Chemistry The Central Science 10 th edition Theodore











- Slides: 27

Chemistry, The Central Science, 10 th edition Theodore L. Brown; H. Eugene Le. May, Jr. ; and Bruce E. Bursten Review Unit 1 (Chp 6, 7): Atoms, Electrons, & Periodicity John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall, Inc.

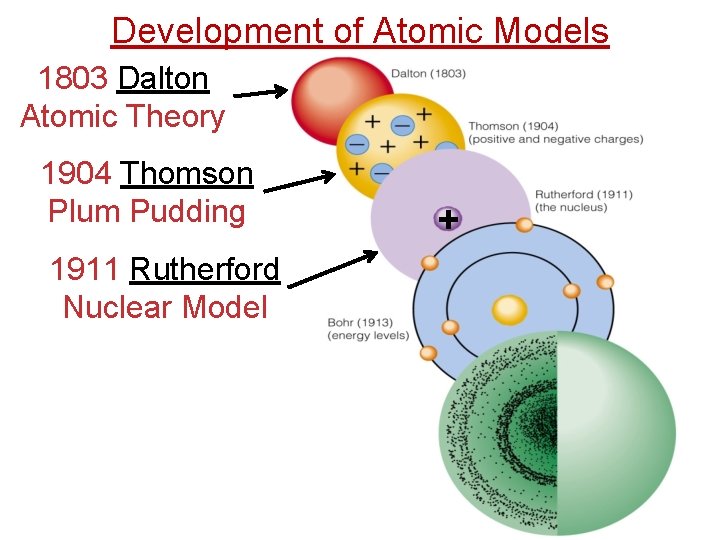
Development of Atomic Models 1803 Dalton Atomic Theory 1904 Thomson Plum Pudding 1911 Rutherford Nuclear Model +

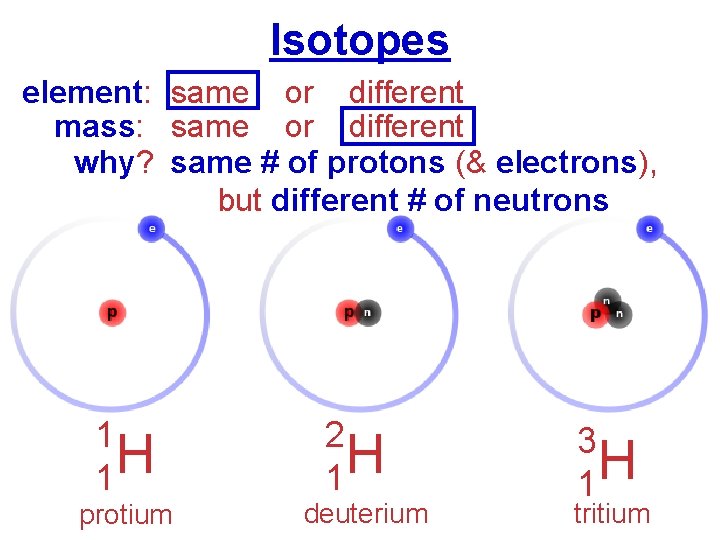
Isotopes element: same or different mass: same or different why? same # of protons (& electrons), but different # of neutrons 1 H 1 protium 2 H 1 deuterium 3 H 1 tritium

Average Atomic Mass • average atomic mass: calculated as a weighted average of isotopes by their relative abundances. • lithium-6 (6. 015 amu), which has a relative abundance of 7. 50%, and • lithium-7 (7. 016 amu), which has a relative abundance of 92. 5%. (6. 015)(0. 0750) + (7. 016)(0. 925) = 6. 94 amu Avg. Mass = (Mass 1)(%) + (Mass 2)(%) …

