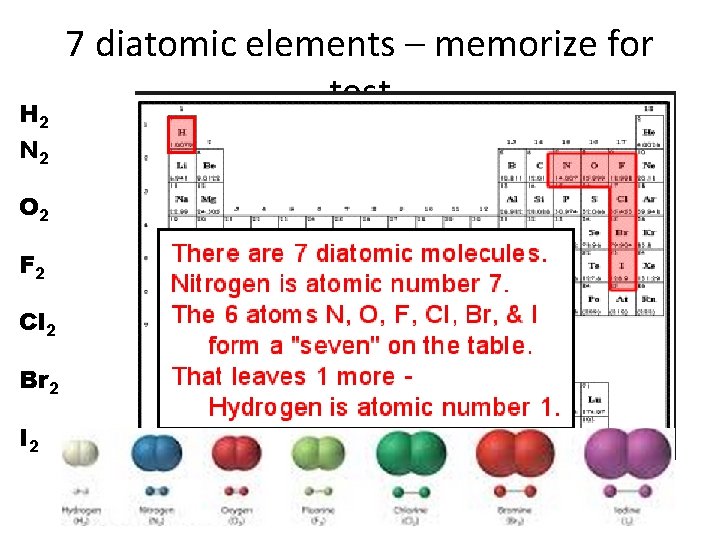
H 2 N 2 O 2 F 2 Cl 2 Br 2 I 2 7 diatomic elements – memorize for test

Compounds – composed of two or more different types of elements chemically bonded together • Compounds can only be separated into the elements that make up the substance by a CHEMICAL REACTION.

Examples of Compounds • 1) Water: H 2 O ATOMIC LEVEL PICTURE • 2) Salt: Na. Cl • 3) Sugar: C 6 H 12 O 6

MIXTURE – Composed of two or more substances which can only be separated by a PHYSICAL PROCESS • Most of the substances we see around us are mixtures • Examples of mixtures • • Salt water Oil and water Apple

Mixtures can be combinations of elements or compounds • Mixture of elements: • Mixture of elements and compounds • Mixture of compounds:


Which of the following are elements and which are compounds? • How many atoms are present? • How many molecules are present?

Which of the following are elements and which are compounds? • How many atoms are present? • How many molecules are present? 1) 2 atoms; 1 molecule 2) 3 atoms; 1 molecule 3) 2 atoms; 1 molecule 4) 5 atoms; 1 molecule 5) 1 atom; 0 molecules Total: 13 atoms; 4 molecules