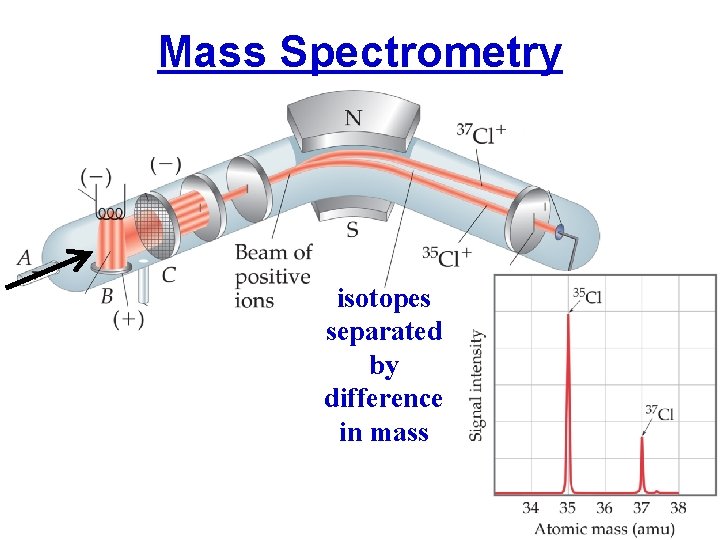
Mass Spectrometry isotopes separated by difference in mass

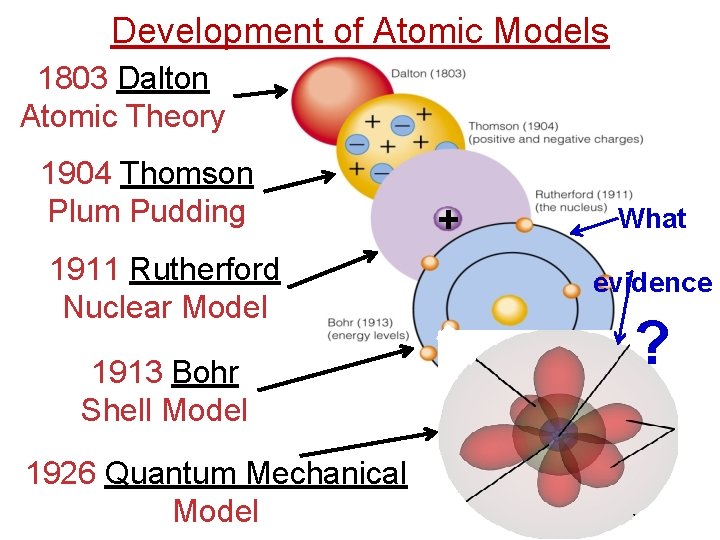
Development of Atomic Models 1803 Dalton Atomic Theory 1904 Thomson Plum Pudding 1911 Rutherford Nuclear Model 1913 Bohr Shell Model 1926 Quantum Mechanical Model + What evidence ?

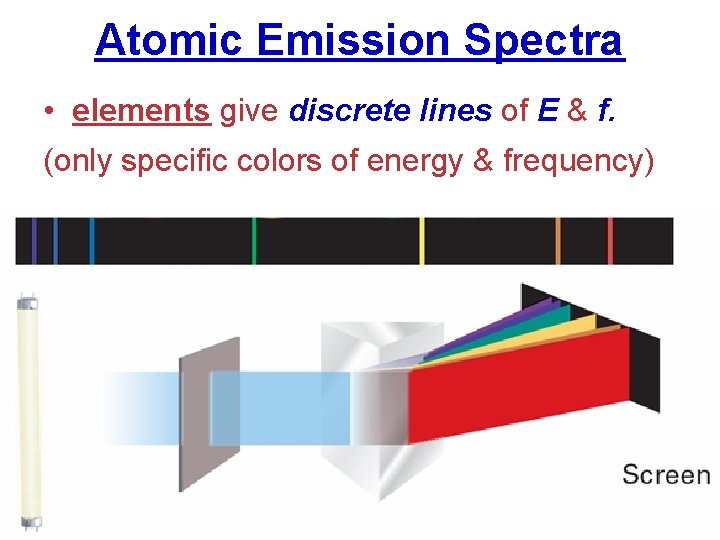
5. 3 Atomic Emission Spectra • elements give discrete lines of E & f. (only specific colors of energy & frequency)

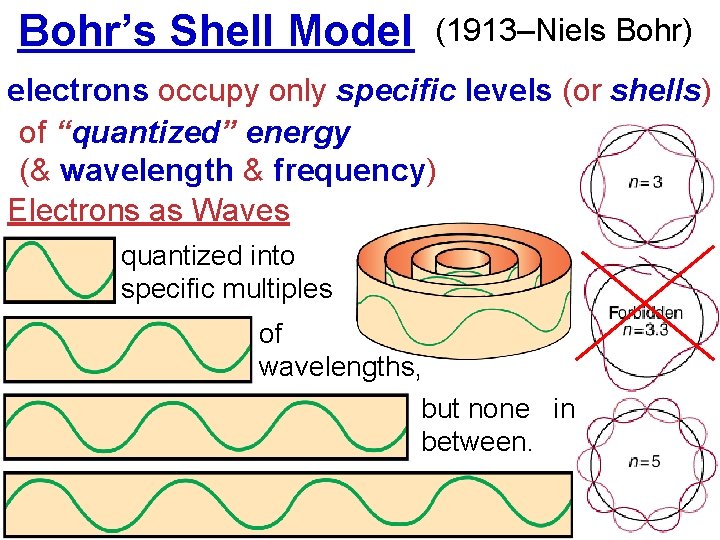
Bohr’s Shell Model (1913–Niels Bohr) electrons occupy only specific levels (or shells) of “quantized” energy (& wavelength & frequency) Electrons as Waves quantized into specific multiples of wavelengths, but none in between.

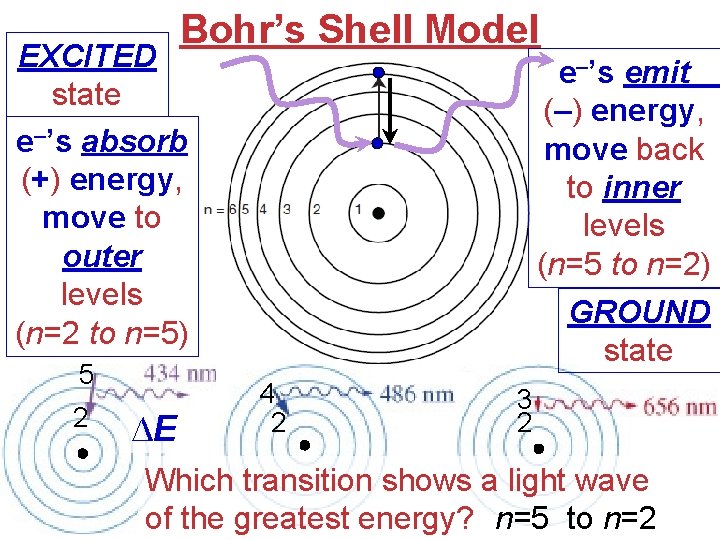
Bohr’s Shell Model EXCITED state e–’s absorb (+) energy, move to outer levels (n=2 to n=5) 5 2 ∆E e–’s emit (–) energy, move back to inner levels (n=5 to n=2) GROUND state 4 2 3 2 Which transition shows a light wave of the greatest energy? n=5 to n=2

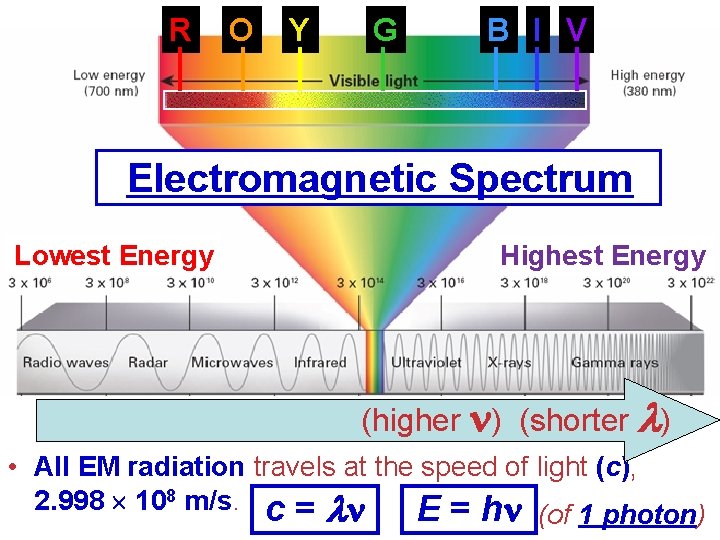
R O Y G B I V Electromagnetic Spectrum Lowest Energy Highest Energy (higher ) (shorter ) • All EM radiation travels at the speed of light (c), 2. 998 108 m/s. c = E = h (of 1 photon)

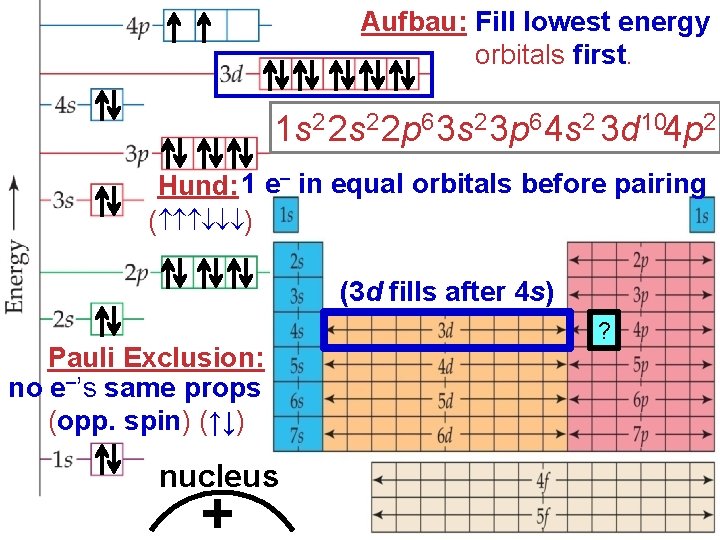
Aufbau: Fill lowest energy orbitals first. 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 104 p 2 Hund: 1 e– in equal orbitals before pairing ( ) (3 d fills after 4 s) Pauli Exclusion: no e–’s same props (opp. spin) (↑↓) nucleus + ?

• Paramagnetic: species are attracted by a magnet (caused by unpaired electrons). Fe: [Ar] ↑↓ ↑↓ ↑ ↑ 4 s 3 d • Diamagnetic: species are slightly repelled by magnets (caused by all paired electrons) Zn: [Ar] ↑↓ ↑↓ ↑↓ 4 s 3 d

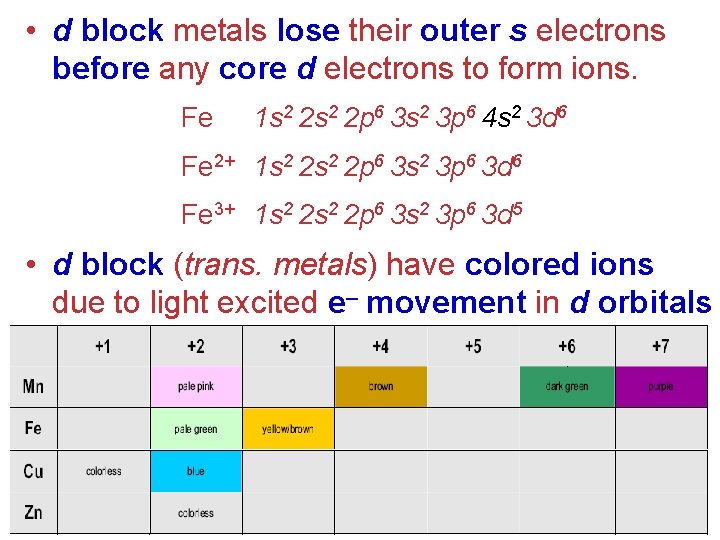
• d block metals lose their outer s electrons before any core d electrons to form ions. Fe 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 Fe 2+ 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6 Fe 3+ 1 s 2 2 p 6 3 s 2 3 p 6 3 d 5 • d block (trans. metals) have colored ions due to light excited e– movement in d orbitals

Other Aspects List 3 species isoelectronic with Ca 2+ & S 2–. P 3– , Cl– , Ar, K+ , Sc 3+ , Ti 4+, V 5+, Cr 6+, Mn 7+ Arrange the following species by increasing size: Ar, K+, Ca 2+, S 2–, Cl– Ca 2+ < K+ < Ar < Cl– < S 2–

Spectroscopy EM REGION SPECTROSCOPIC TECHNIQUE TV/Radio (Rf) Nuclear magnetic resonance (NMR) Infrared Visible/UV X-ray IR APPLICATION Molecular Structure by changes in nuclear spin. Molecular Structure by different bond vibrations UV-Vis, Atomic Emission Spectra Electron Transitions (lines of frequencies/colors) Photoelectron spectroscopy (PES) between energy levels Electronic Structure in an atom (configuration)

Relative # of e–’s Photoelectron Spectroscopy (PES) Which peak is H and which is He? higher peak = more e–’s 1 s 2 He 1 s 1 H 6 5 4 3 2 1 0 Binding Energy. . . or Ionization Energy (MJ/mol) (required to remove e–’s) further left = more energy required (stronger attraction due to more protons)

Relative # of e–’s Photoelectron Spectroscopy (PES) Which peak is H and which is He? 6 2 p – Ne ? higher peak = more e ’s 1 s 2 Identify the 1 He 1 s 2 2 s element H & e-config 6 5 4 3 2 1 0 Binding Energy. . . or Ionization Energy (MJ/mol) (required to remove e–’s) further left = more energy required (stronger attraction due to more protons)

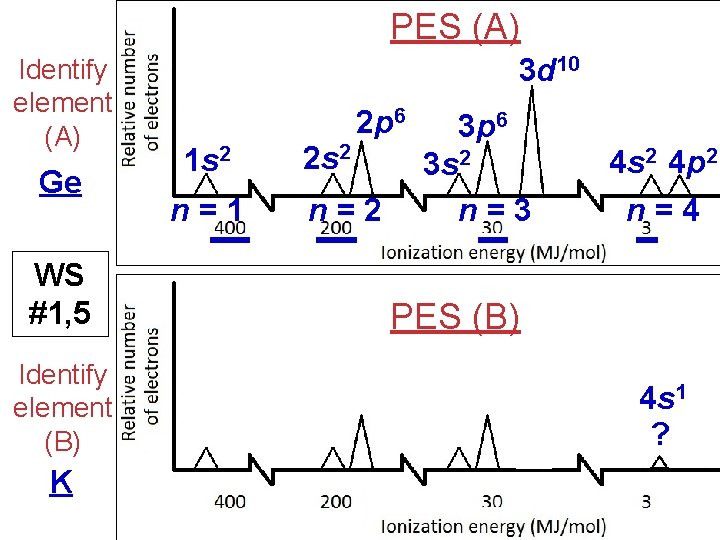
PES (A) Identify element (A) Ge WS #1, 5 Identify element (B) K 3 d 10 2 p 6 1 s 2 n=1 2 s 2 n=2 3 p 6 3 s 2 n=3 4 s 2 4 p 2 n=4 PES (B) 4 s 1 ?

Chemistry, The Central Science, 10 th edition Theodore L. Brown; H. Eugene Le. May, Jr. ; and Bruce E. Bursten Review Unit 1 (Chp 7): Periodicity …or… Periodic Trends in Atomic Properties John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall, Inc.

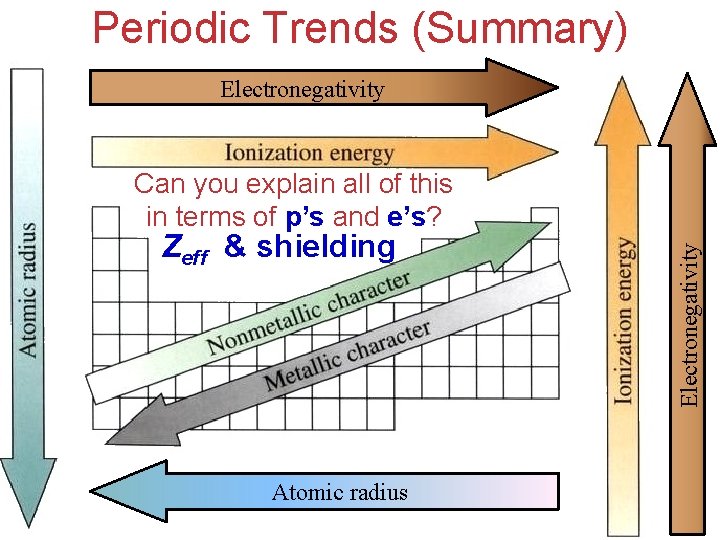
Periodic Trends • We will explain observed trends in size ØAtomic (and Ionic) Radius lose e– ØIonization energy attract e– ØElectronegativity Zeff & shielding (explains all periodic trends and properties)

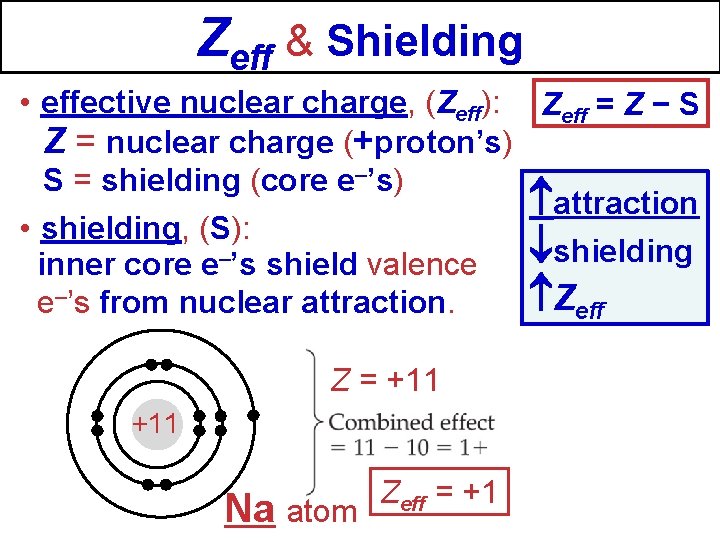
Zeff & Shielding • effective nuclear charge, (Zeff): Zeff = Z − S Z = nuclear charge (+proton’s) S = shielding (core e–’s) attraction • shielding, (S): shielding inner core e–’s shield valence Zeff e–’s from nuclear attraction. Z = +11 Na atom Zeff = +1

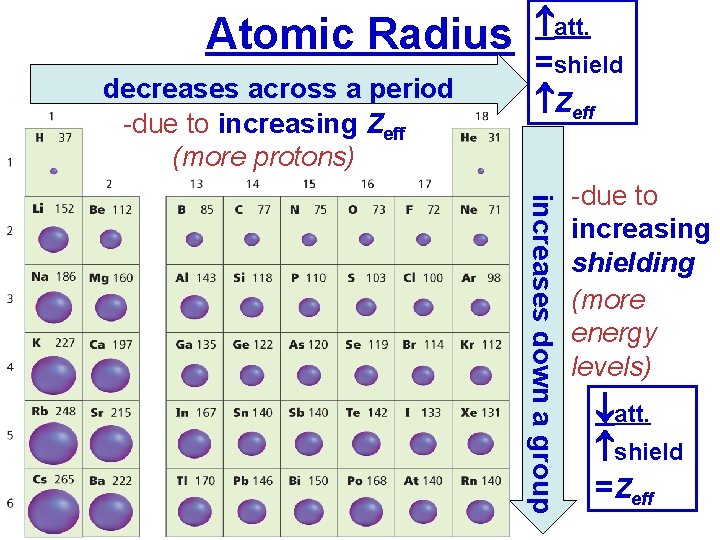
Atomic Radius decreases across a period -due to increasing Zeff (more protons) att. =shield Zeff increases down a group -due to increasing shielding (more energy levels) att. shield =Zeff

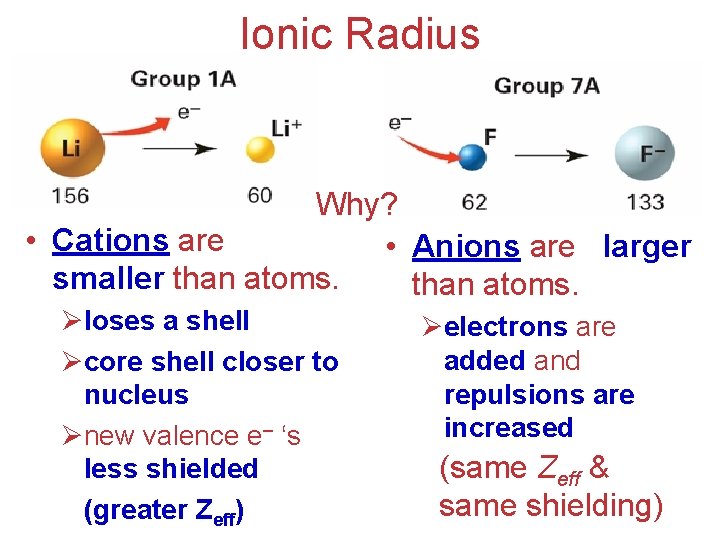
Ionic Radius Why? • Cations are • Anions are larger smaller than atoms. Øloses a shell Øcore shell closer to nucleus Ønew valence e– ‘s less shielded (greater Zeff) Øelectrons are added and repulsions are increased (same Zeff & same shielding)

Ionization Energy (IE) • • energy required to remove an electron more energy to remove each electron IE 1 < IE 2 < IE 3, … look for a huge jump in IE once all valence e–’s are removed, the next e– is on an inner level with attraction ( shielding & Zeff). huge jump in IE 4 b/c 4 th e– on inner level (must have 3 valence e–’s)

Trends in First IE IE tends to… increases across a period decreases down a group -due to increasing Zeff (more protons) att. =shield Zeff -due to increasing shielding (more energy levels) att. shield =Zeff

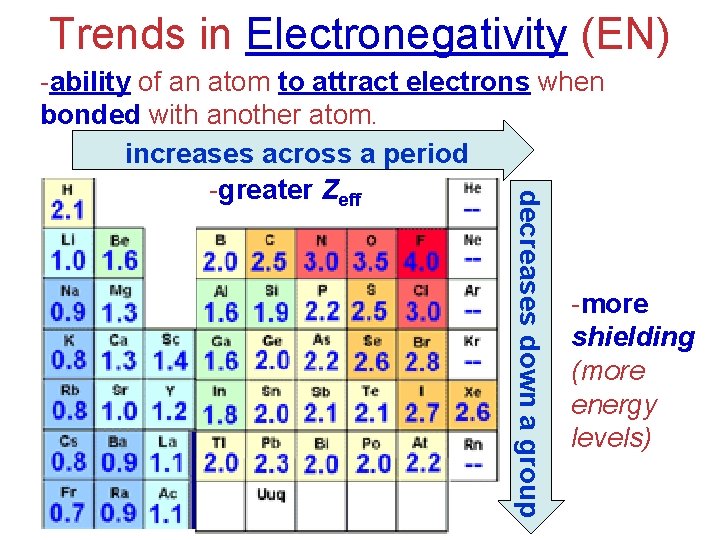
Trends in Electronegativity (EN) decreases down a group -ability of an atom to attract electrons when bonded with another atom. increases across a period -greater Zeff -more shielding (more energy levels)

Periodic Trends (Summary) Electronegativity Zeff & shielding Atomic radius Electronegativity Can you explain all of this in terms of p’s and e’s?